Abstract
Objecive
Women with twins have an a priori increased risk for many of the complications associated with maternal obesity. Thus, the impact of maternal obesity in twins may differ from that reported in singletons. In addition, given the increased metabolic demands in twin pregnancies, the impact of maternal underweight may be greater in twin compared with singleton gestations. Our objective was to test the hypothesis that the relationship between maternal pre-pregnancy body mass index (BMI) and adverse pregnancy outcomes differ between twin and singleton gestations.
Methods
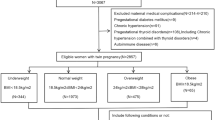
This was a retrospective population-based study of all women who had a singleton or twin hospital birth in Ontario, Canada, between April 2012 and March 2016. Data were obtained from the Better Outcomes Registry & Network (BORN) Ontario. The relationship between maternal BMI category and pregnancy complications was assessed separately in twin and singleton gestations. The primary outcome was a composite variable that included any of the following complications: preeclampsia, gestational diabetes, or preterm birth before 320/7 weeks. Relative risk (aRR) and 95% confidence intervals (CI) for adverse outcomes for each BMI category as defined by WHO (using normal weight category as reference) were generated using modified Poisson regression, adjusting for maternal age, nulliparity, smoking, previous preterm birth, and fetal sex.
Results
A total of 487,870 women with singleton (n = 480,010) and twin (n = 7860) pregnancies met the inclusion criteria. The risk of the composite primary outcome, preeclampsia, gestational diabetes, and cesarean delivery increased with high maternal BMI in both singleton and twin gestations, but these associations were weaker in twin compared with singleton gestations (association of BMI ≥ 40.0 kg/m2 with primary outcome: aRR = 3.10, 95%-CI 2.96–3.24 in singletons compared with aRR = 1.74, 95%-CI 1.37–2.20 in twins). In singleton pregnancies the risk of preterm birth at < 320/7 weeks increased with maternal BMI, mainly due to an increased risk of provider-initiated preterm birth. In twin gestations, however, underweight (but not overweight or obesity) was associated with the greatest risk of preterm birth at < 32 weeks (aRR 1.67, 95%-CI 1.17–2.37), mainly due to an increased risk of spontaneous preterm birth (aRR 2.10, 95%-CI 1.44–3.08).
Conclusion
In healthy women with twin pregnancies, underweight is associated with the greatest risk for preterm birth, while the association of maternal obesity with adverse pregnancy outcomes is weaker than that observed in singletons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mission JF, Marshall NE, Caughey AB. Pregnancy risks associated with obesity. Obstet Gynecol Clin North Am. 2015;42:335–53.
Lu GC, Rouse DJ, DuBard M, Cliver S, Kimberlin D, Hauth JC. The effect of the increasing prevalence of maternal obesity on perinatal morbidity. Am J Obstet Gynecol. 2001;185:845–9.
Kim SY, Dietz PM, England L, Morrow B, Callaghan WM. Trends in pre-pregnancy obesity in nine states, 1993–2003. Obesity. 2007;15:986–93.
Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser. 2000;894(i–xii):1–253.
Rasmussen KM, Yaktine AL, editors. Weight gain during pregnancy: reexamining the guidelines. The National Academies Collection: Reports funded by National Institutes of Health. Washington (DC); 2009.
ACOG Practice Bulletin No 156: obesity in pregnancy. Obstet Gynecol. 2015;126:e112–26.
Aamir AH. The obese pregnancy. J Pak Med Assoc. 2016;66(9Suppl 1):S65–8.
Doherty DA, Magann EF, Francis J, Morrison JC, Newnham JP. Pre-pregnancy body mass index and pregnancy outcomes. Int J Gynaecol Obstet. 2006;95:242–7.
Malik VS, Willett WC, Hu FB. Global obesity: trends, risk factors and policy implications. Nat Rev Endocrinol. 2013;9:13–27.
Orpana HM, Berthelot JM, Kaplan MS, Feeny DH, McFarland B, Ross NA. BMI and mortality: results from a national longitudinal study of Canadian adults. Obesity. 2010;18:214–8.
Viswanathan M, Siega-Riz AM, Moos MK, Deierlein A, Mumford S, Knaack J, et al. Outcomes of maternal weight gain. Evid Rep Technol Assess (Full Rep). 2008;168:1–223.
Callaway LK, Prins JB, Chang AM, McIntyre HD. The prevalence and impact of overweight and obesity in an Australian obstetric population. Med J Aust. 2006;184:56–9.
Davies GA, Maxwell C, McLeod L, Gagnon R, Basso M, Bos H, et al. Obesity in pregnancy. J Obstet Gynaecol Can. 2010;32:165–73.
Robinson HE, O’Connell CM, Joseph KS, McLeod NL. Maternal outcomes in pregnancies complicated by obesity. Obstet Gynecol. 2005;106:1357–64.
Salihu HM, Dunlop AL, Hedayatzadeh M, Alio AP, Kirby RS, Alexander GR. Extreme obesity and risk of stillbirth among black and white gravidas. Obstet Gynecol. 2007;110:552–7.
Sebire NJ, Jolly M, Harris JP, Wadsworth J, Joffe M, Beard RW, et al. Maternal obesity and pregnancy outcome: a study of 287,213 pregnancies in London. Int J Obes Relat Metab Disord. 2001;25:1175–82.
Yogev Y, Catalano PM. Pregnancy and obesity. Obstet Gynecol Clin North Am. 2009;36:285–300. viii
Al Wattar BH, Pidgeon C, Learner H, Zamora J, Thangaratinam S. Online health information on obesity in pregnancy: a systematic review. Eur J Obstet Gynecol Reprod Biol. 2016;206:147–52.
Vinturache AE, Chaput KH, Tough SC. Pre-pregnancy body mass index (BMI) and macrosomia in a Canadian birth cohort. J Matern Fetal Neonatal Med. 2017;30:109–16.
Catalano PM, Shankar K. Obesity and pregnancy: mechanisms of short term and long term adverse consequences for mother and child. BMJ. 2017;356:j1.
Avci ME, Sanlikan F, Celik M, Avci A, Kocaer M, Gocmen A. Effects of maternal obesity on antenatal, perinatal and neonatal outcomes. J Matern Fetal Neonatal Med. 2015;28:2080–3.
Dixit A, Girling JC. Obesity and pregnancy. J Obstet Gynaecol. 2008;28:14–23.
Chu SY, Callaghan WM, Kim SY, Schmid CH, Lau J, England LJ, et al. Maternal obesity and risk of gestational diabetes mellitus. Diabetes Care. 2007;30:2070–6.
Weiss JL, Malone FD, Emig D, Ball RH, Nyberg DA, Comstock CH, et al. Obesity, obstetric complications and cesarean delivery rate—a population-based screening study. Am J Obstet Gynecol. 2004;190:1091–7.
Gilead R, Yaniv Salem S, Sergienko R, Sheiner E. Maternal “isolated” obesity and obstetric complications. J Matern Fetal Neonatal Med. 2012;25:2579–82.
Kogan MD, Alexander GR, Kotelchuck M, MacDorman MF, Buekens P, Martin JA, et al. Trends in twin birth outcomes and prenatal care utilization in the United States, 1981–1997. JAMA. 2000;284:335–41.
Mathews TJ, MacDorman MF. Infant mortality statistics from the 2003 period linked birth/infant death data set. Natl Vital Stat Rep. 2006;54:1–29.
Chauhan SP, Scardo JA, Hayes E, Abuhamad AZ, Berghella V. Twins: prevalence, problems, and preterm births. Am J Obstet Gynecol. 2010;203:305–15.
Salihu HM, Bekan B, Aliyu MH, Rouse DJ, Kirby RS, Alexander GR. Perinatal mortality associated with abruptio placenta in singletons and multiples. Am J Obstet Gynecol. 2005;193:198–203.
Powers WF, Kiely JL. The risks confronting twins: a national perspective. Am J Obstet Gynecol. 1994;170:456–61.
American College of O, Gynecologists Committee on Practice B-O, Society for Maternal-Fetal M, Committee AJE. ACOG Practice Bulletin #56: Multiple gestation: complicated twin, triplet, and high-order multifetal pregnancy. Obstet Gynecol. 2004;104:869–83.
Blickstein I. Growth aberration in multiple pregnancy. Obstet Gynecol Clin North Am. 2005;32:39–54. viii
Blondel B, Kogan MD, Alexander GR, Dattani N, Kramer MS, Macfarlane A, et al. The impact of the increasing number of multiple births on the rates of preterm birth and low birthweight: an international study. Am J Public Health. 2002;92:1323–30.
Helmerhorst FM, Perquin DA, Donker D, Keirse MJ. Perinatal outcome of singletons and twins after assisted conception: a systematic review of controlled studies. BMJ. 2004;328:261.
Kilpatrick SJ, Jackson R, Croughan-Minihane MS. Perinatal mortality in twins and singletons matched for gestational age at delivery at > or = 30 weeks. Am J Obstet Gynecol. 1996;174(1 Pt 1):66–71.
Luke B, Keith LG. The contribution of singletons, twins and triplets to low birth weight, infant mortality and handicap in the United States. J Reprod Med. 1992;37:661–6.
Lynch A, McDuffie R Jr., Murphy J, Faber K, Orleans M. Preeclampsia in multiple gestation: the role of assisted reproductive technologies. Obstet Gynecol. 2002;99:445–51.
Schwartz DB, Daoud Y, Zazula P, Goyert G, Bronsteen R, Wright D, et al. Gestational diabetes mellitus: metabolic and blood glucose parameters in singleton versus twin pregnancies. Am J Obstet Gynecol. 1999;181:912–4.
Seoud MA, Toner JP, Kruithoff C, Muasher SJ. Outcome of twin, triplet, and quadruplet in vitro fertilization pregnancies: the Norfolk experience. Fertil Steril. 1992;57:825–34.
Sibai BM, Hauth J, Caritis S, Lindheimer MD, MacPherson C, Klebanoff M, et al. Hypertensive disorders in twin versus singleton gestations. National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. Am J Obstet Gynecol. 2000;182:938–42.
Spellacy WN, Handler A, Ferre CD. A case-control study of 1253 twin pregnancies from a 1982–1987 perinatal data base. Obstet Gynecol. 1990;75:168–71.
Weissman A, Drugan A. Glucose tolerance in singleton, twin and triplet pregnancies. J Perinat Med. 2016;44:893–7.
Shinagawa S, Suzuki S, Chihara H, Otsubo Y, Takeshita T, Araki T. Maternal basal metabolic rate in twin pregnancy. Gynecol Obstet Invest. 2005;60:145–8.
Luke B. Nutrition in multiple gestations. Clin Perinatol. 2005;32:403–29. vii
Luke B. Improving multiple pregnancy outcomes with nutritional interventions. Clin Obstet Gynecol. 2004;47:146–62.
Fox NS, Roman AS, Saltzman DH, Klauser CK, Rebarber A. Obesity and adverse pregnancy outcomes in twin pregnancies. J Matern Fetal Neonatal Med. 2014;27:355–9.
Lucovnik M, Blickstein I, Verdenik I, Steblovnik L, Trojner Bregar A, Tul N. Impact of pre-gravid body mass index and body mass index change on preeclampsia and gestational diabetes in singleton and twin pregnancies. J Matern Fetal Neonatal Med. 2014;27:1901–4.
Lucovnik M, Blickstein I, Verdenik I, Trojner-Bregar A, Tul N. Maternal obesity in singleton versus twin gestations: a population-based matched case-control study. J Matern Fetal Neonatal Med. 2015;28:623–5.
Sung SJ, Lee SM, Kim S, Kim BJ, Park CW, Park JS, et al. The risk of spontaneous preterm birth according to maternal pre-pregnancy body mass index in twin gestations. J Korean Med Sci. 2018;33:e103.
Vinturache A, McKeating A, Daly N, Sheehan S, Turner M. Maternal body mass index and the prevalence of spontaneous and elective preterm deliveries in an Irish obstetric population: a retrospective cohort study. BMJ Open. 2017;7:e015258.
Aliyu MH, Alio AP, Lynch O, Mbah A, Salihu HM. Maternal pre-gravid body weight and risk for placental abruption among twin pregnancies. J Matern Fetal Neonatal Med. 2009;22:745–50.
Al-Obaidly S, Parrish J, Murphy KE, Maxwell C. Maternal pre-gravid body mass index and obstetric outcomes in twin gestations. J Perinatol. 2014;34:425–8.
Salihu HM, Alio AP, Belogolovkin V, Aliyu MH, Wilson RE, Reddy UM, et al. Prepregnancy obesity and risk of stillbirth in viable twin gestations. Obesity. 2010;18:1795–800.
Colletto GM, Segre CA. Lack of effect of maternal body mass index on anthropometric characteristics of newborns in twin gestations. Genet Mol Res. 2005;4:47–54.
Simoes T, Queiros A, Correia L, Rocha T, Dias E, Blickstein I. Gestational diabetes mellitus complicating twin pregnancies. J Perinat Med. 2011;39:437–40.
Suzuki S, Yoneyama Y, Sawa R, Shin S, Araki T. Clinical usefulness of maternal body mass index in twin pregnancies. Hypertens Pregnancy. 2000;19:273–9.
Suzuki S, Inde Y, Miyake H. Maternal obesity as a risk factor for very pre-term delivery in dichorionic twin pregnancies. J Obstet Gynaecol. 2010;30:354–6.
Kosinska-Kaczynska K, Szymusik I, Kaczynski B, Wielgos M. Observational study of associations between gestational weight gain and perinatal outcomes in dichorionic twin pregnancies. Int J Gynaecol Obstet. 2017;138:94–9.
White IR, Royston P, Wood AM. Multiple imputation using chained equations: Issues and guidance for practice. Stat Med. 2011;30:377–99.
Kramer MS, Platt RW, Wen SW, Joseph KS, Allen A, Abrahamowicz M, et al. A new and improved population-based Canadian reference for birth weight for gestational age. Pediatrics. 2001;108:E35.
Altman DG, Bland JM. Interaction revisited: the difference between two estimates. BMJ. 2003;326:219.
Lynch AM, Hart JE, Agwu OC, Fisher BM, West NA, Gibbs RS. Association of extremes of prepregnancy BMI with the clinical presentations of preterm birth. Am J Obstet Gynecol. 2014;210:428 e1–9.
Han Z, Mulla S, Beyene J, Liao G, McDonald SD, Knowledge Synthesis G. Maternal underweight and the risk of preterm birth and low birth weight: a systematic review and meta-analyses. Int J Epidemiol. 2011;40:65–101.
Shaw GM, Wise PH, Mayo J, Carmichael SL, Ley C, Lyell DJ, et al. Maternal prepregnancy body mass index and risk of spontaneous preterm birth. Paediatr Perinat Epidemiol. 2014;28:302–11.
Hoellen F, Hornemann A, Haertel C, Reh A, Rody A, Schneider S, et al. Does maternal underweight prior to conception influence pregnancy risks and outcome? In Vivo. 2014;28:1165–70.
Sharifzadeh F, Kashanian M, Jouhari S, Sheikhansari N. Relationship between pre-pregnancy maternal BMI with spontaneous preterm delivery and birth weight. J Obstet Gynaecol. 2015;35:354–7.
Luke B. The evidence linking maternal nutrition and prematurity. J Perinat Med. 2005;33:500–5.
Salihu HM, Mbah AK, Alio AP, Clayton HB, Lynch O. Low pre-pregnancy body mass index and risk of medically indicated versus spontaneous preterm singleton birth. Eur J Obstet Gynecol Reprod Biol. 2009;144:119–23.
Siega-Riz AM, Adair LS, Hobel CJ. Maternal underweight status and inadequate rate of weight gain during the third trimester of pregnancy increases the risk of preterm delivery. J Nutr. 1996;126:146–53.
Lechtig A, Yarbrough C, Delgado H, Martorell R, Klein RE, Behar M. Effect of moderate maternal malnutrition on the placenta. Am J Obstet Gynecol. 1975;123:191–201.
Hediger ML, Luke B, Gonzalez-Quintero VH, Martin D, Nugent C, Witter FR, et al. Fetal growth rates and the very preterm delivery of twins. Am J Obstet Gynecol. 2005;193:1498–507.
Bukowski R, Gahn D, Denning J, Saade G. Impairment of growth in fetuses destined to deliver preterm. Am J Obstet Gynecol. 2001;185:463–7.
Lysikiewicz A, Bracero LA, Tejani N. Sonographically estimated fetal weight percentile as a predictor of preterm delivery. J Matern Fetal Med. 2001;10:44–7.
Zhang Q, Ananth CV, Li Z, Smulian JC. Maternal anaemia and preterm birth: a prospective cohort study. Int J Epidemiol. 2009;38:1380–9.
Allen LH. Biological mechanisms that might underlie iron’s effects on fetal growth and preterm birth. J Nutr. 2001;131(2S-2):581S–9S.
Funding
This study was funded by the Canadian Institute of Health Research (CIHR) (Grant#146442; Non-communicable Diseases in Obstetrics: Improving Quality of Care and Maternal-infant Outcomes Through an Obstetrical Research Network). Matched funding was provided by the Department of Obstetrics and Gynecology at the University of Toronto, McMaster University, Sunnybrook Research Institute, and Providence St. Joseph’s and St. Michael’s Healthcare. Dr. Sarah D. McDonald is supported by a Tier II Canada Research Chair. Dr. Beth Murray-Davis is supported by a Hamilton Health Sciences Early Career Award. None of the funding agencies had any role in the idea, design, analyses, interpretation of data, writing of the manuscript or decision to submit the manuscript.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41366_2019_362_MOESM3_ESM.docx
Table S3: Association between maternal BMI and maternal and neonatal adverse outcomes in singleton pregnancies – multivariable analysis
41366_2019_362_MOESM4_ESM.docx
Table S4: Association between maternal BMI and maternal and neonatal adverse outcomes in twin pregnancies – multivariable analysis
Rights and permissions
About this article
Cite this article
Ram, M., Berger, H., Lipworth, H. et al. The relationship between maternal body mass index and pregnancy outcomes in twin compared with singleton pregnancies. Int J Obes 44, 33–44 (2020). https://doi.org/10.1038/s41366-019-0362-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-019-0362-8
This article is cited by
-
Pre-pregnancy body mass index and adverse maternal and perinatal outcomes in twins: A population retrospective cohort study
International Journal of Obesity (2023)
-
Association of parental prepregnancy BMI with neonatal outcomes and birth defect in fresh embryo transfer cycles: a retrospective cohort study
BMC Pregnancy and Childbirth (2021)
-
Association of maternal pre-pregnancy low or increased body mass index with adverse pregnancy outcomes
Scientific Reports (2021)
-
Association between pregravid liver enzyme levels and gestational diabetes in twin pregnancies: a secondary analysis of national cohort study
Scientific Reports (2021)
-
Challenges for better care based on the course of maternal body mass index, weight gain and multiple outcome in twin pregnancies: a population-based retrospective cohort study in Hessen/Germany within 15 years
Archives of Gynecology and Obstetrics (2020)