Abstract
Background/objective
Adaptation to the extrauterine environment depends on a switch from glycolysis to catabolism of fatty acids (FA) provided as milk lipids. We sought to learn whether the postnatal induction of muscle FA oxidation in mice could reflect propensity to obesity and to characterize the mechanisms controlling this induction.
Methods
Experiments were conducted using obesity-resistant A/J and obesity-prone C57BL/6J (B6) mice maintained at 30 °C, from 5 to 28 days after birth. At day 10, both A/J and B6 mice with genetic ablation (KO) of α2 subunit of AMP-activated protein kinase (AMPK) were also used. In skeletal muscle, expression of selected genes was determined using quantitative real-time PCR, and AMPK subunits content was evaluated using Western blotting. Activities of both AMPK and pyruvate dehydrogenase (PDH), as well as acylcarnitine levels in the muscle were measured.
Results
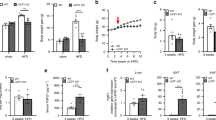
Acylcarnitine levels and gene expression indicated transient increase in FA oxidation during the first 2 weeks after birth, with a stronger increase in A/J mice. These data correlated with (i) the surge in plasma leptin levels, which peaked at day 10 and was higher in A/J mice, and (ii) relatively low activity of PDH linked with up-regulation of PDH kinase 4 gene (Pdk4) expression in the 10-day-old A/J mice. In contrast with the Pdk4 expression, transient up-regulation of uncoupling protein 3 gene was observed in B6 but not A/J mice. AMPK activity changed during the development, without major differences between A/J and B6 mice. Expression of neither Pdk4 nor other muscle genes was affected by AMPK-KO.
Conclusions
Our results indicate a relatively strong postnatal induction of FA oxidation in skeletal muscle of the obesity-resistant A/J mice. This induction is transient and probably results from suppression of PDH activity, linked with a postnatal surge in plasma leptin levels, independent of AMPK.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ferre P, Decaux JF, Issad T, Girard J. Changes in energy metabolism during the suckling and weaning period in the newborn. Reprod Nutr Dev. 1986;26(2B):619–31.
Koldovsky O, Dobiasova M, Drahota Z, Hahn P. Developmental aspects of lipid metabolism. Physiol Res. 1995;44:353–6.
Hahn P. Effect of litter size on plasma cholesterol and insulin and some liver and adipose tissue enzymes in adult rodents. J Nutr. 1984;114:1231–4.
Stocker CJ, Cawthorne MA. The influence of leptin on early life programming of obesity. Trends Biotechnol. 2008;26:545–51.
Pico C, Jilkova ZM, Kus V, Palou A, Kopecky J. Perinatal programming of body weight control by leptin: putative roles of AMP kinase and muscle thermogenesis. Am J Clin Nutr. 2011;94:1830S–7S.
Palou M, Pico C, Palou A. Leptin as a breast milk component for the prevention of obesity. Nutr Rev. 2018;76:875–892.
Koldovsky O, Hahn P, Hromadova M, Krecek J, Macho L. Late effects of early nutritional manipulations. Physiol Res. 1995;44:357–60.
Pico C, Oliver P, Sanchez J, Miralles O, Caimari A, Priego T, et al. The intake of physiological doses of leptin during lactation in rats prevents obesity in later life. Int J Obes. 2007;31:1199–209.
Sanchez J, Priego T, Palou M, Tobaruela A, Palou A, Pico C. Oral supplementation with physiological doses of leptin during lactation in rats improves insulin sensitivity and affects food preferences later in life. Endocrinology. 2008;149:733–40.
Schuster S, Hechler C, Gebauer C, Kiess W, Kratzsch J. Leptin in maternal serum and breast milk: association with infants’ body weight gain in a longitudinal study over 6 months of lactation. Pediatr Res. 2011;70:633–7.
Devaskar SU, Ollesch C, Rajakumar RA, Rajakumar PA. Developmental changes in ob gene expression and circulating leptin peptide concentrations. Biochem Biophys Res Commun. 1997;238:44–7.
Ahima RS, Prabakaran D, Flier JS. Postnatal leptin surge and regulation of circadian rhythm of leptin by feeding. Implications for energy homeostasis and neuroendocrine function. J Clin Invest. 1998;101:1020–7.
Mistry AM, Swick A, Romsos DR. Leptin alters metabolic rates before acquisition of its anorectic effect in developing neonatal mice. Am J Physiol. 1999;277:R742–R7.
Flier JS, Maratos-Flier E. Obesity and the hypothalamus: novel peptides for new pathways. Cell. 1998;92:437–40.
Stehling O, Doring H, Nuesslein-Hildesheim B, Olbort M, Schmidt I. Leptin does not reduce body fat content but augments cold defense abilities in thermoneutrally reared rat pups. Pflugers Arch. 1997;434:694–7.
Blumberg MS, Deaver K, Kirby RF. Leptin disinhibits nonshivering thermogenesis in infants after maternal separation. Am J Physiol. 1999;276:R606–R10.
Ukropec J, Anunciado RV, Ravussin Y, Kozak LP. Leptin is required for uncoupling protein-1-independent thermogenesis during cold stress. Endocrinology. 2006;147:2468–80.
Muoio DM, Dohm GL, Fiedorek FT Jr, Tapscott EB, Coleman RA. Leptin directly alters lipid partitioning in skeletal muscle. Diabetes . 1997;46:1360–3.
Hardie DG, Hawley SA, Scott JW. AMP-activated protein kinase--development of the energy sensor concept. J Physiol . 2006;574(Pt 1):7–15.
Viollet B, Andreelli F, Jorgensen SB, Perrin C, Flamez D, Mu J, et al. Physiological role of AMP-activated protein kinase (AMPK): insights from knockout mouse models. Biochem Soc Trans. 2003;31(Pt 1):216–9.
Solinas G, Summermatter S, Mainieri D, Gubler M, Pirola L, Wymann MP, et al. The direct effect of leptin on skeletal muscle thermogenesis is mediated by substrate cycling between de novo lipogenesis and lipid oxidation. FEBS Lett. 2004;577:539–44.
Minokoshi Y, Kim YB, Peroni OD, Fryer LG, Muller C, Carling D, et al. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature. 2002;415:339–43.
Suzuki A, Okamoto S, Lee S, Saito K, Shiuchi T, Minokoshi Y. Leptin stimulates fatty acid oxidation and peroxisome proliferator-activated receptor alpha gene expression in mouse C2C12 myoblasts by changing the subcellular localization of the alpha2 form of AMP-activated protein kinase. Mol Cell Biol. 2007;27:4317–27.
Brun S, Carmona MC, Mampel T, Vinas O, Giralt M, Iglesias R, et al. Activators of peroxisome proliferator-activated receptor-a induce the expression of the uncoupling protein-3 gene in skeletal muscle. Diabetes. 1999;48:1217–22.
Garratt ES, Vickers MH, Gluckman PD, Hanson MA, Burdge GC, Lillycrop KA. Tissue-specific 5’ heterogeneity of PPARalpha transcripts and their differential regulation by leptin. PLoS ONE. 2013;8:e67483.
Pawlak M, Lefebvre P, Staels B. Molecular mechanism of PPARalpha action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J Hepatol. 2015;62:720–33.
Fritzen AM, Lundsgaard AM, Jeppesen J, Christiansen ML, Bienso R, Dyck JR, et al. 5’-AMP activated protein kinase alpha2 controls substrate metabolism during post-exercise recovery via regulation of pyruvate dehydrogenase kinase 4. J Physiol. 2015;593:4765–80.
Flachs P, Adamcova K, Zouhar P, Marques C, Janovska P, Viegas I, et al. Induction of lipogenesis in white fat during cold exposure in mice: link to lean phenotype. Int J Obes. 2017;41:372–80.
Horakova O, Hansikova J, Bardova K, Gardlo A, Rombaldova M, Kuda O, et al. Plasma acylcarnitines and amino acid levels as an early complex biomarker of propensity to high-fat diet-induced obesity in mice. PLoS ONE. 2016;11:e0155776.
Kus V, Prazak T, Brauner P, Hensler M, Kuda O, Flachs P, et al. Induction of muscle thermogenesis by high-fat diet in mice: association with obesity-resistance. Am J Physiol Endocrinol Metab. 2008;295:E356–67.
Bardova K, Horakova O, Janovska P, Hansikova J, Kus V, van Schothorst EM, et al. Early differences in metabolic flexibility between obesity-resistant and obesity-prone mice. Biochimie. 2016;124:163–70.
Collins S, Daniel KW, Petro AE, Surwit RS. Strain-specific response to beta3-adrenergic receptor agonist treatment of diet-induced obesity in mice. Endocrinology. 1997;138:405–13.
Guerra C, Koza RA, Yamashita H, King KW, Kozak LP. Emergence of brown adipocytes in white fat in mice is under genetic control. Effects on body weight and adiposity. J Clin Invest. 1998;102:412–20.
Viollet B, Andreelli F, Jorgensen SB, Perrin C, Geloen A, Flamez D, et al. The AMP-activated protein kinase alpha2 catalytic subunit controls whole-body insulin sensitivity. J Clin Invest. 2003;111:91–8.
Cahova M, Chrastina P, Hansikova H, Drahota Z, Trnovska J, Skop V, et al. Carnitine supplementation alleviates lipid metabolism derangements and protects against oxidative stress in non-obese hereditary hypertriglyceridemic rats. Appl Physiol Nutr Metab. 2015;40:280–91.
Pomar CA, Kuda O, Kopecky J, Rombaldova M, Castro H, Pico C, et al. Alterations in plasma acylcarnitine and amino acid profiles may be indicative of poor nutrition during the suckling period due to maternal intake of an unbalanced diet and predict later metabolic dysfunction. FASEB J. 2018:fj201800327RR.
Jelenik T, Rossmeisl M, Kuda O, Jilkova ZM, Medrikova D, Kus V, et al. AMP-activated protein kinase {alpha}2 subunit is required for the preservation of hepatic insulin sensitivity by n-3 polyunsaturated fatty acids. Diabetes. 2010;59:2737–46.
Spacilova J, Hulkova M, Hrustincova A, Capek V, Tesarova M, Hansikova H, et al. Analysis of expression profiles of genes involved in F(o)F(1)-ATP synthase biogenesis during perinatal development in rat liver and skeletal muscle. Physiol Res. 2016;65:597–608.
McGarry JD, Mills SE, Long CS, Foster DW. Observations on the affinity for carnitine, and malonyl-CoA sensitivity, of carnitine palmitoyltransferase I in animal and human tissues. Demonstration of the presence of malonyl-CoA in non-hepatic tissues of the rat1. Biochem J. 1983;214:21–8.
Cha SH, Rodgers JT, Puigserver P, Chohnan S, Lane MD. Hypothalamic malonyl-CoA triggers mitochondrial biogenesis and oxidative gene expression in skeletal muscle: Role of PGC-1alpha. Proc Natl Acad Sci USA. 2006;103:15410–5.
Shabalina IG, Hoeks J, Kramarova TV, Schrauwen P, Cannon B, Nedergaard J. Cold tolerance of UCP1-ablated mice: a skeletal muscle mitochondria switch toward lipid oxidation with marked UCP3 up-regulation not associated with increased basal, fatty acid- or ROS-induced uncoupling or enhanced GDP effects. Biochem. Biophys. Acta. 2010;1797:968–80.
MacLellan JD, Gerrits MF, Gowing A, Smith PJ, Wheeler MB, Harper ME. Physiological increases in uncoupling protein 3 augment fatty acid oxidation and decrease reactive oxygen species production without uncoupling respiration in muscle cells. Diabetes. 2005;54:2343–50.
Kwon HS, Harris RA. Mechanisms responsible for regulation of pyruvate dehydrogenase kinase 4 gene expression. Adv Enzyme Regul. 2004;44:109–21.
Schooneman MG, Achterkamp N, Argmann CA, Soeters MR, Houten SM. Plasma acylcarnitines inadequately reflect tissue acylcarnitine metabolism. Biochim Biophys Acta. 2014;1841:987–94.
Mihalik SJ, Goodpaster BH, Kelley DE, Chace DH, Vockley J, Toledo FG, et al. Increased levels of plasma acylcarnitines in obesity and type 2 diabetes and identification of a marker of glucolipotoxicity. Obesity (Silver Spring). 2010;18:1695–700.
Koves TR, Ussher JR, Noland RC, Slentz D, Mosedale M, Ilkayeva O, et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 2008;7:45–56.
Muoio DM, Noland RC, Kovalik JP, Seiler SE, Davies MN, DeBalsi KL, et al. Muscle-specific deletion of carnitine acetyltransferase compromises glucose tolerance and metabolic flexibility. Cell Metab. 2012;15:764–77.
Mounier R, Lantier L, Leclerc J, Sotiropoulos A, Pende M, Daegelen D, et al. Important role for AMPKalpha1 in limiting skeletal muscle cell hypertrophy. FASEB J. 2009;23:2264–73.
Fu X, Zhu M, Zhang S, Foretz M, Viollet B, Du M. Obesity Impairs skeletal muscle regeneration through inhibition of AMPK. Diabetes. 2016;65:188–200.
Porter C, Constantin-Teodosiu D, Constantin D, Leighton B, Poucher SM, Greenhaff PL. Muscle carnitine availability plays a central role in regulating fuel metabolism in the rodent. J Physiol. 2017;595:5765–80.
Soeters MR, Serlie MJ, Sauerwein HP, Duran M, Ruiter JP, Kulik W, et al. Characterization of D-3-hydroxybutyrylcarnitine (ketocarnitine): an identified ketosis-induced metabolite. Metabolism. 2012;61:966–73.
Gong DW, He Y, Karas M, Reitman M. Uncoupling protein-3 is a mediator of thermogenesis regulated by thyroid hormone, beta3-adrenergic agonists, and leptin. J Biol Chem. 1997;272:24129–32.
Brun S, Carmona MC, Mampel T, Vinas O, Giralt M, Iglesias R, et al. Uncoupling protein-3 gene expression in skeletal muscle during development is regulated by nutritional factors that alter circulating non-esterified fatty acids. FEBS Lett. 1999;453:205–9.
Brauner P, Kopecky P, Flachs P, Kuda O, Vorlicek J, Planickova L, et al. Expression of uncoupling protein 3 and GLUT4 gene in skeletal muscle of preterm newborns: possible control by AMP-activated protein kinase. Pediatr Res. 2006;60:569–75.
Rinnankoski-Tuikka R, Silvennoinen M, Torvinen S, Hulmi JJ, Lehti M, Kivela R, et al. Effects of high-fat diet and physical activity on pyruvate dehydrogenase kinase-4 in mouse skeletal muscle. Nutr Metab. 2012;9:53.
Zhang S, Hulver MW, McMillan RP, Cline MA, Gilbert ER. The pivotal role of pyruvate dehydrogenase kinases in metabolic flexibility. Nutr Metab. 2014;11:10.
Hue L, Taegtmeyer H. The Randle cycle revisited: a new head for an old hat. Am J Physiol Endocrinol Metab. 2009;297:E578–E91.
Broderick TL, Quinney HA, Lopaschuk GD. Carnitine stimulation of glucose oxidation in the fatty acid perfused isolated working rat heart. J Biol Chem. 1992;267:3758–63.
Wan Z, Thrush AB, Legare M, Frier BC, Sutherland LN, Williams DB, et al. Epinephrine-mediated regulation of PDK4 mRNA in rat adipose tissue. Am J Physiol Cell Physiol. 2010;299:C1162–70.
Puthanveetil P, Wang Y, Wang F, Kim MS, Abrahani A, Rodrigues B. The increase in cardiac pyruvate dehydrogenase kinase-4 after short-term dexamethasone is controlled by an Akt-p38-forkhead box other factor-1 signaling axis. Endocrinology. 2010;151:2306–18.
Acknowledgements
The research was supported by the Czech Science Foundation (MITOCENTRE, GB14-36804G). We thank J. Bemova, S. Hornova, and D. Salkova for technical assistance, D. Grahame Hardie (University of Dundee, UK) for the AMPKα1 and AMPKα2 antibodies, and B. Viollet (Institute Cochin, Paris, France) for the AMPK-KO-B6 mice [34].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
Buresova, J., Janovska, P., Kuda, O. et al. Postnatal induction of muscle fatty acid oxidation in mice differing in propensity to obesity: a role of pyruvate dehydrogenase. Int J Obes 44, 235–244 (2020). https://doi.org/10.1038/s41366-018-0281-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-018-0281-0