Abstract
Background
Potentially driven by the lack of mother-to-infant transmission of microbiota at birth, cesarean delivery has been associated with higher risk of offspring obesity. Yet, no studies have examined when delivery-mode differences in adiposity begin to emerge. In this study, we examine differences in infant weight and adiposity trajectories from birth to 12 months by delivery mode.
Methods
From 2013 to 2015, we recruited pregnant women into the Nurture Study and followed up their 666 infants. We ascertained maternal delivery method and infant birth weight from medical records. We measured weight, length, and skinfold thicknesses (subscapular, triceps, abdominal) when infants were 3, 6, 9, and 12 months of age. The main outcome, infant weight-for-length z score, was derived based on the WHO Child Growth Standards. We used linear regression models to assess the difference at each time point and used linear mixed models to examine the growth rate for infant weight and adiposity trajectories. We controlled for maternal age, race, marital status, education level, household income, smoking status, maternal pre-pregnancy body mass index, and infant birth weight.
Results
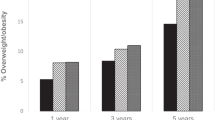
Of the 563 infants in our final sample, 179 (31.8%) were cesarean delivered. From birth to 12 months, the rate of increase in weight-for-length z score was 0.02/month (p = 0.03) greater for cesarean-delivered than vaginally-delivered infants. As a result of more rapid growth, cesarean-delivered infants had higher weight-for-length z score (0.26, 95% CI: 0.05, 0.47) and sum of subscapular and triceps (SS + TR) skinfold thickness (0.95 mm, 95% CI: 0.30, 1.60)—an indicator for overall adiposity—at 12 months, compared to vaginally-delivered infants.
Conclusions
Compared to vaginal delivery, cesarean delivery was associated with greater offspring rate of weight gain over the first year and differences in adiposity that appear as early as 3 months of age. Monitoring cesarean-delivered infants closely for excess weight gain may help guide primordial prevention of obesity later in life.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Skinner AC, Ravanbakht SN, Skelton JA, Perrin EM, Armstrong SC. Prevalence of Obesity and Severe Obesity in US Children, 1999–2016. Pediatrics. https://doi.org/10.1542/peds.2017-3459 (2018).
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444:1027–31. https://doi.org/10.1038/nature05414.
Ley RE, et al. Obesity alters gut microbial ecology. Proc Natl Acad Sci USA. 2005;102:11070–5. https://doi.org/10.1073/pnas.0504978102.
Boulange CL, Neves AL, Chilloux J, Nicholson JK, Dumas ME. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 2016;8:42 https://doi.org/10.1186/s13073-016-0303-2.
DeFrances CJ, Cullen KA, Kozak LJ. National Hospital Discharge Survey: 2005 annual summary with detailed diagnosis and procedure data. Vital and Health Statistics. Series 13, Data from the National Health Survey, 165:1–209 (2007).
Dominguez-Bello MG, et al. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci USA. 2010;107:11971–5. 1002601107 [pii]10.1073/pnas.1002601107.
Yassour M, et al. Natural history of the infant gut microbiome and impact of antibiotic treatment on bacterial strain diversity and stability. Sci Transl Med. 2016;8:343ra381 https://doi.org/10.1126/scitranslmed.aad0917.
Bokulich NA, et al. Antibiotics, birth mode, and diet shape microbiome maturation during early life. Sci Transl Med. 2016;8:343ra382 https://doi.org/10.1126/scitranslmed.aad7121.
Mueller NT. et al. Delivery Mode and the Transition of Pioneering Gut-Microbiota Structure, Composition and Predicted Metabolic Function. Genes 8, https://doi.org/10.3390/genes8120364 (2017).
Ye J, Betran AP, Guerrero Vela M, Souza JP, Zhang J. Searching for the optimal rate of medically necessary cesarean delivery. Birth. 2014;41:237–44. https://doi.org/10.1111/birt.12104.
(UK)., N. C. C. f. W. s. a. C. s. H. Caesarean Section, NICE Clinical Guidelines, No. 132, 2011).
Li HT, Zhou YB, Liu JM. The impact of cesarean section on offspring overweight and obesity: a systematic review and meta-analysis. Int J Obes. 2013;37:893–9. https://doi.org/10.1038/ijo.2012.195.
Kuhle S, Tong OS, Woolcott CG. Association between caesarean section and childhood obesity: a systematic review and meta-analysis. Obes Rev. 2015;16:295–303. https://doi.org/10.1111/obr.12267.
Darmasseelane K, Hyde MJ, Santhakumaran S, Gale C, Modi N. Mode of delivery and offspring body mass index, overweight and obesity in adult life: a systematic review and meta-analysis. PLoS ONE. 2014;9:e87896 https://doi.org/10.1371/journal.pone.0087896.
Huh SY, et al. Delivery by caesarean section and risk of obesity in preschool age children: a prospective cohort study. Arch Dis Child. 2012;97:610–6. https://doi.org/10.1136/archdischild-2011-301141.
Mueller NT, et al. Prenatal exposure to antibiotics, cesarean section and risk of childhood obesity. Int J Obes. 2015;39:665–70. https://doi.org/10.1038/ijo.2014.180.
Blustein J, et al. Association of caesarean delivery with child adiposity from age 6 weeks to 15 years. Int J Obes. 2013;37:900–6. https://doi.org/10.1038/ijo.2013.49.
Benjamin Neelon SE, et al. Cohort profile for the Nurture Observational Study examining associations of multiple caregivers on infant growth in the Southeastern USA. BMJ Open. 2017;7:e013939 https://doi.org/10.1136/bmjopen-2016-013939.
Shorr IJ. How to Weight and Measure Children: Assessing the Nutritional Status of Young Children in Household Surveys. (United Nations Department of Technical Co-operation for Development and Statistical Office, 1986).
Child Growth WHO. Standards based on length/height, weight and age. Acta Paediatr. 2006;450:76–85.
Villar J, et al. International standards for newborn weight, length, and head circumference by gestational age and sex: the Newborn Cross-Sectional Study of the INTERGROWTH-21st Project. Lancet. 2014;384:857–68. https://doi.org/10.1016/s0140-6736(14)60932-6.
Martin JA, Hamilton BE, Osterman MJK. Births in the United States, 2016. NCHS Data Brief, 1–8 (2017).
Martinez KA 2nd, et al. Increased weight gain by C-section: functional significance of the primordial microbiome. Sci Adv. 2017;3:eaao1874 https://doi.org/10.1126/sciadv.aao1874.
Keyes KM, Galea S. Population Health Science. (Oxford University Press, 2016).
Taveras EM, et al. Weight status in the first 6 months of life and obesity at 3 years of age. Pediatrics. 2009;123:1177–83. https://doi.org/10.1542/peds.2008-1149.
Cox LM, et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell. 2014;158:705–21. https://doi.org/10.1016/j.cell.2014.05.052.
Steinborn A, et al. Spontaneous labour at term is associated with fetal monocyte activation. Clin Exp Immunol. 1999;117:147–52. https://doi.org/10.1046/j.1365-2249.1999.00938.x.
Lim FT, et al. Association of stress during delivery with increased numbers of nucleated cells and hematopoietic progenitor cells in umbilical cord blood. Am J Obstet Gynecol. 2000;183:1144–52. https://doi.org/10.1067/mob.2000.108848.
Redzko S, Przepiesc J, Zak J, Urban J, Wysocka J. Influence of perinatal factors on hematological variables in umbilical cord blood. J Perinat Med. 2005;33:42–45. https://doi.org/10.1515/jpm.2005.007.
Glasser L, Sutton N, Schmeling M, Machan JT. A comprehensive study of umbilical cord blood cell developmental changes and reference ranges by gestation, gender and mode of delivery. J Perinatol. 2015;35:469–75. https://doi.org/10.1038/jp.2014.241.
Greig PC, et al. Maternal serum interleukin-6 during pregnancy and during term and preterm labor. Obstet Gynecol. 1997;90:465–9.
Arntzen KJ, Lien E, Austgulen R. Maternal serum levels of interleukin-6 and clinical characteristics of normal delivery at term. Acta Obstet Gynecol Scand. 1997;76:55–60.
Stallmach T, et al. Cytokine production and visualized effects in the feto-maternal unit. Quantitative and topographic data on cytokines during intrauterine disease. Lab Investig. 1995;73:384–92.
Mueller NT, et al. Birth mode-dependent association between pre-pregnancy maternal weight status and the neonatal intestinal microbiome. Sci Rep. 2016;6:23133 https://doi.org/10.1038/srep23133.
Mueller NT, et al. Does vaginal delivery mitigate or strengthen the intergenerational association of overweight and obesity? Findings from the Boston Birth Cohort. Int J Obes. 2017;41:497–501. https://doi.org/10.1038/ijo.2016.219.
Kozyrskyj AL, Kalu R, Koleva PT, Bridgman SL. Fetal programming of overweight through the microbiome: boys are disproportionately affected. J Dev Orig Health Dis. 2016;7:25–34. https://doi.org/10.1017/S2040174415001269.
Funding
This study was supported by a grant from the National Institutes of Health (R01DK094841). The funders had no role in the design of the study, data collection and analysis, decision to publish, or preparation of the manuscript. NTM is supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health under award number K01HL141589, and by grants from the Mid-Atlantic Nutrition Obesity Research Center (P30DK072488) and the Foundation for Gender Specific Medicine. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Mueller, N.T., Zhang, M., Hoyo, C. et al. Does cesarean delivery impact infant weight gain and adiposity over the first year of life?. Int J Obes 43, 1549–1555 (2019). https://doi.org/10.1038/s41366-018-0239-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-018-0239-2
This article is cited by
-
Pre-labor and post-labor cesarean delivery and early childhood adiposity in the Canadian Healthy Infant Longitudinal Development (CHILD) Cohort Study
International Journal of Obesity (2024)
-
Cord blood epigenome-wide meta-analysis in six European-based child cohorts identifies signatures linked to rapid weight growth
BMC Medicine (2023)
-
Associations between KCNQ1 and ITIH4 gene polymorphisms and infant weight gain in early life
Pediatric Research (2022)
-
Association between mode of delivery and body mass index at 4-5 years in White British and Pakistani children: the Born in Bradford birth cohort
BMC Public Health (2021)
-
Mode of delivery, type of labor, and measures of adiposity from childhood to teenage: Project Viva
International Journal of Obesity (2021)