Abstract
Epoxyeicosatrienoic acids (EETs) are metabolites of arachidonic acid that are rapidly metabolized into diols by soluble epoxide hydrolase (sEH). sEH inhibition has been shown to increase the biological activity of EETs, which are known to have anti-inflammatory properties. However, the role of EETs in pulmonary fibrosis remains unexplored. Liquid chromatography with tandem mass spectrometry (LC-MS/MS) was used to analyze EETs in the lung tissues of patients with idiopathic pulmonary fibrosis (IPF, n = 29) and controls (n = 15), and the function of 11,12-EET was evaluated in in vitro and in vivo in pulmonary fibrosis models. EET levels in IPF lung tissues, including those of 8,9-EET, 11,12-EET, and 14,15-EET, were significantly lower than those in control tissues. The 11,12-EET/11,12-DHET ratio in human lung tissues also differentiated IPF from control tissues. 11,12-EET significantly decreased transforming growth factor (TGF)-β1-induced expression of α-smooth muscle actin (SMA) and collagen type-I in MRC-5 cells and primary fibroblasts from IPF patients. sEH-specific siRNA and 1-trifluoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) urea (TPPU; sEH inhibitor) also decreased TGF-β1-induced expression of α-SMA and collagen type-I in fibroblasts. Moreover, 11,12-EET and TPPU decreased TGF-β1-induced p-Smad2/3 and extracellular-signal-regulated kinase (ERK) expression in primary fibroblasts from patients with IPF and fibronectin expression in Beas-2B cells. TPPU decreased the levels of hydroxyproline in the lungs of bleomycin-induced mice. 11,12-EET or sEH inhibitors could inhibit pulmonary fibrosis by regulating TGF-β1-induced profibrotic signaling, suggesting that 11,12-EET and the regulation of EETs could serve as potential therapeutic targets for IPF treatment.
Similar content being viewed by others
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic progressive fibrosing interstitial pneumonia of unknown etiology characterized by irreversible loss of lung function and poor prognosis1. Aberrant wound healing responses to lung injury have been implicated in the pathogenesis of pulmonary fibrosis2. The characteristic progressive fibrosis in IPF is due to fibroblast activation, which is regulated by various metabolic mediators, including lipid metabolites3,4,5. Previous studies have suggested that metabolic dysregulation, such as reduced sphingolipid metabolism, altered fatty acid oxidation, and an increased number of dysfunctional mitochondria, contributes to the pathogenesis of IPF6,7,8. However, the metabolic mediators associated with the pathogenesis of IPF have not been fully identified.
Eicosanoid metabolites of arachidonic acid play various roles in disease pathogenesis, such as anti-inflammatory, antifibrogenic and antiapoptotic roles3,5,9,10. Some eicosanoids, including prostaglandin E2, lipoxin A4, and prostacyclin, have shown antifibrotic effects on fibrosis models3,11,12. Epoxyeicosatrienoic acids (EETs), including 5,6-EET, 8,9-EET, 11,12-EET, and 14,15-EET, are biologically active eicosanoids that are produced by the actions of cytochrome P450 (CYP) epoxygenases on arachidonic acid. EETs are rapidly metabolized by soluble epoxide hydrolase (sEH) into dihydroxyeicosatrienoic acids (DHETs), which are the less active diol forms, and inhibiting sEH activity increases the levels of EETs in tissues and plasma13. Previous studies have suggested that epoxide hydrolase and epoxygenase metabolites can serve as key mediators during the pathogenesis of fibrosis and as therapeutic targets14,15,16,17,18. In the lung, EETs are the dominant eicosanoids produced in response to microbial challenge19 and are involved in the development of many pulmonary diseases, including asthma20, chronic obstructive pulmonary disease21, and pulmonary fibrosis17. In a nephrectomized rat model, EETs inhibited α-SMA expression and TGF-β1-Smad signaling and protected against renal fibrosis14. Moreover, inhibiting sEH prevents renal15, liver16, heart22, and pulmonary fibrosis17. However, the compositional changes in EETs in the lung tissues of patients with IPF and the roles of EETs in the pathogenesis of IPF have not been completely explored.
In this study, we hypothesized that patients with IPF exhibited changes in eicosanoid composition, which would affect IPF pathogenesis. We performed eicosanoid profiling in human lung tissues using LC-MS/MS and found that the levels of EETs, especially 11,12-EET, were reduced in the lung tissues of patients with IPF compared to those in control subjects. The role of 11,12-EET was investigated using in vitro and in vivo pulmonary fibrosis models.
Materials and methods
Human subjects
All patients with IPF met the diagnostic criteria set by the American Thoracic Society/European Respiratory Society/Japanese Respiratory Society/Latin American Thoracic Association23. IPF lung tissues (n = 29) were obtained at the time of surgical lung biopsy, and control human lung tissues (n = 15) with no histological evidence of disease were obtained from the Bio-Resource Center of Asan Medical Center, Seoul, Republic of Korea.
Ethics statement
Lung tissue procurement was completed under Protocol #2016-1131, which was approved by the Institutional Review Board of Asan Medical Center, Seoul, Republic of Korea. Informed written consent was obtained from all study participants. Mouse experiments were performed in accordance with the Guiding Principles for the Care and Use of Animals, and protocols were approved by the Animal Care and Handling Committee of Asan Medical Center (protocol #2017-12-034). While conducting the experiments, care was taken so that animal suffering was minimized.
LC-MS/MS analysis of eicosanoids
Eicosanoids were extracted from human lung tissues (~20 mg) using solid phase extraction (SPE). Eicosanoids were eluted with 0.5 mL of methanol followed by 1.5 mL of ethyl acetate and analyzed by a liquid chromatography-tandem mass spectrometry system. Detailed protocols for the analysis of eicosanoids can be found in the online supplement.
Animal model
Six-week-old female C57BL/6J mice were obtained from Orient Bio (Seongnam, South Korea) and acclimatized for 2 weeks before the experiments. The mice were housed under specific pathogen-free conditions in an air conditioned (22 ± 2 °C) and humidity-controlled (45–55%) room under a 12-h light and 12-h dark cycle with ad libitum access to food and water. The mice were randomly divided into four groups and were administered (1) saline plus vehicle, (2) saline plus TPPU, (3) bleomycin (3 U/kg) plus vehicle, or (4) bleomycin plus TPPU. The mice were anesthetized with intraperitoneal injections of 50 mg/kg alfaxalone (Jurox, Rutherford, NSW, Australia) and 5 mg/kg xylazine (Bayer, Leverkusen, Germany). Saline or bleomycin in saline was injected intratracheally. After bleomycin administration, 0.25 mg/kg TPPU was administered immediately by intraperitoneal injection (5 days per week for 3 weeks). The mice were euthanized, and the lungs were harvested on day 21, snap frozen in liquid nitrogen, and stored at −80 °C.
Hydroxyproline assay
To estimate the amount of collagen in lung tissues, a hydroxyproline assay was performed using a commercial kit (BioVision, Milpitas, CA, USA) according to the manufacturer’s protocol. The lungs were weighed, homogenized in sterile water, and hydrolyzed in 12 N HCl at 120 °C for 3 h. The hydrolyzed samples were incubated with 4‐(dimethylamino) benzaldehyde for 90 min at 60 °C, and the absorbance of the oxidized hydroxyproline was measured at 560 nm. The amount of hydroxyproline is expressed as μg/mg of lung tissue.
Cell culture
Beas-2B cells, a human bronchial epithelial cell line (ATCC, Manassas, VA, USA), were cultured in bronchial epithelial cell culture medium (Lonza, Bend, OR, USA) at 37 °C in a humidified atmosphere containing 5% CO2. MRC-5 cells, a normal human fetal lung fibroblast cell line (ATCC), were maintained in Eagle’s minimal essential medium (ATCC) supplemented with 100 units/mL penicillin, 100 µg/mL streptomycin (Invitrogen, Carlsbad, CA, USA), and 10% fetal bovine serum (FBS, HyClone, Logan, UT, USA) at 37 °C in a humidified incubator containing 5% CO2. Cells were treated with 5 ng/mL TGF-β1 for 24 h in the presence or absence of 11,12-EET (Abcam, Cambridge, MA, USA), 1-trifluoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) urea (TPPU, Sigma-Aldrich, St. Louis, MO, USA) or sulphaphenazole (Abcam). To isolate primary fibroblasts, IPF lung tissues were cut into 1 × 1 mm2 pieces and cultured at 37 °C in a humidified atmosphere containing 5% CO2 for 7‒10 days, the medium was changed every 3 days. Cells at passages 3 to 7 were used for all experiments. Detailed protocols for other experiments can be found in the online supplement.
Statistical analysis
All values are represented as the mean ± standard error. Experiments were repeated at least three times, and the data were analyzed for statistical significance using GraphPad Prism 5 software (GraphPad Inc. La Jolla, CA, USA). For in vitro assays, significant differences were analyzed using one-way analysis of variance (ANOVA), followed by the Newman‒Keuls multiple comparison test when comparing >3 groups. Statistical analysis of eicosanoid levels in human lung tissues was performed using the Mann‒Whitney nonparametric test. Receiver operating characteristic (ROC) curve analysis was performed to confirm the optimal cutoff value of EETs for IPF diagnosis. All p-values were two-tailed, with statistical significance set at p < 0.05.
Results
EET expression is reduced in IPF lung tissues
The levels of eicosanoids in human IPF lung tissues (n = 29) and control tissues (n = 15) were measured using LC-MS/MS. The IPF group had lower lung function than the control group (Supplementary Table S1). Although the levels of several eicosanoids differed significantly between the IPF and control groups (Fig. 1a), EETs exhibited the most notable differences. The levels of EETs, including 8,9-EET (mean, 13.39 vs. 25.00 fmol/mg lung tissue, p < 0.001), 11,12-EET (mean, 2.07 vs. 4.87 fmol/mg lung tissue, p < 0.001), and 14,15-EET (mean, 3.44 vs. 7.19 fmol/mg lung tissue, p < 0.001), in IPF lung tissues were significantly lower than those in control lung tissues (Fig. 1b–d), and 4,5-EET was not detected. ROC curve analysis revealed that EETs were useful in discriminating IPF from control conditions; the optimal cutoff values of 8,9-EET, 11,12-EET, and 14,15-EET were 22.5 fmol/mg lung tissue (AUC 0.88, p < 0.001), 3.367 fmol/mg lung tissue (AUC 0.94, p < 0.001), and 4.285 fmol/mg lung tissue (AUC 0.87, p < 0.001), respectively, (Supplementary Fig. S1).
Quantification of arachidonic acid-related metabolites in human idiopathic pulmonary fibrosis (IPF; n = 29) or control lung tissues (n = 15) was performed using liquid chromatography with tandem mass spectrometry. a Heatmap visualization of arachidonic acid-related metabolites in human idiopathic pulmonary fibrosis or control lung tissues. Epoxyeicosatrienoic acids (EETs) and soluble epoxy hydrolase-related metabolites are labeled in the heatmap. b–d IPF lung tissues exhibited lower levels of EETs (8,9-EET [b], 11,12-EET [c], and 14,15-EET [d]) than control lung tissues. * indicates p < 0.0001 compared to control lung tissues.
Increased expression of sEH contributes to changes in EET levels in IPF lung tissues
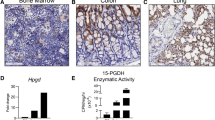
As EETs are rapidly metabolized into their corresponding diol forms by sEH, we measured the levels of DHETs. While the levels of 8,9-DHET and 11,12-DHET did not show significant differences between the two groups, the levels of 14,15-DHET in IPF lung tissues were significantly lower than those in the control (Supplementary Fig. S2). We also determined the activity of sEH by measuring the ratio of EETs to DHETs. The ratio of 11,12-EET/11,12-DHET in IPF lung tissues was significantly lower than that in the control (mean, 4.63 vs. 9.00 p < 0.01, Fig. 2b), while the ratios other EETs did not exhibit any significant differences between the two groups (Fig. 2a, c). We next measured the expression of sEH in lung tissues. The levels of sEH were significantly higher in IPF lung tissues than in control tissues (Fig. 2d). Our data suggest that the reduced levels of 11,12-EET in IPF lung tissues might be due to the upregulation of sEH.
a–c The ratio of 8,9-EET to 8,9-dihydroxyeicosatrienoic acid (DHET) (a), 11,12-EET to 11,12-DHET (b), and 14,15-EET to 14,15-DHET (c) in idiopathic pulmonary fibrosis (IPF) lung tissues compared with that in control lung tissues. ** indicates p < 0.01 compared to control lung tissues. d The levels of soluble epoxide hydrolase in IPF and control lung tissues were analyzed by western blotting. Densitometry was used to analyze fold changes in the levels of soluble epoxide hydrolase/α-actinin (p < 0.05). These data suggest that the reduced ratio of 11,12-EET to 11,12-DHET in IPF lung tissues might be due to the upregulated expression of soluble epoxide hydrolase.
11,12-EET and sEH inhibition suppresses fibroblast activation
TGF-β1 induces epithelial cell injury and stimulates fibroblast activation24. In MRC-5 cells, which are human lung fibroblasts, TGF-β1 increased the expression of α-SMA and collagen type 1, and 11,12-EET significantly reduced the TGF-β1-induced expression of these proteins (Supplementary Fig. S3a, b). We next examined the effects of the sEH inhibitor TPPU, which increased the levels of EETs. TPPU significantly reduced the TGF-β1-induced expression of α-SMA and collagen type 1 in MRC-5 cells (Supplementary Fig. S3c). Under identical conditions, 11,12-EET and TPPU did not significantly affect the cytotoxicity (Supplementary Fig. S4a, b) or viability (Supplementary Fig. S4e, f) of MRC-5 cells.
To ascertain whether sEH activity was essential for fibroblast activation, we investigated the effects of sEH knockdown using specific siRNA. MRC-5 cells were transfected with control siRNA or sEH-specific siRNA for 48 h and then treated with TGF-β1 for 24 h. TGF-β1 upregulated the expression of sEH, and sEH-specific siRNA effectively downregulated the basal expression or TGF-β1-induced expression of sEH (Supplementary Fig. S3d). sEH knockdown significantly inhibited the TGF-β1-induced expression of α-SMA and collagen type 1 (Supplementary Fig. S3d).
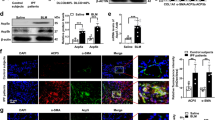
We confirmed our results in primary fibroblasts from patients with IPF. Consistent with the results in MRC-5 cells, 11,12-EET significantly reduced the TGF-β1-induced expression of α-SMA and collagen type 1 in primary fibroblasts (Fig. 3a, b). In addition, the inhibition of sEH by TPPU (Fig. 3c, d) and sEH-specific siRNA (Fig. 3e, f) reduced the effects of TGF-β1 on primary fibroblasts. These results suggest that 11,12-EET and sEH inhibition may reduce fibroblast activation.
a Primary human lung fibroblasts were treated with the indicated concentration of 11,12-EET and then stimulated with 5 ng/mL TGF-β1 for 24 h. Total cell extracts were prepared and subjected to western blotting using antibodies against collagen type 1, α-smooth muscle actin, and α-actinin. Representative immunoblots are shown. b Densitometry was used to analyze fold changes in the levels of α-smooth muscle actin (α-SMA) and collagen type 1 (Col-1). c Primary human lung fibroblasts were treated with the indicated amount of TPPU and then stimulated with 5 ng/mL TGF-β1 for 24 h. Total cell extracts were prepared and subjected to western blotting. d Densitometry was used to analyze fold changes in the levels of α-smooth muscle actin (α-SMA) and collagen type 1 (Col-1). e Primary human lung fibroblasts were transfected with control or soluble epoxide hydrolase siRNA for 48 h. Cells were treated with 5 ng/mL TGF-β1 for 24 h, and total cell extracts were prepared and subjected to western blotting. f Densitometry was used to analyze fold changes in the levels of α-smooth muscle actin (α-SMA) and collagen type 1 (Col-1). * indicates p < 0.05 compared to the control, and # indicates p < 0.05 compared to TGF-β1 treatment alone.
11,12-EET and sEH inhibition suppresses TGF-β1-induced Smad signaling in fibroblasts
TGF-β1 stimulates the Smad signaling pathway via the phosphorylation of Smad2/325 and activates the extracellular-signal-regulated kinase (ERK) pathway26. In this study, TGF-β1 increased the phosphorylation of Smad2/3 and ERK in primary fibroblasts from patients with IPF, and treatment with 11,12-EET and TPPU suppressed these effects (Fig. 4a, b). Moreover, as oxidative stress may contribute to pulmonary fibrosis27, we measured the levels of reactive oxygen species (ROS) and found that 11,12-EET and TPPU reduced TGFβ1-induced ROS levels in primary fibroblasts (Fig. 4c).
a Primary human lung fibroblasts were exposed to 5 ng/mL TGF-β1 for 1 h with/without 1 µM 11,12-EET. b Primary human lung fibroblasts were pretreated with 10 µM TPPU for 23 h and then stimulated with 5 ng/mL TGF-β1 for 1 h. Total cell extracts were prepared and subjected to western blotting using antibodies against p-Smad2/3, p-Erk, Erk, and α-actinin. c Primary human lung fibroblasts were pretreated with 1 µM 11,12-EET for 1 h or 10 µM TPPU for 23 h and then stimulated with 5 ng/mL TGF-β1 for 1 h. ROS levels were measured using 2’,7’-dichlorodihydrofluorescein diacetate dye. The data are expressed as the mean values of triplicate experiments. * and ** indicate p < 0.05 and p < 0.01, respectively.
Inhibition of CYP activity increases fibroblast activation
The most abundant cytochrome P450s (CYPs) in human lung tissue are CYP2C9, CYP1B1, CYP2B6, and CYP2E128. CYP2C9 is a good target for studying EET metabolism in the lung, as CYP2C and CYP2J metabolize arachidonic acid into EETs. Therefore, we investigated whether CYP2C9 activity was involved in fibroblast activation. The mRNA levels of CYP2C9 did not show significant differences between human IPF and control lung tissues (Fig. 5a). Sulphaphenazole, a CYP2C9 inhibitor, dose-dependently enhanced TGF-β1-induced expression of α-SMA in MRC-5 cells (Fig. 5b). To examine whether the effects of CYP2C9 inhibition were dependent on the levels of 11,12-EET, we investigated the effects of 11,12-EET on the TGF-β1- and sulphaphenazole-induced upregulation of α-SMA. 11,12-EET reduced the TGF-β1- and sulphaphenazole-induced upregulation of α-SMA in MRC-5 cells (Fig. 5c) and primary fibroblasts from patients with IPF (Fig. 5d).
a The mRNA levels of CYP2C9 in human idiopathic pulmonary fibrosis (IPF; n = 7) or control lung tissues (n = 7) were measured by real-time polymerase chain reaction. b MRC-5 cells were treated with the indicated amount of sulphaphenazole (Sulpha) and then stimulated with 5 ng/mL TGF-β1 for 24 h. Densitometry was used to analyze fold changes in the levels of α-smooth muscle actin (α-SMA). c MRC-5 cells were stimulated with 5 ng/mL TGF-β1 for 24 h with/without 20 µM sulphaphenazole and 1 µM 11,12-EET. Densitometry was used to analyze fold changes in the levels of α-smooth muscle actin (α-SMA). d Primary human lung fibroblasts were stimulated with 5 ng/mL TGF-β1 for 24 h with/without 20 µM sulphaphenazole and 1 µM 11,12-EET. Total cell extracts were prepared and subjected to western blotting using antibodies against α-smooth muscle actin and α-actinin. Densitometry was used to analyze fold changes in the levels of α-smooth muscle actin (α-SMA). * indicate p < 0.05.
11,12-EET reduces the expression of fibronectin in Beas-2B cells
To evaluate the effect of 11,12-EET on epithelial-mesenchymal transition (EMT) in the context lung fibrosis29, a migration assay was performed in Beas-2B cells. Beas-2B cells were treated with 11,12-EET or TPPU for 48 h, which resulted in significant reductions in TGF-β1-induced cell migration (Fig. 6a, b). To confirm the antifibrotic effect, we analyzed the expression of the epithelial cell marker E-cadherin and the mesenchymal cell marker fibronectin in TGF-β1-treated Beas-2B cells. The increased levels of fibronectin in TGF-β1-treated Beas-2B cells were reduced in response to 11,12-EET and TPPU treatment (Fig. 6c, d), whereas sulphaphenazole did not exert any effect (Supplementary Fig. S5c). However, 11,12-EET, TPPU, and sulphaphenazole did not affect the reductions in the levels of E-cadherin in TGF-β1-treated Beas-2B cells (Supplementary Fig. S5). Under identical conditions, 11,12-EET and TPPU did not affect the cytotoxicity (Supplementary Fig. S4c, d) or viability (Supplementary Fig. S4g, h) of Beas-2B cells. We next analyzed the mRNA expression of SLUG and VIM, which are TGF-β1-induced EMT-related genes30,31. TPPU significantly inhibited TGF-β1-induced mRNA levels of SLUG and VIM (Fig. 6e). Our results suggest that 11,12-EET and TPPU may exert antifibrotic effects on epithelial cells by reducing the expression of fibronectin.
a, b The migrated cells on the lower surface of the upper part of the transwell membrane were stained with 0.2% crystal violet and observed using a phase-contrast microscope (a). The stain was eluted, and the absorbance was measured at 540 nm (b). c, d Beas-2B cells were treated with 5 ng/mL TGF-β1 for 24 h with/without 1 μM 11,12-EET (c) or 10 μM TPPU (d). Total cell extracts were prepared and subjected to western blotting using antibodies against fibronectin and β-actin. e Beas-2B cells were treated with 5 ng/mL TGF-β1 for 24 h with/without 10 μM TPPU. The mRNA levels of SLUG and VIM were determined by real-time polymerase chain reaction. * indicates p < 0.01.
TPPU attenuates pulmonary fibrosis in a bleomycin-induced mouse model
To determine whether the in vitro effects of TPPU could be recapitulated in vivo, we evaluated the antifibrotic effects of TPPU on a bleomycin-induced mouse model. After intratracheal injection of bleomycin (3 U/kg), 0.25 mg/kg TPPU was immediately administered to the mice 5 days per week for 3 weeks by intraperitoneal injection, and the mice were euthanized after 21 days. The bleomycin-induced group showed severe weight loss after bleomycin administration compared to that of the control group; however, TPPU treatment significantly attenuated weight loss in the bleomycin-induced group (Fig. 7a). Histopathological examination of lung sections (Supplementary Fig. S6) and the fibrotic lesion scores (Fig. 7b) showed that TPPU prevented the development of bleomycin-induced pulmonary fibrosis. Mice treated with bleomycin showed significantly higher levels of collagen, as measured by hydroxyproline assays (Fig. 7c) and Mason’s trichrome staining (Fig. 7d), in lung tissues than control mice, while TPPU significantly attenuated bleomycin-induced increases in collagen levels. In summary, these data suggest that sEH inhibition by TPPU is a promising therapeutic strategy for pulmonary fibrosis.
a TPPU (0.25 mg/kg) was administered to mice 5 days/week for 3 weeks after bleomycin administration (3 U/kg). Body weights were analyzed in the four groups of mice: control (n = 3), bleomycin treatment (n = 4), TPPU treatment (n = 3), and bleomycin + TPPU treatment (n = 6). * and ** indicate p < 0.05 and p < 0.01, respectively, compared to the control group. # indicates p < 0.05 compared to the bleomycin treatment group. b Lung fibrosis scores were quantified by a modified Ashcroft scale (grade 0~8). c Collagen content was estimated by hydroxyproline assays. * and ** indicate p < 0.05 and p < 0.01, respectively (one-way ANOVA test with the Newman‒Keuls multiple comparison test). d Representative histological lung sections from each group stained with Masson’s trichrome.
Discussion
In the present study, we demonstrated changes in the levels of eicosanoids, including EETs, in IPF lung tissues. Among the EETs, the levels of 11,12-EET were the most significantly different between the IPF and control groups. 11,12-EET reduced the TGF-β1-induced activation of fibroblasts and epithelial cell EMT. TPPU also showed antifibrotic effects both in vitro and in vivo. These data suggest that 11,12-EET and the regulation of its metabolic pathway prevent pulmonary fibrosis.
The effect of EETs or sEH inhibitors on pulmonary fibrosis is associated with some signaling pathways, such as the Smad, p38 MAPK, and EMT-related signaling pathways29,32. In our study, the levels of EETs in IPF lung tissues were lower than those in control lung tissues. Therefore, we hypothesized that the dysregulation of EET-related signaling was associated with the development of pulmonary fibrosis. We found that 11,12-EET or sEH inhibitors downregulated TGF-β1-induced Smad2/3 and ERK pathways in TGF-β1-treated fibroblasts and downregulated EMT in TGF-β1-treated Beas-2B cells. Thus, our results suggest that the EET-related metabolic pathway can serve as a therapeutic target for IPF treatment.
Recent studies suggested that IPF develops as a result of an aberrant wound healing response after recurrent alveolar injury2 and that dysregulation of lipid metabolism is involved in the pathogenesis of IPF6,7,31. EETs serve as important antifibrotic lipid mediators in fibrotic diseases such as pulmonary and renal fibrosis17,18,33. In our study, EET levels were reduced in the lung tissues of patients with IPF compared to control lung tissues. This reduction may be due to the dysregulated activity of two key enzymes, sEH and CYP2C9, in the biosynthetic pathway of EETs. sEH, a key enzyme that is responsible for metabolizing EETs into DHETs, is widely distributed in mammalian tissues, including the lungs34. The inhibition of sEH activity by a pharmacological inhibitor or genetic disruption can induce the accumulation of EETs and exert protective effects against lung, liver, and renal fibrosis14,15,16,17. In our study, increased expression of sEH was observed in IPF lung tissues, and TPPU-mediated inhibition of sEH activity effectively reduced lung fibrosis in in vitro and in vivo in lung fibrosis models. Overall, our data suggest that the regulation of EET-related enzymes may be an important strategy in the treatment of IPF.
Additionally, the hydration of EETs by epoxide hydrolase is regioselective35. While 5,6-EET is a poor substrate of sEH, 8,9-EET, 11,12-EET and 14,15-EET are effectively utilized by sEH36. Moreover, among 8,9-EET, 11,12-EET and 14,15-EET, the hydration rate of 11,12-EET by lung cytosolic epoxide hydrolase is the lowest35. However, in the present study, we only observed a reduction in the 11,12-EET/11,12-DHET ratio in IPF lung tissues, and the 8,9-EET/8,9-DHET and 14,15-EET/14,15-DHET ratios were not significantly different between the IPF and control lung tissues. These results might be associated with the increases in 8,9-DHET and 14,15-DHET levels, while higher hydration rates for a long time might cause 8,9-EET and 14,15-EET to act as poor substrates of sEH to maintain homeostasis or might be associated with the excretion ratio of diol products.
CYP2C9 is one of the CYPs expressed in human lung tissues28 that metabolizes arachidonic acid into EETs, thereby eliciting various physiological responses, including antifibrotic effects17,37. The production of 11,12-EET by CYP2C9 in endothelial cells induces the expression of COX-238, and the COX-2-PGE2 axis exerts an antifibrotic effect by promoting the survival of epithelial cells and the death of fibroblasts/myofibroblasts in IPF3, suggesting that CYP2C9 may be involved in the pathogenesis of IPF. However, the role of CYP2C9 in lung fibrosis is incompletely understood. We therefore tested the hypothesis that sulphaphenazole-mediated inhibition of CYP2C9 could reduce the production of 11,12-EET and exacerbate lung fibrosis. We found that sulphaphenazole increased the TGF-β1-induced upregulation of α-SMA in human lung fibroblasts, which was reduced by treatment with 11,12-EET. These data suggest that 11,12-EET production might increase due to the activity of CYP2C9, thereby reducing the activation of human lung fibroblasts via the regulation of the COX-2-PGE2 axis.
In conclusion, our data suggest that 11,12-EET and sEH inhibition may prevent pulmonary fibrosis by inhibiting the activation of fibroblasts and epithelial cell EMT. Therefore, the regulation of EETs might serve as a therapeutic strategy for IPF treatment.
References
Adegunsoye, A. & Strek, M. E. Therapeutic approach to adult fibrotic lung diseases. Chest 150, 1371–1386 (2016).
Richeldi, L., Collard, H. R. & Jones, M. G. Idiopathic pulmonary fibrosis. Lancet 389, 1941–1952 (2017).
Maher, T. M. et al. Diminished prostaglandin E2 contributes to the apoptosis paradox in idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 182, 73–82 (2010).
Tang, N. et al. Lysophosphatidic acid accelerates lung fibrosis by inducing differentiation of mesenchymal stem cells into myofibroblasts. J. Cell Mol. Med. 18, 156–169 (2014).
Castelino, F. V. Lipids and eicosanoids in fibrosis: emerging targets for therapy. Curr. Opin. Rheumatol. 24, 649–655 (2012).
Zhao, Y. D. et al. Metabolic heterogeneity of idiopathic pulmonary fibrosis: a metabolomic study. BMJ Open Respir. Res. 4, e000183 (2017).
Kang, Y. P. et al. Metabolic profiling regarding pathogenesis of idiopathic pulmonary fibrosis. J. Proteome Res. 15, 1717–1724 (2016).
Mora, A. L., Bueno, M. & Rojas, M. Mitochondria in the spotlight of aging and idiopathic pulmonary fibrosis. J. Clin. Invest. 127, 405–414 (2017).
Dennis, E. A. & Norris, P. C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 15, 511–523 (2015).
Huang, S. K. & Peters-Golden, M. Eicosanoid lipid mediators in fibrotic lung diseases: ready for prime time? Chest 133, 1442–1450 (2008).
Roach, K. M., Feghali-Bostwick, C. A., Amrani, Y. & Bradding, P. Lipoxin A4 attenuates constitutive and TGF-beta1-dependent profibrotic activity in human lung myofibroblasts. J. Immunol. 195, 2852–2860 (2015).
Soberman, R. J. & Christmas, P. Revisiting prostacyclin: new directions in pulmonary fibrosis and inflammation. Am. J. Physiol. Lung Cell Mol. Physiol. 291, L142–L143 (2006).
Imig, J. D., Navar, L. G., Roman, R. J., Reddy, K. K. & Falck, J. R. Actions of epoxygenase metabolites on the preglomerular vasculature. J. Am. Soc. Nephrol. 7, 2364–2370 (1996).
Zhao, G. et al. Delivery of AAV2-CYP2J2 protects remnant kidney in the 5/6-nephrectomized rat via inhibition of apoptosis and fibrosis. Hum. Gene Ther. 23, 688–699 (2012).
Chiang, C. W. et al. Genetic deletion of soluble epoxide hydrolase attenuates inflammation and fibrosis in experimental obstructive nephropathy. Mediators Inflamm. 2015, 693260 (2015).
Zhang, C. H. et al. Soluble epoxide hydrolase inhibition with t-TUCB alleviates liver fibrosis and portal pressure in carbon tetrachloride-induced cirrhosis in rats. Clin. Res. Hepatol. Gastroenterol. 42, 118–125 (2018).
Zhou, Y. et al. Soluble epoxide hydrolase inhibitor 1-trifluoromethoxyphenyl-3- (1-propionylpiperidin-4-yl) urea attenuates bleomycin-induced pulmonary fibrosis in mice. Cell Tissue Res. 363, 399–409 (2016).
Zhang, C. Y. et al. COX-2/sEH dual inhibitor PTUPB alleviates bleomycin-induced pulmonary fibrosis in mice via inhibiting senescence. FEBS J. 287, 1666–1680 (2020).
Kiss, L. et al. Epoxyeicosatrienoates are the dominant eicosanoids in human lungs upon microbial challenge. Eur. Respir. J. 36, 1088–1098 (2010).
Chung, K. F. Lipoxins and epoxyeicosatrienoic acids. Potential for inhibitors of soluble epoxide hydrolase in severe asthma? Am. J. Respir. Crit. Care Med. 190, 848–850 (2014).
Ma, W. J. et al. Epoxyeicosatrienoic acids attenuate cigarette smoke extract-induced interleukin-8 production in bronchial epithelial cells. Prostaglandins Leukot. Ess. Fat. Acids 94, 13–19 (2015).
Zhou, C. et al. Pharmacological inhibition of soluble epoxide hydrolase ameliorates chronic ethanol-induced cardiac fibrosis by restoring autophagic flux. Alcohol Clin. Exp. Res. 42, 1970–1978 (2018).
Raghu, G. et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am. J. Respir. Crit. Care Med. 183, 788–824 (2011).
Sakai, N. & Tager, A. M. Fibrosis of two: Epithelial cell-fibroblast interactions in pulmonary fibrosis. Biochim Biophys. Acta 1832, 911–921 (2013).
Chitra, P., Saiprasad, G., Manikandan, R. & Sudhandiran, G. Berberine inhibits Smad and non-Smad signaling cascades and enhances autophagy against pulmonary fibrosis. J. Mol. Med. (Berl.) 93, 1015–1031 (2015).
Biernacka, A., Dobaczewski, M. & Frangogiannis, N. G. TGF-beta signaling in fibrosis. Growth Factors 29, 196–202 (2011).
Cheresh, P., Kim, S. J., Tulasiram, S. & Kamp, D. W. Oxidative stress and pulmonary fibrosis. Biochim. Biophys. Acta 1832, 1028–1040 (2013).
Castell, J. V., Donato, M. T. & Gomez-Lechon, M. J. Metabolism and bioactivation of toxicants in the lung. The in vitro cellular approach. Exp. Toxicol. Pathol. 57(Suppl 1), 189–204 (2005).
Liang, Y. et al. Soluble epoxide hydrolase inhibition ameliorates proteinuria-induced epithelial-mesenchymal transition by regulating the PI3K-Akt-GSK-3beta signaling pathway. Biochem Biophys. Res Commun. 463, 70–75 (2015).
Naber, H. P., Drabsch, Y., Snaar-Jagalska, B. E., ten Dijke, P. & van Laar, T. Snail and Slug, key regulators of TGF-beta-induced EMT, are sufficient for the induction of single-cell invasion. Biochem. Biophys. Res. Commun. 435, 58–63 (2013).
Kim, H. S. et al. Stearic acid attenuates profibrotic signalling in idiopathic pulmonary fibrosis. Respirology 26, 255–263 (2021).
Dong, X. W. et al. Soluble epoxide hydrolase inhibitor AUDA decreases bleomycin-induced pulmonary toxicity in mice by inhibiting the p38/Smad3 pathways. Toxicology 389, 31–41 (2017).
Skibba, M. et al. Epoxyeicosatrienoic acid analog decreases renal fibrosis by reducing epithelial-to-mesenchymal transition. Front. Pharm. 8, 406 (2017).
Harris, T. R. & Hammock, B. D. Soluble epoxide hydrolase: gene structure, expression and deletion. Gene 526, 61–74 (2013).
Zeldin, D. C. et al. Regio- and enantiofacial selectivity of epoxyeicosatrienoic acid hydration by cytosolic epoxide hydrolase. J. Biol. Chem. 268, 6402–6407 (1993).
Nayeem, M. A. Role of oxylipins in cardiovascular diseases. Acta Pharm. Sin. 39, 1142–1154 (2018).
Singh, S. P. et al. CYP-450 epoxygenase derived epoxyeicosatrienoic acid contribute to reversal of heart failure in obesity-induced diabetic cardiomyopathy via PGC-1 alpha activation. Cardiovasc. Pharm. Open Access 7, 1000233 (2018).
Michaelis, U. R., Falck, J. R., Schmidt, R., Busse, R. & Fleming, I. Cytochrome P4502C9-derived epoxyeicosatrienoic acids induce the expression of cyclooxygenase-2 in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 25, 321–326 (2005).
Acknowledgements
This study was supported by a Basic Science Research Program grant through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (NRF-2016R1A2B4016318, NRF-2019R1A2C2008541). H.S.K. was supported by a grant from the Basic Science Research Program through the National Research Foundation of Korea (NRF-2017R1D1A1B03030214) and a VHS Medical Center Research Grant (VHSMC20025). H.J.Y. was supported by a grant from the Basic Science Research Program through the National Research Foundation of Korea (NRF-2016R1D1A1B03935517).
Author information
Authors and Affiliations
Contributions
J.W.S. takes full responsibility for the content of this manuscript, including the data and analyses. H.S.K. and J.W.S. made substantial contributions to the concept and design of the study. H.S.K., S.M., and S.E.L. performed experiments. H.S.K., H.J.Y., and J.W.S. analyzed the data. H.S.K. and J.W.S. wrote the first draft of the manuscript. All authors discussed the results and reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, H.S., Moon, SJ., Lee, S.E. et al. The arachidonic acid metabolite 11,12-epoxyeicosatrienoic acid alleviates pulmonary fibrosis. Exp Mol Med 53, 864–874 (2021). https://doi.org/10.1038/s12276-021-00618-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s12276-021-00618-7
This article is cited by
-
Blood lipid profiles as a prognostic biomarker in idiopathic pulmonary fibrosis
Respiratory Research (2024)
-
Epoxyeicosatrienoic Acids Attenuate LPS-Induced NIH/3T3 Cell Fibrosis through the A2AR and PI3K/Akt Signaling Pathways
Bulletin of Experimental Biology and Medicine (2024)