Abstract
Background
Epidemiological studies reported inconsistent results on the associations between maternal caffeine intake during pregnancy and risk of low birth weight (LBW) and childhood overweight and obesity in their offspring.
Methods
We conducted a meta-analysis of cohort studies to quantitatively assess these associations. Pertinent studies were identified by searching PubMed and Embase through June 2019. Study-specifics risk estimates were combined using fixed effects models, or random-effects models when significant heterogeneity was detected. Dose-response analysis was modeled by using restricted cubic splines.
Results
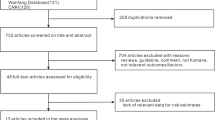
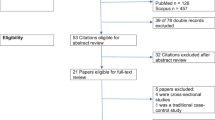
A total of 15 cohort studies, with 102,347 pregnancy women, was included in the meta-analysis. The pooled relative risk (RR) for LBW was 1.33 (95% CI: 1.12, 1.57) for mothers with the highest compared with the lowest level of caffeine intake during pregnancy, with significant heterogeneity across studies (I2 = 49.3%, P = 0.032). The pooled RR was 1.07 (95% CI: 1.02, 1.11) for each 100 mg/day increase of caffeine intake. The pooled RR for childhood overweight and obesity was 1.39 (95% CI: 1.15, 1.69) for mothers with the highest compared with the lowest level of caffeine intake during pregnancy. No significant heterogeneity across studies was detected (I2 = 38.9%, P = 0.179). The pooled RR was 1.31 (95% CI: 1.11, 1.55) for each 100 mg/day increase of caffeine intake. No evidence of publication bias was indicated.
Conclusions
Maternal caffeine intake during pregnancy is associated with higher risk of LBW and childhood overweight and obesity. Further studies may focus on investigating the potential mechanisms before the recommendation of complete avoidance of caffeine intake during pregnancy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Poole R, Kennedy OJ, Roderick P, Fallowfield JA, Hayes PC, Parkes J. Coffee consumption and health: umbrella review of meta-analyses of multiple health outcomes. BMJ. 2017;359:j5024.
Grosso G, Godos J, Galvano F, Giovannucci EL. Coffee, caffeine, and health outcomes: an umbrella review. Annu Rev Nutr. 2017;37:131–56.
Rhee J, Kim R, Kim Y, Tam M, Lai Y, Keum N, et al. Maternal caffeine consumption during pregnancy and risk of low birth weight: a dose-response meta-analysis of observational studies. PLoS ONE. 2015;10:e0132334.
American College of O, Gynecologists. ACOG CommitteeOpinion No. 462: moderate caffeine consumption during pregnancy. Obstet Gynecol. 2010;116(2 Pt 1):467–8.
Greenwood DC, Thatcher NJ, Ye J, Garrard L, Keogh G, King LG, et al. Caffeine intake during pregnancy and adverse birth outcomes: a systematic review and dose-response meta-analysis. Eur J Epidemiol. 2014;29:725–34.
Li DK, Ferber JR, Odouli R. Maternal caffeine intake during pregnancy and risk of obesity in offspring: a prospective cohort study. Int J Obes. 2015;39:658–64. https://doi.org/10.1038/ijo.2014.196. Epub 12 Nov 2014.
Papadopoulou E, Botton J, Brantsaeter AL, Haugen M, Alexander J, Meltzer HM, et al. Maternal caffeine intake during pregnancy and childhood growth and overweight: results from a large Norwegian prospective observational cohort study. BMJ Open. 2018;8:e018895. https://doi.org/10.1136/bmjopen-2017-018895.
Voerman E, Jaddoe VW, Gishti O, Hofman A, Franco OH, Gaillard R. Maternal caffeine intake during pregnancy, early growth, and body fat distribution at school age. Obesity. 2016;24:1170–7. https://doi.org/10.1002/oby.21466.
Chen LW, Murrin CM, Mehegan J, Kelleher CC, Phillips CM. Maternal, but not paternal or grandparental, caffeine intake is associated with childhood obesity and adiposity: The Lifeways Cross-Generation Cohort Study. Am J Clin Nutr. 2019;109:1648–55. https://doi.org/10.1093/ajcn/nqz019.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Orsini N, Li R, Wolk A, Khudyakov P, Spiegelman D. Meta-analysis for linear and nonlinear dose-response relations: examples, an evaluation of approximations, and software. Am J Epidemiol. 2012;175:66–73.
Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992;135:1301–9.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Mills JL, Holmes LB, Aarons JH, Simpson JL, Brown ZA, Jovanovic-Peterson LG, et al. Moderate caffeine use and the risk of spontaneous abortion and intrauterine growth retardation. Scand J Gastroenterol. 1993;28:285–8.
Grosso LM, Rosenberg KD, Belanger K, Saftlas AF, Leaderer B, Bracken MB. Maternal caffeine intake and intrauterine growth retardation. Epidemiology. 2001;12:447–55.
Group CS. Maternal caffeine intake during pregnancy and risk of fetal growth restriction: a large prospective observational study. BMJ. 2008;337:a2332.
Sengpiel V, Elind E, Bacelis J, Nilsson S, Grove J, Myhre R, et al. Maternal caffeine intake during pregnancy is associated with birth weight but not with gestational length: results from a large prospective observational cohort study. BMC Med. 2013;11:42.
Sasaki S, Limpar M, Sata F, Kobayashi S, Kishi R. Interaction between maternal caffeine intake during pregnancy and CYP1A2 C164A polymorphism affects infant birth size in the Hokkaido study. Pediatr Res. 2017;82:19–28.
van der Hoeven T, Browne JL, Uiterwaal C, van der Ent CK, Grobbee DE, Dalmeijer GW. Antenatal coffee and tea consumption and the effect on birth outcome and hypertensive pregnancy disorders. PLoS ONE. 2017;12:e0177619.
Peacock A, Hutchinson D, Wilson J, McCormack C, Bruno R, Olsson CA, et al. Adherence to the caffeine intake guideline during pregnancy and birth outcomes: a prospective cohort study. Nutrients. 2018;10:319.
Modzelewska D, Bellocco R, Elfvin A, Brantsæter AL, Meltzer HM, Jacobsson B, et al. Caffeine exposure during pregnancy, small for gestational age birth and neonatal outcome-results from the Norwegian mother and child cohort study. BMC Pregnancy Childbirth. 2019;19:80.
Linn S, Schoenbaum SC, Monson RR, Rosner B, Stubblefield PG, Ryan KJ. No association between coffee consumption and adverse outcomes of pregnancy. N Engl J Med. 1982;306:141–5.
McDonald AD, Armstrong BG, Sloan M. Cigarette, alcohol, and coffee consumption and prematurity. Am J Public Health. 1992;82:87–90.
Vitti FP, Grandi C, Cavalli RC, Simoes VMF, Batista RFL, Cardoso VC. Association between caffeine consumption in pregnancy and low birth weight and preterm birth in the birth cohort of Ribeirao Preto. Rev Bras Ginecol Obstet. 2018;40:749–56. https://doi.org/10.1055/s-0038-1675806. Epub 7 Dec 2018.
Chen LW, Fitzgerald R, Murrin CM, Mehegan J, Kelleher CC, Phillips CM. Associations of maternal caffeine intake with birth outcomes: results from the Lifeways Cross Generation Cohort Study. Am J Clin Nutr. 2018;108:1301–8. https://doi.org/10.1093/ajcn/nqy219.
Okubo H, Miyake Y, Tanaka K, Sasaki S, Hirota Y. Maternal total caffeine intake, mainly from Japanese and Chinese tea, during pregnancy was associated with risk of preterm birth: the Osaka Maternal and Child Health Study. Nutr Res. 2015;35:309–16. https://doi.org/10.1016/j.nutres.2015.02.009. Epub 2 Mar 2015.
Bakker R, Steegers EA, Obradov A, Raat H, Hofman A, Jaddoe VW. Maternal caffeine intake from coffee and tea, fetal growth, and the risks of adverse birth outcomes: the Generation R Study. Am J Clin Nutr. 2010;91:1691–8. https://doi.org/10.3945/ajcn.2009.28792. Epub 28 Apr 2010.
Bracken MB, Triche EW, Belanger K, Hellenbrand K, Leaderer BP. Association of maternal caffeine consumption with decrements in fetal growth. Am J Epidemiol. 2003;157:456–66. https://doi.org/10.1093/aje/kwf220.
Eskenazi B, Stapleton AL, Kharrazi M, Chee WY. Associations between maternal decaffeinated and caffeinated coffee consumption and fetal growth and gestational duration. Epidemiology. 1999;10:242–9.
Fortier I, Marcoux S, Beaulac-Baillargeon L. Relation of caffeine intake during pregnancy to intrauterine growth retardation and preterm birth. Am J Epidemiol. 1993;137:931–40. https://doi.org/10.1093/oxfordjournals.aje.a116763.
Olsen J, Overvad K, Frische G. Coffee consumption, birthweight, and reproductive failures. Epidemiology. 1991;2:370–4.
Martin TR, Bracken MB. The association between low birth weight and caffeine consumption during pregnancy. Am J Epidemiol. 1987;126:813–21. https://doi.org/10.1093/oxfordjournals.aje.a114718.
Wikoff D, Welsh BT, Henderson R, Brorby GP, Britt J, Myers E, et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem Toxicol. 2017;109(Pt 1):585–648.
Jahanfar S, Jaafar SH. Effects of restricted caffeine intake by mother on fetal, neonatal and pregnancy outcomes. Cochrane Database Syst Rev. 2015;6:CD006965.
Li DK, Ferber JR, Odouli R. Maternal caffeine intake during pregnancy and risk of obesity in offspring: a prospective cohort study. Int J Obes. 2015;39:658–64.
Cho WK, Suh BK. Catch-up growth and catch-up fat in children born small for gestational age. Korean J Pediatr. 2016;59:1–7.
Xu D, Zhang B, Liang G, Ping J, Kou H, Li X, et al. Caffeine-induced activated glucocorticoid metabolism in the hippocampus causes hypothalamic-pituitary-adrenal axis inhibition in fetal rats. PLoS ONE. 2012;7:e44497.
Xu D, Wu Y, Liu F, Liu YS, Shen L, Lei YY, et al. A hypothalamic-pituitary-adrenal axis-associated neuroendocrine metabolic programmed alteration in offspring rats of IUGR induced by prenatal caffeine ingestion. Toxicol Appl Pharmacol. 2012;264:395–403.
Signorello LB, Nordmark A, Granath F, Blot WJ, McLaughlin JK, Anneren G, et al. Caffeine metabolism and the risk of spontaneous abortion of normal karyotype fetuses. Obstet Gynecol. 2001;98:1059–66.
Bech BH, Autrup H, Nohr EA, Henriksen TB, Olsen J. Stillbirth and slow metabolizers of caffeine: comparison by genotypes. Int J Epidemiol. 2006;35:948–53.
Desai M, Ross MG. Fetal programming of adipose tissue: effects of intrauterine growth restriction and maternal obesity/high-fat diet. Semin Reprod Med. 2011;29:237–45.
Roh E, Song DK, Kim MS. Emerging role of the brain in the homeostatic regulation of energy and glucose metabolism. Exp Mol Med. 2016;48:e216.
Acknowledgements
We appreciate Dr. Yang Yang for his inputs on the statistical methods in this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jin, F., Qiao, C. Association of maternal caffeine intake during pregnancy with low birth weight, childhood overweight, and obesity: a meta-analysis of cohort studies. Int J Obes 45, 279–287 (2021). https://doi.org/10.1038/s41366-020-0617-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-020-0617-4
This article is cited by
-
The influence of six polymorphisms of uncoupling protein 3 (UCP3) gene and childhood obesity: a case–control study
BMC Pediatrics (2023)
-
Stoffwechsel und Ernährung in der Schwangerschaft
Monatsschrift Kinderheilkunde (2022)
-
Careful data extraction in meta-analysis: the case of maternal caffeine intake during pregnancy and low-birth weight, childhood overweight, and obesity
International Journal of Obesity (2021)