Abstract
Background
In a Japanese macaque model of diet-induced obesity, we have previously demonstrated that consumption of a high-fat, “Western-style” diet (WSD) is associated with placental dysfunction and adverse pregnancy outcomes, independent of an obese maternal phenotype. Specifically, we have reported decreased uterine placental blood flow and increased inflammation with maternal WSD consumption. We also previously investigated the use of a promising therapeutic intervention that mitigated the adverse placental effects of a WSD but had unexpected detrimental effects on fetal pancreatic development. Thus, the objective of the current study was to determine whether simple preconception diet reversal (REV) would improve placental function.
Methods
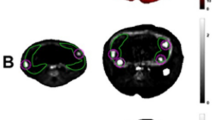
Female Japanese macaques were divided into three groups: REV animals (n = 5) were switched from a chronic WSD (36% fat) to a low fat, CON diet (14% fat) prior to conception and throughout pregnancy. The CON (n = 6) and WSD (n = 6) cohorts were maintained on their respective diets throughout pregnancy. Maternal body weight and composition were regularly assessed and advanced noninvasive imaging was performed at midgestation (gestational day 90, G90, or 0.5 of gestation, where full term is G175), and G129, 1 day prior to C-section delivery at G130 (0.75 of gestation). Imaging studies comprised Doppler ultrasound (US), contrast-enhanced US, and dynamic contrast-enhanced magnetic resonance imaging to assess uteroplacental hemodynamics and maternal-side placental perfusion.
Results
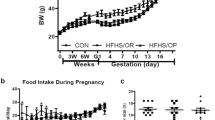
Dietary intervention resulted in significant maternal weight loss prior to pregnancy, and improved lean to fat mass ratio. By advanced imaging we demonstrated that a chronic WSD led to decreased blood flow velocity in the intervillous space, delayed blood flow transfer through the maternal spiral arteries, and reduced total placental blood flow compared to CON fed animals. Dietary reversal ameliorated these concerning derangements, restoring these hemodynamic parameters to CON levels.
Conclusions
Preconception dietary modification has beneficial effects on the maternal metabolic phenotype, and results in improved placental hemodynamics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity in the United States, 2009–2010. NCHS Data Brief. 2012;82:1–8.
Fisher SC, Kim SY, Sharma AJ, Rochat R, Morrow B. Is obesity still increasing among pregnant women? Prepregnancy obesity trends in 20 states, 2003–2009. Prev Med. 2013;56:372–8.
Schummers L, Hutcheon JA, Bodnar LM, Lieberman E, Himes KP. Risk of adverse pregnancy outcomes by prepregnancy body mass index: a population-based study to inform prepregnancy weight loss counseling. Obstet Gynecol. 2015;125:133–43.
Kidron D, Bernheim J, Aviram R. Placental findings contributing to fetal death, a study of 120 stillbirths between 23 and 40 weeks gestation. Placenta. 2009;30:700–4.
Nohr EA, Bech BH, Davies MJ, Frydenberg M, Henriksen TB, Olsen J. Prepregnancy obesity and fetal death: a study within the Danish National Birth Cohort. Obstet Gynecol. 2005;106:250–9.
Roberts DJ, Post MD. The placenta in preeclampsia and intrauterine growth restriction. J Clin Pathol. 2008;61:1254–60.
Challier JC, Basu S, Bintein T, et al. Obesity in pregnancy stimulates macrophage accumulation and inflammation in the placenta. Placenta. 2008;29:274–81.
Cnattingius S, Bergstrom R, Lipworth L, Kramer MS. Prepregnancy weight and the risk of adverse pregnancy outcomes. N Engl J Med. 1998;338:147–52.
Stewart FM, Freeman DJ, Ramsay JE, Greer IA, Caslake M, Ferrell WR. Longitudinal assessment of maternal endothelial function and markers of inflammation and placental function throughout pregnancy in lean and obese mothers. J Clin Endocrinol Metab. 2007;92:969–75.
Frias AE, Morgan TK, Evans AE, et al. Maternal high-fat diet disturbs uteroplacental hemodynamics and increases the frequency of stillbirth in a nonhuman primate model of excess nutrition. Endocrinology. 2011;152:2456–64.
Roberts VH, Pound LD, Thorn SR, et al. Beneficial and cautionary outcomes of resveratrol supplementation in pregnant nonhuman primates. FASEB J. 2014;28:2466–77.
Hayes EK, Lechowicz A, Petrik JJ, et al. Adverse fetal and neonatal outcomes associated with a life-long high-fat diet: role of altered development of the placental vasculature. PLoS One. 2012;7:e33370.
Liang C, DeCourcy K, Prater MR. High-saturated-fat diet induces gestational diabetes and placental vasculopathy in C57BL/6 mice. Metabolism. 2010;59:943–50.
Wallace JM, Bourke DA, Aitken RP, Palmer RM, Da Silva P, Cruickshank MA. Relationship between nutritionally mediated placental growth restriction and fetal growth, body composition and endocrine status during late gestation in adolescent sheep. Placenta. 2000;21:100–8.
McCurdy CE, Bishop JM, Williams SM, et al. Maternal high-fat diet triggers lipotoxicity in the fetal livers of nonhuman primates. J Clin Invest. 2009;119:323–35.
McCurdy CE, Schenk S, Hetrick B, et al. Maternal obesity reduces oxidative capacity in fetal skeletal muscle of Japanese macaques. JCI Insight. 2016;1:e86612.
Nicol LE, Grant WF, Comstock SM, et al. Pancreatic inflammation and increased islet macrophages in insulin-resistant juvenile primates. J Endocrinol. 2013;217:207–13.
Pound LD, Comstock SM, Grove KL. Consumption of a Western-style diet during pregnancy impairs offspring islet vascularization in a Japanese macaque model. Am J Physiol Endocrinol Metab. 2014;307:E115–123.
Guttmacher AE, Maddox YT, Spong CY. The Human Placenta Project: placental structure, development, and function in real time. Placenta. 2014;35:303–4.
Kaufmann BA, Wei K, Lindner JR. Contrast echocardiography. Curr Probl Cardiol. 2007;32:51–96.
Roberts VH, Lo JO, Salati JA, et al. Quantitative assessment of placental perfusion by contrast-enhanced ultrasound in macaques and human subjects. Am J Obstet Gynecol. 2016;214:369 e361–368.
Roberts VHJ, Lo JO, Lewandowski KS, et al. Adverse placental perfusion and pregnancy outcomes in a new nonhuman primate model of gestational protein restriction. Reprod Sci. 2017;25:110–9.
Frias AE, Schabel MC, Roberts VH, et al. Using dynamic contrast-enhanced MRI to quantitatively characterize maternal vascular organization in the primate placenta. Magn Reson Med. 2015;73:1570–8.
Lo JO, Schabel MC, Roberts VH, et al. Vitamin C supplementation ameliorates the adverse effects of nicotine on placental hemodynamics and histology in nonhuman primates. Am J Obstet Gynecol. 2015;212:370 e371–378.
Roberts VH, Rasanen JP, Novy MJ, et al. Restriction of placental vasculature in a nonhuman primate: a unique model to study placental plasticity. Placenta. 2012;33:73–6.
Lee JY, Kim MC. Anesthesia of growing pigs with tiletamine-zolazepam and reversal with flumazenil. J Vet Med Sci. 2012;74:335–9.
Acharya G, Sitras V, Erkinaro T, et al. Experimental validation of uterine artery volume blood flow measurement by Doppler ultrasonography in pregnant sheep. Ultrasound Obstet Gynecol. 2007;29:401–6.
Konje JC, Kaufmann P, Bell SC, Taylor DJ. A longitudinal study of quantitative uterine blood flow with the use of color power angiography in appropriate for gestational age pregnancies. Am J Obstet Gynecol. 2001;185:608–13.
Schabel MC, Morrell GR. Uncertainty in T(1) mapping using the variable flip angle method with two flip angles. Phys Med Biol. 2009;54:N1–8.
Oh KY, Roberts VH, Schabel MC, Grove KL, Woods M, Frias AE. Gadolinium chelate contrast material in pregnancy: fetal biodistribution in the nonhuman primate. Radiology. 2015;276:110–8.
Prola-Netto J, Woods M, Roberts VHJ, et al. Gadolinium chelate safety in pregnancy: barely detectable gadolinium levels in the juvenile nonhuman primate after in utero exposure. Radiology. 2017;286:112–28.
Mayhew TM, Burton GJ. Methodological problems in placental morphometry: apologia for the use of stereology based on sound sampling practice. Placenta. 1988;9:565–81.
Chernyavsky IL, Leach L, Dryden IL, Jensen OE. Transport in the placenta: homogenizing haemodynamics in a disordered medium. Philos Trans A Math Phys Eng Sci. 2011;369:4162–82.
Mayhew TM, Charnock-Jones DS, Kaufmann P. Aspects of human fetoplacental vasculogenesis and angiogenesis. III. Changes in complicated pregnancies. Placenta. 2004;25:127–39.
Chadderdon SM, Belcik JT, Bader L, et al. Proinflammatory endothelial activation detected by molecular imaging in obese nonhuman primates coincides with onset of insulin resistance and progressively increases with duration of insulin resistance. Circulation. 2014;129:471–8.
Grant WF, Nicol LE, Thorn SR, Grove KL, Friedman JE, Marks DL. Perinatal exposure to a high-fat diet is associated with reduced hepatic sympathetic innervation in one-year old male Japanese macaques. PLoS One. 2012;7:e48119.
Rosenberg TJ, Garbers S, Chavkin W, Chiasson MA. Prepregnancy weight and adverse perinatal outcomes in an ethnically diverse population. Obstet Gynecol. 2003;102(5 Pt 1):1022–7.
Armitage JA, Taylor PD, Poston L. Experimental models of developmental programming: consequences of exposure to an energy rich diet during development. J Physiol. 2005;565(Pt 1):3–8.
Whitaker RC, Wright JA, Pepe MS, Seidel KD, Dietz WH. Predicting obesity in young adulthood from childhood and parental obesity. N Engl J Med. 1997;337:869–73.
Acknowledgments
The authors thank Diana Takahashi, Karalee Baquero, Jessica Walker, Peter Blundell, and Tyler Dean (Oregon National Primate Research Center) for technical assistance and guidance with the animal studies.
Funding
Supported by NIH #R21HD076265, and NIH #R24DK0909640. In addition, research reported in this publication was supported by the Office of the Director, National Institutes of Health of the National Institutes of Health under Award Number P51OD011092. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Salati, J.A., Roberts, V.H.J., Schabel, M.C. et al. Maternal high-fat diet reversal improves placental hemodynamics in a nonhuman primate model of diet-induced obesity. Int J Obes 43, 906–916 (2019). https://doi.org/10.1038/s41366-018-0145-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-018-0145-7