Abstract
The gastrointestinal tract is a specialized organ in which dynamic interactions between host cells and the complex environment occur in addition to food digestion. Together with the chemical barrier of the mucosal layer and the cellular immune system, the epithelial cell layer performs a pivotal role as the first physical barrier against external factors and maintains a symbiotic relationship with commensal bacteria. The tight junction proteins, including occludin, claudins, and zonula occludens, are crucial for the maintenance of epithelial barrier integrity. To allow the transport of essential molecules and restrict harmful substances, the intracellular signaling transduction system and a number of extracellular stimuli such as cytokines, small GTPases, and post-translational modifications dynamically modulate the tight junction protein complexes. An imbalance in these regulations leads to compromised barrier integrity and is linked with pathological conditions. Despite the obscurity of the causal relationship, the loss of barrier integrity is considered to contribute to inflammatory bowel disease, obesity, and metabolic disorders. The elucidation of the role of diseases in barrier integrity and the underlying regulatory mechanisms have improved our understanding of the intestinal barrier to allow the development of novel and potent therapeutic approaches.
Similar content being viewed by others
Introduction
The intestinal epithelial layer forms the major barrier that separates our body from the external environment. Trillions of commensal bacteria reside in the gastrointestinal tract and have a vital role in digestion and the development of the immune system. However, they present a risk of infection1. The maintenance of the intestinal epithelial barrier is the essential function of the intestinal epithelial cells (IECs). The IECs integrate positive and negative interactions from the microbiota living in the gut and signal the immune cells to accommodate the microbiota, thereby perpetuating the normal function of the body2,3,4. An imbalance in the intestinal barrier structure can flare up into an uncontrollable immune reaction in the intestinal microenvironment or allow the unrestrained growth of microbiota, which leads to various diseases, including intestinal inflammatory disorders, extra-intestinal autoimmune diseases such as rheumatoid arthritis and multiple sclerosis, and metabolic disorders such as diabetes and obesity4,5,6. Critically ill patients and patients receiving chemotherapy/radiotherapy show severely compromised intestinal barrier integrity. Asymptomatic close relatives of patients with inflammatory bowel diseases (IBDs) also show compromised barrier integrity. Intestinal barrier permeability may therefore be a prognostic marker for disease pathophysiology; similarly, targeting the intestinal barrier permeability holds promise for therapy and for the prevention of disease.
Intestinal epithelial barrier
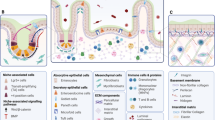
The intestinal epithelial barrier is a one-cell-thick internal lining of the gut that contains different types of epithelial cells. Underneath the epithelial layer, there is a thin layer of connective tissue, the lamina propria, which has a crucial role in nurturing healthy communication between the microbiome and the immune cells. The intestinal epithelial system is also home to immune cells, including dendritic cells, T cells, B cells, and macrophages, which function in close relation with the IECs to maintain intestinal homeostasis3,7. Gut microbiota, consisting of hundreds of trillions of bacteria and viruses, are pivotal for the maintenance of a symbiotic relationship with immune cells8,9,10. Recent studies have reported compelling evidence for the metabolic, immunological, and physiological roles had by the gut microbiota. However, the first layer of defense in the epithelium of the gut is formed by a layer of mucus, which is critical for the limitation of the exposure of epithelial cells to the microbiome11. The absence of mucin, a highly glycosylated polymeric protein in the mucous layer, makes an animal vulnerable to intestinal inflammation, which leads to the development of spontaneous colitis and confers a predisposition to the development of colorectal cancers (Fig. 1).
Epithelial cells form a layer that functions as a physical barrier facilitated by tight connections between each cell. A number of tight junction protein components seal the paracellular pathway and conduct gate and fence functions. The mucosal layer is a chemical barrier that is critical to limit the contact between the microbiome and epithelial cells. Immune cells are also a major participant in the immune response and the tolerance of the host against external substances. The graphical illustration was drawn by using the images from Servier Medical Art by Servier, with slight modifications (http://www.servier.com/Powerpoint-image-bank, https://creativecommons.org/licenses/by/3.0/)
Cellular functions of IECs
The intestinal epithelial layer is highly dynamic and characterized by a remarkable turnover rate; IECs are rapidly renewed and replaced every couple of days12,13. The maintenance of this cell layer renewal requires tight regulation to avoid any imbalance in homeostasis14,15. The intestinal epithelial monolayer is composed of different types of specialized epithelial cells, such as enterocytes, Paneth cells, goblet cells, endocytes, and microfold cells, each with a distinct function. The most abundant of these are IECs or enterocytes, for which the major function is the maintenance of epithelial barrier integrity16,17. Paneth cells reside in the base of crypts and secrete anti-microbial peptides, such as α-defensin, to impede microbial entry to the intestinal lumen. Goblet cells secrete mucous, trefoil peptides, and resistin-like molecule-β, which are central to both the defense and repair of the epithelial layer and have significant roles in epithelial homeostasis18,19,20. Endocytes regulate incoming antigens and microfold cells secrete IgA, which, in addition to goblet cells, helps present bacterial antigens to dendritic cells. Collectively, these cells form a polarized layer to establish a tight barrier by virtue of intracellular tight junctions, adherens junctions, and desmosomes.
IECs are capable of phagocytosing bacteria and can also sequester and neutralize bacterial toxins. These cells are also specialized to recognize bacterial-derived molecules, known as prokaryotic-associated molecular patterns, with the help of the Toll-like receptors on the cell surface and the nucleotide-binding oligomerization domain-like receptors in the cytoplasm, which activate defense mechanisms by the secretion of anti-microbial peptides2. IECs also maintain two-way communication with the underlying immune cells to regulate the inflammatory response against bacterial toxins21. In conjunction with the mucosal layer and specialized cells, the epithelial layer forms a well-equipped, intricately regulated and stringent barrier with continuous scrutiny by immune cells to create an immune-silent environment.
Mechanism of regulation of intestinal epithelial tight junction proteins
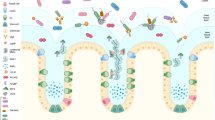
A crucial function of IECs is the maintenance of barrier integrity, which allows the permeability of essential ions, nutrients, and water but restricts the entry of bacterial toxins and pathogens21. The transport of molecules across the epithelial layer occurs through three major pathways: the trans-cellular pathway (passive diffusion across the cell membranes), the carrier-mediated pathway (carrier/receptor-mediated trans-cellular pathway), and the paracellular pathway (passive diffusion between the spaces through adjacent cells). The epithelial tight junction proteins, the most apical component of epithelial intracellular junctions, equip IECs with this function, which seals the paracellular space between the cells and tightly restricts the transport of hydrophilic molecules22,23,24. That is, the main function attributed to the tight junction proteins is the “gate and fence function,” which allows the paracellular transport of some solutes and molecules but prevents the intramembrane transport of proteins, lipids, and microbial-derived peptides25,26. Any alteration in the tight junction structure can prove to be detrimental to the organism.
The tight junction is composed of several transmembrane and cytosolic proteins, including occludin, claudins, zonula occludens (ZOs), tricellulin, cingulin, and junctional adhesion molecules (JAM), which interact with each other, as well as with the cytoskeleton, and form a complex architecture27. Most of these proteins, except for cingulin and ZO, are integral membrane proteins that extend into the paracellular spaces between the cells. Cingulin and ZOs are cytoskeletal linker proteins, which interact with the cytoplasmic peripheral membrane proteins, occludin, claudin, and JAM to form strong cross-links and interact with the membrane cytoskeleton composed of F-actin and myosin. Together with intracellular signaling proteins, tight junction proteins activate a plethora of cellular processes to maintain barrier integrity28. Tight junction complexes are the rate-limiting factor for paracellular permeability; they are programmed to rapidly open and seal the barrier in the event of injury and other signals. They form a highly dynamic entity, continuously transmitting signals to the individual components that undergo a series of regulations to enhance or modulate the integrity of the intestinal barrier.
Although it is well-accepted that tight junctions are crucial for the maintenance of barrier integrity, the exact function of the individual tight junction proteins remain elusive. Over the years, researchers have highlighted the diverse functions performed by tight junction proteins. Occludin, the first identified tight junction protein,29,30 has a dual role in the intestinal barrier; it provides structural integrity to the tight junction and is an integral component in the barrier function of tight junctions31. The expression level of occludin was found to be closely correlated with the barrier properties in vitro and in vivo32,33,34,35. Interestingly, occludin knockout mice had morphologically intact tight junctions but displayed complex histological phenotypes, with chronic inflammation and a defective epithelial barrier, which implicated that its crucial role was in tight junction stability rather than tight junction assembly. In contrast, certain other studies reported normal barrier function in occludin-deficient mice but showed chronic inflammation and hyperplasia in the gastric epithelium and testicular atrophy36,37. Severely compromised occludin expression has been observed in disease models of intestinal inflammatory diseases, which suggests it has a critical role in the maintenance of barrier integrity38,39,40. Collectively, these studies indicated that the functions of occludin are complex, and the mechanism by which occludin regulates the tight junction should be investigated in great detail.
Claudins, the other major tight junction proteins, are responsible for the regulation of paracellular space27,41,42. There are several isoforms of claudin, each having potentially different roles43,44, and a fine balance between them is needed for the maintenance of paracellular integrity. Alterations in the claudin levels can affect the intestinal barrier integrity in different ways depending on the type of claudin isoform45. For example, the downregulation of claudin 5 and 8 can drastically reduce the barrier integrity46; in contrast, claudin-2, a tight junction protein required for the formation of paracellular water channels that is highly expressed in leaky epithelial tissues, is upregulated in IBDs and promotes inflammation27,46,47. ZOs are peripheral membrane-associated proteins ubiquitously expressed in epithelial and endothelial cells. The various isoforms, ZO-1, ZO-2, and ZO-3, are all characterized by their ability to interact with different cellular proteins through a multitude of protein binding domains, such as the SH3 domain, the PDZ domain, and the leucine-zipper domain,48,49 and are also essential for scaffold formation and the connection of other tight junction proteins to the cytoskeleton. JAM-A, another tight junction protein, is also implicated in the maintenance of intestinal barrier integrity. JAM-A-deficient mice have increased barrier permeability with elevated bacterial translocation; however, they do not develop spontaneous colitis50,51. The roles of other tight junction proteins and their mechanism of action remain largely unknown.
Tight junction proteins are closely regulated, which is imperative for the maintenance of normal barrier integrity. IECs proliferate rapidly and renew quickly, and it is essential that the tight junction proteins are also strictly regulated to avoid any detrimental effect on membrane integrity52. They are also capable of efficiently adapting to the different demands of the cell by sealing, opening, and maintaining paracellular transport under various physiological and pathological conditions53.
The mechanism of the regulation of tight junction proteins is intricate and somewhat obscure. The tight junction proteins are regulated by multiple signaling proteins and signaling molecules. Several molecules involved in the signal transduction processes, including small GTP-binding proteins and tyrosine kinases, such as c-Src, c-Yes, and protein kinase C (PKC), have been found to be localized at these tight junctions, presumably indicating their pivotal role in the maintenance of tight junction integrity54,55. A significant body of evidence has highlighted the role of cytokines in the regulation of various tight junction proteins in a multitude of pathological conditions.
Tumor necrosis factor-α (TNFα), interferon-γ (IFN-γ), and interleukins all are well-known for their indisputable role in the regulation of tight junction integrity56. TNFα is a key player in the caveolin-1-mediated internalization of occludin, which elevates gut permeability; further, the overexpression of occludin alleviates the cytokine-induced increase in gut permeability57. TNFα stimulation of the NFκB signal transduction pathway is another major mechanism involved in tight junction regulation58. NFκB inhibition protected mice from severe water loss and diarrhea, which indicated its role in the regulation of the barrier property of IECs. The mechanism through which IFNγ modulates epithelial permeability is still under investigation; however, the acto-myosin cytoskeletal interaction with tight junction proteins is thought to be altered by IFNγ treatment59,60,61. IFNγ also induces an increase in barrier permeability through the reduction of ZO-1 and occludin expression in an adenosine monophosphate-activated protein kinase (AMPK)-dependent pathway, irrespective of the cellular energy levels62. The simultaneous presence of both these cytokines has a detrimental effect on intestinal integrity through the disassociation of tight junction proteins63,64.
A prominent player in cytokine-mediated tight junction regulation is myosin light chain kinase (MLCK)56,65,66, which disrupts the interaction between the tight junction proteins and the actin-myosin cytoskeleton, subsequently damaging the tight junction scaffold, which is crucial for the maintenance of barrier integrity66,67,68. TNF-mediated endocytosis of the tight junction requires enhanced MLCK transcription and activity at the tight junctions. Cytokines are also responsible for occludin redistribution from the tight junction to caveolin-containing vesicles69, and MLCK is also involved in the regulation of tight junction proteins through the alteration of ZO-1 protein dynamics70.
Another major mechanism of tight junction regulation is post-translational phosphorylation, which drastically alters their membrane distribution and turnover. Among the tight junction proteins, the post-translational phosphorylation of occludin has been widely studied and is responsible for opening and sealing tight junctions71. The phosphorylation status of occludin is intricately regulated by several kinases. Various research groups have suggested that serine/threonine phosphorylation is the predominant phosphorylation modification of occludin; however, recent research advances in this area have emphasized the importance of tyrosine phosphorylation. Hence, kinases such as PKC and c-Src and phosphatases, including PP2A, PP1, and PTP1B, which phosphorylate and dephosphorylate occludin, have a crucial role to play in intestinal barrier integrity72,73,74.
Occludin is highly phosphorylated at serine and threonine residues in the basal epithelium72. The specific kinases involved in occludin phosphorylation remain elusive; however, cellular/tight junction localization studies have suggested PKC as one of the cardinal players. Phosphorylated occludin interacts with ZO-1 and other tight junction proteins. An alteration in the phosphorylation pattern, such as an increase in tyrosine phosphorylation, which results from pathological conditions, such as inflammation or elevated ROS, can alter the protein–protein interactions of occludin with ZO-1, ZO-2, and ZO-3, and thereby alter the membrane integrity75. Oxidative-stress-induced intestinal permeability is thought to be mediated through the tyrosine phosphorylation of occludin and the redistribution of occludin, ZO-1, E-cadherin, and β-catenin from the intracellular junctions76,77. The tyrosine kinases involved in occludin phosphorylation remain largely obscure, although certain studies have speculated the role of c-Src family kinases in hydrogen peroxide-induced occludin tyrosine phosphorylation at the cellular level31,78. Tyrosine phosphorylated occludin could delocalize/disassemble from the tight junctions and undergo proteasome-mediated degradation, a phenomenon observed in patients suffering from inflammatory bowel syndrome79,80. The cellular energy sensor, AMPK, has been implicated in tight junction assembly by several studies81. AMPK is activated during the calcium switch-induced assembly of ZO-1, which facilitates tight junction assembly81. Butyrate, a short chain fatty acid abundant in the gut after the bacterial fermentation of carbohydrates, also induces tight junction assembly of ZO-1 and occludin through an AMPK-dependent pathway82. Although studies in this field have elucidated some of the mechanisms of tight junction regulation, in vivo studies that describe their role in pathological conditions are lacking. Despite the discoveries identifying the indisputable role of the post-translational phosphorylation of tight junction proteins, future studies to decipher the kinases and phosphatases involved, in addition to the putative binding partners and protein–protein interactions, are imperative for a better understanding of their regulatory mechanisms.
Regulation of barrier integrity during pathological conditions
The significance of the gut barrier in disease pathogenesis has recently attracted attention. Compromised intestinal barrier integrity is observed in both intestinal and systemic diseases, including IBDs, autoimmune diseases, and other metabolic diseases83. However, the scientific community has not yet determined whether the loss of barrier integrity is the cause or consequence of these diseases. Pathophysiological or environmental factors may be the crucial factors that usurp normal physiology and increase the permeability of the barrier. Hence, it is imperative to understand the factors that contribute to the loss of barrier integrity under pathological conditions (Fig. 2).
Inflammatory bowel disease and ulcerative colitis
IBD is a group of conditions in which patients have severe inflammation in the gastrointestinal tract84,85,86. It is a chronic condition that affects one out of 250 people in the European population as well as a significant population of people of other ethnic origins, and it has an unknown etiology, poor prognosis, and lifelong morbidity in patients87,88. There are two major classifications of IBD: ulcerative colitis (UC) and Crohn’s disease (CD). In UC, the inflammation is restricted to the rectum and the colon and never affects the small intestine; in contrast, in CD, there can be severe inflammation in any part of the intestine, including the small intestine and the large intestine89. The mechanism and etiology of disease progression are entirely different for these two types of IBD. Genetic predisposition to the disease is observed widely; in most cases, patients have at least one or more members in the family with the disease. Although the exact cause and mechanism of both types of IBD are yet to be completely understood, environmental or autoimmune-related factors are major causative factors90,91. The dysregulation of the mucosal immune response is also thought to be primarily responsible for disease progression92,93.
The loss of barrier integrity is a characteristic feature of IBD. A leaky gut may be an initial event in the pathogenesis of inflammatory bowel disorders, allowing bacteria-derived molecules into the mucosa and flaring up uncontrollable inflammatory signal cascades. The altered expression of tight junction proteins is observed in patients with UC and IBD94. Although there is an unresolved dispute over the contribution of the tight junction barrier to the disease pathology, recent studies have concluded that even in the presence of a normal underlying immunity, functional abnormalities of tight junction proteins could result in these diseases. In patients with CD, an increase in epithelial permeability precedes disease relapse, which emphasizes the essential role of tight junction proteins95,96. It has been reported that first-degree relatives of patients with IBD have abnormal intestinal permeability97, but it is still unclear whether individuals are genetically pre-disposed to compromised barrier integrity or if diet or environmental factors make them susceptible to the disease. The increase in apoptosis in IECs and cell shedding could also be responsible for the leakiness observed in these patients. However, some genetic studies have revealed a potential link between mutations in TJ-associated proteins, such as myosin IXB (MYO9B), partitioning defect protein (PARD3), PDZ containing protein 2 gene (MAGI2), and the development of IBD and celiac diseases98,99,100. All these studies reinforce the important role played by tight junction proteins and highlight the central role of epithelial barrier function in the pathogenesis of IBD.
Studies on in vivo experimental and spontaneous colitis models have identified the quintessential role played by tight junction proteins in the pathogenesis of UC. The roles of several kinases and proteins in the regulation of tight junction protein expression in pathological conditions were also investigated in these studies. Recently, it was reported in an experimental colitis model that dextran sodium sulfate (DSS) treatment, a chemical agent known to induce colitis, elevated the c-Src-mediated tyrosine phosphorylation of occludin in a phospholipase D2 (PLD2)-mediated pathway in PLD2 knockout mice. The inhibition of this pathway was shown to ameliorate DSS-induced colitis101.
Obesity and metabolic disorders
The loss of barrier integrity is closely associated with the onset of metabolic disorders, including obesity and type-II diabetes (T2D). Clinical studies have shown that increased intestinal permeability decreased to within the normal range after weight reduction in patients with obesity102. Recent studies have also demonstrated a difference in the intestinal permeability between individuals with or without T2D, which implicated a crucial contribution of intestinal permeability to metabolic disorders103. However, further studies are necessary before the regulation of barrier permeability can be introduced into clinical practice104.
Metabolic endotoxemia, arising from the loss of barrier integrity, is thought to be a major factor that contributes to insulin resistance and obesity105,106,107. Damage to the intestinal epithelial layer causes the leakage of gut microbiota-derived lipopolysaccharide (LPS) and other toxins into the blood stream, resulting in metabolic endotoxemia. Mice fed a high-fat diet for 4 weeks showed a three- to fourfold increase in serum LPS, which is defined as metabolic endotoxemia. This condition can subsequently lead to low-grade systemic inflammation and insulin resistance that is central to metabolic diseases. LPS itself is known to impact the increase in gut permeability. Physiologically relevant LPS concentrations (0–10 ng/ml) can induce intestinal tight junction permeability in enterocytes via an increase in TLR-4 and CD14 expression, without inducing cell death108. As bacterial-derived LPS has a key role in the increase in gut permeability, changes in the gut microbiota must be a contributory factor to the pathogenesis of obesity and diabetes109.
Changes in the gut microbial composition are a characteristic feature of many metabolic diseases; obese and diabetic individuals show drastic differences in their gut microbiomes compared with healthy counterparts, and the gut microbiome plays a pivotal role in the maintenance of barrier integrity110,111,112. The two major bacterial phyla in the gut, Bacteriodetes and Firmicutes, have been investigated widely for their effect on IECs. Mice fed with an high fat diet (HFD), or patients with obesity or diabetes show a change in this ratio with elevated levels of Firmicutes and Proteobacteria compared with the beneficial species, Bacteriodetes. Akkermansia muciniphila is one such beneficial bacterial species known to be less abundant in obese and diabetic individuals and has been implicated in the regulation of intestinal barrier integrity113. The daily administration of live A. muciniphila has been shown to mitigate HFD-induced gut barrier dysfunction114,115. Bacterial-derived components have also been reported to directly affect tight junction assembly. Plovier et al.116 recently identified that a specific outer membrane protein of A. muciniphila, termed Amuc100, improved the gut barrier integrity and that administration of this protein alone could partly recapitulate the beneficial effects shown by A. muciniphila. Extracellular vesicles (EVs) secreted from bacteria are other major components that are known to interact directly with IECs and exert their function. EVs from A. muciniphila have been shown to improve the symptoms of DSS-induced colitis in mice117. Another study from our group recently showed that EVs derived from A. muciniphila improved barrier integrity and glucose tolerance and reduced body weight gain in HFD-fed mice (in press). The reduction in metabolic endotoxemia through the improvement of gut barrier permeability could therefore present an interesting strategy to treat metabolic disorders such as obesity and diabetes.
Future perspectives
Recent studies have provided substantial evidence for the role of intestinal permeability in the regulation of several intestinal and extra-intestinal diseases. The improvement of barrier integrity by the regulation of tight junction protein expression or through other mechanisms has shown promising results with improvement of the disease symptoms in UC, CD, and metabolic diseases; strategies to identify and develop novel therapeutic targets to improve gut barrier integrity have become increasingly more attractive. The improvement of gut barrier integrity alone might not be sufficient in severe inflammatory diseases. However, in combination with conventional immunosuppressant drugs, such as TNFα inhibitors, approaches to improve intestinal barrier might prove beneficial. Metagenomic studies on gut microbiota from individuals with various diseases have shown that gut microbiota also actively interact with IECs for the regulation of barrier integrity. Various research groups are also investigating the strategy of probiotic and prebiotic administration to improve intestinal barrier integrity, as well as metabolic diseases. However, therapeutic intervention for the regulation of barrier integrity is an emerging topic and more investigations are essential to understand the role of intestinal barrier integrity in various diseases. The elucidation of signaling pathways involved in the regulation of the tight junction would allow the identification of novel barrier-restoring agents, which is imperative for deciphering novel and potent approaches for disease treatment.
References
Koch, S. & Nusrat, A. The life and death of epithelia during inflammation: lessons learned from the gut. Ann. Rev. Pathol. 7, 35–60 (2012).
Artis, D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat. Rev. Immunol. 8, 411–420 (2008).
Eri, R. & Chieppa, M. Messages from the inside. The dynamic environment that favors intestinal homeostasis. Front. Immunol. 4, 323 (2013).
De Mey, J. R. & Freund, J. N. Understanding epithelial homeostasis in the intestine: an old battlefield of ideas, recent breakthroughs and remaining controversies. Tissue Barriers 1, e24965 (2013).
Madara, J. L., Nash, S., Moore, R. & Atisook, K. Structure and function of the intestinal epithelial barrier in health and disease. Monogr. Pathol. 31, 306–324 (1990).
Catalioto, R. M., Maggi, C. A. & Giuliani, S. Intestinal epithelial barrier dysfunction in disease and possible therapeutical interventions. Curr. Med. Chem. 18, 398–426 (2011).
Saenz, S. A., Taylor, B. C. & Artis, D. Welcome to the neighborhood: epithelial cell-derived cytokines license innate and adaptive immune responses at mucosal sites. Immunol. Rev. 226, 172–190 (2008).
Guinane, C. M. & Cotter, P. D. Role of the gut microbiota in health and chronic gastrointestinal disease: understanding a hidden metabolic organ. Ther. Adv. Gastroenterol. 6, 295–308 (2013).
Kahrstrom, C. T., Pariente, N. & Weiss, U. Intestinal microbiota in health and disease. Nature 535, 47 (2016).
Clemente, J. C., Ursell, L. K., Parfrey, L. W. & Knight, R. The impact of the gut microbiota on human health: an integrative view. Cell 148, 1258–1270 (2012).
Yu, Y., Sitaraman, S. & Gewirtz, A. T. Intestinal epithelial cell regulation of mucosal inflammation. Immunol. Res. 29, 55–68 (2004).
Bjerknes, M. & Cheng, H. Gastrointestinal stem cells. II. Intest. stem Cells Am. J. Physiol. Gastrointest. Liver Physiol. 289, G381–G387 (2005).
Okamoto, R. & Watanabe, M. Molecular and clinical basis for the regeneration of human gastrointestinal epithelia. J. Gastroenterol. 39, 1–6 (2004).
Ruemmele, F. M., Seidman, E. G. & Lentze, M. J. Regulation of intestinal epithelial cell apoptosis and the pathogenesis of inflammatory bowel disorders. J. Pediatr. Gastroenterol. Nutr. 34, 254–260 (2002).
Williams, J. M. et al. Epithelial cell shedding and barrier function: a matter of life and death at the small intestinal villus tip. Vet. Pathol. 52, 445–455 (2015).
Groschwitz, K. R. & Hogan, S. P. Intestinal barrier function: molecular regulation and disease pathogenesis. J. Allergy Clin. Immunol. 124, 3–20 (2009). quiz 21-22.
Kagnoff, M. F. The intestinal epithelium is an integral component of a communications network. J. Clin. Invest. 124, 2841–2843 (2014).
Birchenough, G. M., Johansson, M. E., Gustafsson, J. K., Bergstrom, J. H. & Hansson, G. C. New developments in goblet cell mucus secretion and function. Mucosal Immunol. 8, 712–719 (2015).
Kim, Y. S. & Ho, S. B. Intestinal goblet cells and mucins in health and disease: recent insights and progress. Curr. Gastroenterol. Rep. 12, 319–330 (2010).
Specian, R. D. & Oliver, M. G. Functional biology of intestinal goblet cells Am. J. Physiol. 260, C183–C193 (1991).
Rescigno, M. The intestinal epithelial barrier in the control of homeostasis and immunity. Trends Immunol. 32, 256–264 (2011).
Suzuki, T. Regulation of intestinal epithelial permeability by tight junctions. Cell Mol. Life Sci. 70, 631–659 (2013).
Maloy, K. J. & Powrie, F. Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature 474, 298–306 (2011).
Khor, B., Gardet, A. & Xavier, R. J. Genetics and pathogenesis of inflammatory bowel disease. Nature 474, 307–317 (2011).
Diamond, J. M. Twenty-first Bowditch lecture. The epithelial junction: bridge, gate, and fence. Physiologist 20, 10–18 (1977).
Berkes, J., Viswanathan, V. K., Savkovic, S. D. & Hecht, G. Intestinal epithelial responses to enteric pathogens: effects on the tight junction barrier, ion transport, and inflammation. Gut 52, 439–451 (2003).
Gunzel, D. & Yu, A. S. Claudins and the modulation of tight junction permeability. Physiol. Rev. 93, 525–569 (2013).
Cereijido, M. et al. New diseases derived or associated with the tight junction. Med Res Arch. 38, 465–478 (2007).
Farquhar, M. G. & Palade, G. E. Junctional complexes in various epithelia. J. Cell Biol. 17, 375–412 (1963).
Furuse, M. et al. Occludin: a novel integral membrane protein localizing at tight junctions. J. Cell Biol. 123, 1777–1788 (1993).
Feldman, G. J., Mullin, J. M. & Ryan, M. P. Occludin: structure, function and regulation. Adv. Drug Deliv. Rev. 57, 883–917 (2005).
Kevil, C. G. et al. Expression of zonula occludens and adherens junctional proteins in human venous and arterial endothelial cells: role of occludin in endothelial solute barriers. Microcirculation 5, 197–210 (1998).
Balda, M. S. et al. Functional dissociation of paracellular permeability and transepithelial electrical resistance and disruption of the apical-basolateral intramembrane diffusion barrier by expression of a mutant tight junction membrane protein. J. Cell Biol. 134, 1031–1049 (1996).
Balda, M. S., Flores-Maldonado, C., Cereijido, M. & Matter, K. Multiple domains of occludin are involved in the regulation of paracellular permeability. J. Cell Biochem. 78, 85–96 (2000).
Chen, Y., Merzdorf, C., Paul, D. L. & Goodenough, D. A. COOH terminus of occludin is required for tight junction barrier function in early Xenopus embryos. J. Cell Biol. 138, 891–899 (1997).
Schulzke, J. D. et al. Epithelial transport and barrier function in occludin-deficient mice. Biochim. Biophys. Acta 1669, 34–42 (2005).
Saitou, M. et al. Complex phenotype of mice lacking occludin, a component of tight junction strands. Mol. Biol. Cell 11, 4131–4142 (2000).
Li, X. et al. Somatostatin regulates tight junction proteins expression in colitis mice. Int. J. Clin. Exp. Pathol. 7, 2153–2162 (2014).
Nighot, P. et al. Matrix metalloproteinase 9-induced increase in intestinal epithelial tight junction permeability contributes to the severity of experimental DSS colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 309, G988–G997 (2015).
Mennigen, R. et al. Probiotic mixture VSL#3 protects the epithelial barrier by maintaining tight junction protein expression and preventing apoptosis in a murine model of colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G1140–G1149 (2009).
Tsukita, S. & Furuse, M. Occludin and claudins in tight-junction strands: leading or supporting players? Trends Cell Biol. 9, 268–273 (1999).
Mrsny, R. J. et al. A key claudin extracellular loop domain is critical for epithelial barrier integrity. Am. J. Pathol. 172, 905–915 (2008).
Furuse, M., Furuse, K., Sasaki, H. & Tsukita, S. Conversion of zonulae occludentes from tight to leaky strand type by introducing claudin-2 into Madin-Darby canine kidney I cells J. Cell Biol. 153, 263–272 (2001).
Tsukita, S. & Furuse, M. Overcoming barriers in the study of tight junction functions: from occludin to claudin. Genes Cells 3, 569–573 (1998).
Findley, M. K. & Koval, M. Regulation and roles for claudin-family tight junction proteins. IUBMB Life 61, 431–437 (2009).
Zeissig, S. et al. Changes in expression and distribution of claudin-2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn’s disease. Gut 56, 61–72 (2007).
Ahmad, R. et al. Targeted colonic claudin-2 expression renders resistance to epithelial injury, induces immune suppression, and protects from colitis. Mucosal Immunol. 7, 1340–1353 (2014).
Itoh, M. et al. Direct binding of three tight junction-associated MAGUKs, ZO-1, ZO-2, and ZO-3, with the COOH termini of claudins. J. Cell Biol. 147, 1351–1363 (1999).
Beatch, M., Jesaitis, L. A., Gallin, W. J., Goodenough, D. A. & Stevenson, B. R. The tight junction protein ZO-2 contains three PDZ (PSD-95/Discs-Large/ZO-1) domains and an alternatively spliced region. J. Biol. Chem. 271, 25723–25726 (1996).
Laukoetter, M. G. et al. JAM-A regulates permeability and inflammation in the intestine in vivo. J. Exp. Med. 204, 3067–3076 (2007).
Khounlotham, M. et al. Compromised intestinal epithelial barrier induces adaptive immune compensation that protects from colitis. Immunity 37, 563–573 (2012).
Marchiando, A. M., Graham, W. V. & Turner, J. R. Epithelial barriers in homeostasis and disease. Ann. Rev. Pathol. 5, 119–144 (2010).
Harhaj, N. S. & Antonetti, D. A. Regulation of tight junctions and loss of barrier function in pathophysiology. Int. J. Biochem. Cell Biol. 36, 1206–1237 (2004).
Tsukita, S. et al. Specific proto-oncogenic tyrosine kinases of src family are enriched in cell-to-cell adherens junctions where the level of tyrosine phosphorylation is elevated. J. Cell Biol. 113, 867–879 (1991).
Zahraoui, A. et al. A small rab GTPase is distributed in cytoplasmic vesicles in non polarized cells but colocalizes with the tight junction marker ZO-1 in polarized epithelial cells J. Cell Biol. 124, 101–115 (1994).
Capaldo, C. T. & Nusrat, A. Cytokine regulation of tight junctions. Biochim. Biophys. Acta 1788, 864–871 (2009).
Marchiando, A. M. et al. Caveolin-1-dependent occludin endocytosis is required for TNF-induced tight junction regulation in vivo. J. Cell Biol. 189, 111–126 (2010).
Wang, F. et al. Interferon-gamma and tumor necrosis factor-alpha synergize to induce intestinal epithelial barrier dysfunction by up-regulating myosin light chain kinase expression. Am. J. Pathol. 166, 409–419 (2005).
Zolotarevsky, Y. et al. A membrane-permeant peptide that inhibits MLC kinase restores barrier function in in vitro models of intestinal disease. Gastroenterol 123, 163–172 (2002).
Utech, M. et al. Mechanism of IFN-gamma-induced endocytosis of tight junction proteins: myosin II-dependent vacuolarization of the apical plasma membrane. Mol. Biol. Cell 16, 5040–5052 (2005).
Bruewer, M. et al. Interferon-gamma induces internalization of epithelial tight junction proteins via a macropinocytosis-like process. FASEB J. 19, 923–933 (2005).
Scharl, M., Paul, G., Barrett, K. E. & McCole, D. F. AMP-activated protein kinase mediates the interferon-gamma-induced decrease in intestinal epithelial barrier function. J. Biol. Chem. 284, 27952–27963 (2009).
Ozaki, H. et al. Cutting edge: combined treatment of TNF-alpha and IFN-gamma causes redistribution of junctional adhesion molecule in human endothelial cells J. Immunol. 163, 553–557 (1999).
Ye, D., Ma, I. & Ma, T. Y. Molecular mechanism of tumor necrosis factor-alpha modulation of intestinal epithelial tight junction barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 290, G496–G504 (2006).
Graham, W. V. et al. Tumor necrosis factor-induced long myosin light chain kinase transcription is regulated by differentiation-dependent signaling events. Characterization of human long myosin light chain kinase promoter. J. Biol. Chem. 281, 26205–26215 (2006).
Su, L. et al. TNFR2 activates MLCK-dependent tight junction dysregulation to cause apoptosis-mediated barrier loss and experimental colitis. Gastroenterol 145, 407–415 (2013).
Yu, D. et al. MLCK-dependent exchange and actin binding region-dependent anchoring of ZO-1 regulate tight junction barrier function. Proc. . Natl Acad. Sci. USA 107, 8237–8241 (2010).
Shen, L. et al. Myosin light chain phosphorylation regulates barrier function by remodeling tight junction structure. J. Cell Sci. 119, 2095–2106 (2006).
Shen, L. Tight junctions on the move: molecular mechanisms for epithelial barrier regulation. Ann. N. Y. Acad. Sci. 1258, 9–18 (2012).
Cunningham, K. E. & Turner, J. R. Myosin light chain kinase: pulling the strings of epithelial tight junction function. Ann. N. Y. Acad. Sci. 1258, 34–42 (2012).
Farshori, P. & Kachar, B. Redistribution and phosphorylation of occludin during opening and resealing of tight junctions in cultured epithelial cells J. Membr. Biol. 170, 147–156 (1999).
Rao, R. Occludin phosphorylation in regulation of epithelial tight junctions. Ann. N. Y. Acad. Sci. 1165, 62–68 (2009).
Seth, A., Sheth, P., Elias, B. C. & Rao, R. Protein phosphatases 2A and 1 interact with occludin and negatively regulate the assembly of tight junctions in the CACO-2 cell monolayer. J. Biol. Chem. 282, 11487–11498 (2007).
Nunbhakdi-Craig, V. et al. Protein phosphatase 2A associates with and regulates atypical PKC and the epithelial tight junction complex. J. Cell Biol. 158, 967–978 (2002).
Kale, G., Naren, A. P., Sheth, P. & Rao, R. K. Tyrosine phosphorylation of occludin attenuates its interactions with ZO-1, ZO-2, and ZO-3. Biochem. Biophys. Res. Commun. 302, 324–329 (2003).
Wong, V. Phosphorylation of occludin correlates with occludin localization and function at the tight junction. Am. J. Physiol. 273, C1859–C1867 (1997).
Rao, R. K., Basuroy, S., Rao, V. U., Karnaky, K. J. Jr & Gupta, A. Tyrosine phosphorylation and dissociation of occludin-ZO-1 and E-cadherin-beta-catenin complexes from the cytoskeleton by oxidative stress. Biochem. J. 368, 471–481 (2002).
Basuroy, S. et al. Expression of kinase-inactive c-Src delays oxidative stress-induced disassembly and accelerates calcium-mediated reassembly of tight junctions in the Caco-2 cell monolayer. J. Biol. Chem. 278, 11916–11924 (2003).
Coeffier, M. et al. Increased proteasome-mediated degradation of occludin in irritable bowel syndrome. Am. J. Gastroenterol. 105, 1181–1188 (2010).
Raikwar, N. S., Vandewalle, A. & Thomas, C. P. Nedd4-2 interacts with occludin to inhibit tight junction formation and enhance paracellular conductance in collecting duct epithelia. Am. J. Physiol. Ren. Physiol. 299, F436–F444 (2010).
Zhang, L., Li, J., Young, L. H. & Caplan, M. J. AMP-activated protein kinase regulates the assembly of epithelial tight junctions. Proc. . Natl Acad. Sci. USA 103, 17272–17277 (2006).
Peng, L., Li, Z. R., Green, R. S., Holzman, I. R. & Lin, J. Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J. Nutr. 139, 1619–1625 (2009).
Ma, T. Y. Intestinal epithelial barrier dysfunction in Crohn’s disease. Proc. Soc. Exp. Biol. Med. Soc. Exp. Biol. Med. 214, 318–327 (1997).
Hanauer, S. B. Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflamm. Bowel Dis. 12(Suppl 1), S3–S9 (2006).
Schmidt, C. & Stallmach, A. Etiology and pathogenesis of inflammatory bowel disease. Minerva Gastroenterol. Dietol. 51, 127–145 (2005).
Leake, I. IBD: treatment for acute severe ulcerative colitis. Nat. Rev. Gastroenterol. Hepatol. 13, 436 (2016).
Nguyen, G. C. et al. Inflammatory bowel disease characteristics among African Americans, Hispanics, and non-Hispanic Whites: characterization of a large North American cohort. Am. J. Gastroenterol. 101, 1012–1023 (2006).
Karlinger, K., Gyorke, T., Mako, E., Mester, A. & Tarjan, Z. The epidemiology and the pathogenesis of inflammatory bowel disease. Eur. J. Radiol. 35, 154–167 (2000).
Sartor, R. B. Current concepts of the etiology and pathogenesis of ulcerative colitis and Crohn’s disease. Gastroenterol. Clin. North Am. 24, 475–507 (1995).
Xavier, R. J. & Podolsky, D. K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 448, 427–434 (2007).
Stefanelli, T., Malesci, A., Repici, A., Vetrano, S. & Danese, S. New insights into inflammatory bowel disease pathophysiology: paving the way for novel therapeutic targets. Curr. Drug. Targets 9, 413–418 (2008).
Kucharzik, T. et al. Recent understanding of IBD pathogenesis: implications for future therapies. Inflamm. Bowel Dis. 12, 1068–1083 (2006).
MacDermott, R. P. Alterations of the mucosal immune system in inflammatory bowel disease. J. Gastroenterol. 31, 907–916 (1996).
Takeuchi, K., Maiden, L. & Bjarnason, I. Genetic aspects of intestinal permeability in inflammatory bowel disease. Novartis Found. Symp. 263, 151–158 (2004). discussion 159-163, 211-158.
D’Inca, R. et al. Intestinal permeability test as a predictor of clinical course in Crohn’s disease. Am. J. Gastroenterol. 94, 2956–2960 (1999).
Arnott, I. D., Kingstone, K. & Ghosh, S. Abnormal intestinal permeability predicts relapse in inactive Crohn disease. Scand. J. Gastroenterol. 35, 1163–1169 (2000).
Soderholm, J. D. et al. Different intestinal permeability patterns in relatives and spouses of patients with Crohn’s disease: an inherited defect in mucosal defence? Gut 44, 96–100 (1999).
Wapenaar, M. C. et al. Associations with tight junction genes PARD3 and MAGI2 in Dutch patients point to a common barrier defect for coeliac disease and ulcerative colitis. Gut 57, 463–467 (2008).
van Bodegraven, A. A. et al. Genetic variation in myosin IXB is associated with ulcerative colitis. Gastroenterol 131, 1768–1774 (2006).
Monsuur, A. J. et al. Myosin IXB variant increases the risk of celiac disease and points toward a primary intestinal barrier defect. Nat. Genet. 37, 1341–1344 (2005).
Chelakkot, C. et al. Intestinal epithelial cell-specific deletion of PLD2 alleviates DSS-induced colitis by regulating occludin. Sci. Rep. 7, 1573 (2017).
Damms-Machado, A. et al. Gut permeability is related to body weight, fatty liver disease, and insulin resistance in obese individuals undergoing weight reduction. Am. J. Clin. Nutr. 105, 127–135 (2017).
Cox, A. J. et al. Increased intestinal permeability as a risk factor for type 2 diabetes. Diabetes Metab. 43, 163–166 (2017).
Fasano, A. Gut permeability, obesity, and metabolic disorders: who is the chicken and who is the egg?. Am. J. Clin. Nutr. 105, 3–4 (2017).
Cani, P. D. et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56, 1761–1772 (2007).
Cani, P. D. et al. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57, 1470–1481 (2008).
Moreira, A. P., Texeira, T. F., Ferreira, A. B., Peluzio Mdo, C. & Alfenas Rde, C. Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. Br. J. Nutr. 108, 801–809 (2012).
Guo, S., Al-Sadi, R., Said, H. M. & Ma, T. Y. Lipopolysaccharide causes an increase in intestinal tight junction permeability in vitro and in vivo by inducing enterocyte membrane expression and localization of TLR-4 and CD14. Am. J. Pathol. 182, 375–387 (2013).
Hur, K. Y. Is GDF15 a novel biomarker to predict the development of prediabetes or diabetes? Diabetes Metab. J. 38, 437–438 (2014).
Cani, P. D. et al. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 58, 1091–1103 (2009).
Cani, P. D., Osto, M., Geurts, L. & Everard, A. Involvement of gut microbiota in the development of low-grade inflammation and type 2 diabetes associated with obesity. Gut Microbes 3, 279–288 (2012).
Qin, J. et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60 (2012).
Dao, M. C. et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut 65, 426–436 (2016).
Everard, A. et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl Acad. Sci. USA 110, 9066–9071 (2013).
Shin, N. R. et al. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut 63, 727–735 (2014).
Plovier, H. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 23, 107–113 (2017).
Kang, C. S. et al. Extracellular vesicles derived from gut microbiota, especially Akkermansia muciniphila, protect the progression of dextran sulfate sodium-induced colitis. PLoS ONE 8, e76520 (2013).
Acknowledgements
This work was supported by the Samsung Research Funding Center of Samsung Electronics under Project Number SRFC-MA1502-14.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chelakkot, C., Ghim, J. & Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp Mol Med 50, 1–9 (2018). https://doi.org/10.1038/s12276-018-0126-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s12276-018-0126-x
This article is cited by
-
The protective role of prebiotics and probiotics on diarrhea and gut damage in the rotavirus-infected piglets
Journal of Animal Science and Biotechnology (2024)
-
Carotid artery vascular stenosis causes the blood-CSF barrier damage and neuroinflammation
Journal of Neuroinflammation (2024)
-
Intestinal Piezo1 aggravates intestinal barrier dysfunction during sepsis by mediating Ca2+ influx
Journal of Translational Medicine (2024)
-
Single-cell sequencing of the vermiform appendix during development identifies transcriptional relationships with appendicitis in preschool children
BMC Medicine (2024)
-
The effect of an acute aspirin challenge on intestinal permeability in healthy adults with and without prophylactic probiotic consumption: a double-blind, placebo-controlled, randomized trial
BMC Gastroenterology (2024)