Abstract
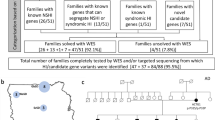
Hearing loss (HL) is a common heterogeneous trait that involves variants in more than 200 genes. In this study, we utilized exome (ES) and genome sequencing (GS) to effectively identify the genetic cause of presumably non-syndromic HL in 322 families from South and West Asia and Latin America. Biallelic GJB2 variants were identified in 58 probands at the time of enrollment these probands were excluded. In addition, upon review of phenotypic findings, 38/322 probands were excluded based on syndromic findings at the time of ascertainment and no further evaluation was performed on those samples. We performed ES as a primary diagnostic tool on one or two affected individuals from 212/226 families. Via ES we detected a total of 78 variants in 30 genes and showed their co-segregation with HL in 71 affected families. Most of the variants were frameshift or missense and affected individuals were either homozygous or compound heterozygous in their respective families. We employed GS as a primary test on a subset of 14 families and a secondary tool on 22 families which were unsolved by ES. Although the cumulative detection rate of causal variants by ES and GS is 40% (89/226), GS alone has led to a molecular diagnosis in 7 of 14 families as the primary tool and 5 of 22 families as the secondary test. GS successfully identified variants present in deep intronic or complex regions not detectable by ES.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Morton CC, Nance WE. Newborn hearing screening—a silent revolution. N Engl J Med. 2006;354:2151–64.
Yamasoba T, Lin FR, Someya S, Kashio A, Sakamoto T, Kondo K. Current concepts in age-related hearing loss: epidemiology and mechanistic pathways. Hear Res. 2013;303:30–8.
Gettelfinger JD, Dahl JP. Syndromic hearing loss: a brief review of common presentations and genetics. J Pediatr Genet. 2018;7:001–8.
Hilgert N, Smith RJ, Van Camp G. Forty-six genes causing nonsyndromic hearing impairment: which ones should be analyzed in DNA diagnostics? Mutat Res/Rev Mutat Res. 2009;681:189–96.
Kimberling WJ, Hildebrand MS, Shearer AE, Jensen ML, Halder JA, Trzupek K, et al. Frequency of Usher syndrome in two pediatric populations: Implications for genetic screening of deaf and hard of hearing children. Genet Med. 2010;12:512–6.
Brodie KD, Moore AT, Slavotinek AM, Meyer AK, Nadaraja GS, Conrad DE, et al. Genetic testing leading to early identification of childhood ocular manifestations of usher syndrome. Laryngoscope. 2021;131:E2053–E9.
Fromer M, Moran JL, Chambert K, Banks E, Bergen SE, Ruderfer DM, et al. Discovery and statistical genotyping of copy-number variation from whole-exome sequencing depth. Am J Hum Genet. 2012;91:597–607.
Krumm N, Sudmant PH, Ko A, O’Roak BJ, Malig M, Coe BP, et al. Copy number variation detection and genotyping from exome sequence data. Genome Res. 2012;22:1525–32.
Huang J, Liang X, Xuan Y, Geng C, Li Y, Lu H, et al. A reference human genome dataset of the BGISEQ-500 sequencer. Gigascience 2017;6:gix024.
Bademci G, Abad C, Incesulu A, Rad A, Alper O, Kolb SM, et al. MPZL2 is a novel gene associated with autosomal recessive nonsyndromic moderate hearing loss. Hum Genet. 2018;137:479–86.
Oza AM, DiStefano MT, Hemphill SE, Cushman BJ, Grant AR, Siegert RK, et al. Expert specification of the ACMG/AMP variant interpretation guidelines for genetic hearing loss. Hum Mutat. 2018;39:1593–613.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–23.
Tavtigian SV, Harrison SM, Boucher KM, Biesecker LG. Fitting a naturally scaled point system to the ACMG/AMP variant classification guidelines. Hum Mutat. 2020;41:1734–7.
Cartegni L, Wang J, Zhu Z, Zhang MQ, Krainer AR. ESEfinder: a web resource to identify exonic splicing enhancers. Nucleic Acids Res. 2003;31:3568–71.
Zhang Y. I-TASSER server for protein 3D structure prediction. BMC Bioinform. 2008;9:1–8.
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJ. The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc. 2015;10:845–58.
Naz S, Imtiaz A, Mujtaba G, Maqsood A, Bashir R, Bukhari I, et al. Genetic causes of moderate to severe hearing loss point to modifiers. Clin Genet. 2017;91:589–98.
Rehman AU, Santos-Cortez RLP, Drummond MC, Shahzad M, Lee K, Morell RJ, et al. Challenges and solutions for gene identification in the presence of familial locus heterogeneity. Eur J Hum Genet. 2015;23:1207–15.
Richard EM, Santos‐Cortez RLP, Faridi R, Rehman AU, Lee K, Shahzad M, et al. Global genetic insight contributed by consanguineous Pakistani families segregating hearing loss. Hum Mutat. 2019;40:53–72.
Bademci G, Foster J, Mahdieh N, Bonyadi M, Duman D, Cengiz FB, et al. Comprehensive analysis via exome sequencing uncovers genetic etiology in autosomal recessive nonsyndromic deafness in a large multiethnic cohort. Genet Med. 2016;18:364–71.
Denoyelle F, Weil D, Maw MA, Wilcox SA, Lench NJ, Allen-Powell DR, et al. Prelingual deafness: high prevalence of a 30delG mutation in the connexin 26 gene. Hum Mol Genet. 1997;6:2173–7.
de la Luz Arenas-Sordo M, Menendez I, Hernández-Zamora E, Sirmaci A, Gutiérrez-Tinajero D, McGetrick M, et al. Unique spectrum of GJB2 mutations in Mexico. Int J Pediatr Otorhinolaryngol. 2012;76:1678–80.
Batissoco AC, Abreu-Silva RS, Braga MCC, Lezirovitz K, Della-Rosa V, Alfredo T Jr, et al. Prevalence of GJB2 (connexin-26) and GJB6 (connexin-30) mutations in a cohort of 300 Brazilian hearing-impaired individuals: implications for diagnosis and genetic counseling. Ear Hear. 2009;30:1–7.
Bouzaher MH, Worden CP, Jeyakumar A. Systematic review of pathogenic GJB2 variants in the Latino population. Otol Neurotol. 2020;41:e182–e91.
Grangeiro CHP, Picanço-Albuquerque CG, dos Anjos TO, De Molfetta GA, Silva WA. Ferraz VEdF. Contribution of SLC26A4 to the molecular diagnosis of nonsyndromic prelingual sensorineural hearing loss in a Brazilian cohort. BMC Res Notes. 2018;11:1–7.
Manzoli GN, Bademci G, Acosta AX, Félix TM, Cengiz FB, Foster J, et al. Targeted resequencing of deafness genes reveals a founder MYO15A variant in northeastern Brazil. Ann Hum Genet. 2016;80:327–31.
Demain LA, Urquhart JE, O’Sullivan J, Williams SG, Bhaskar SS, Jenkinson EM, et al. Expanding the genotypic spectrum of Perrault syndrome. Clin Genet. 2017;91:302–12.
Baker S, Booth C, Fillman C, Shapiro M, Blair MP, Hyland JC, et al. A loss of function mutation in the COL9A2 gene causes autosomal recessive Stickler syndrome. Am J Med Genet Part A. 2011;155:1668–72.
Kjellström U, Martell S, Brobeck C, Andréasson S. Autosomal recessive Stickler syndrome associated with homozygous mutations in the COL9A2 gene. Ophthalmic Genet. 2021;42:161–9.
Nixon TR, Alexander P, Richards A, McNinch A, Bearcroft PW, Cobben J, et al. Homozygous type IX collagen variants (COL9A1, COL9A2, and COL9A3) causing recessive Stickler syndrome—expanding the phenotype. Am J Med Genet Part A. 2019;179:1498–506.
Hatch EP, Noyes CA, Wang X, Wright TJ, Mansour SL. Fgf3 is required for dorsal patterning and morphogenesis of the inner ear epithelium. 2007.
Ramsebner R, Ludwig M, Parzefall T, Lucas T, Baumgartner WD, Bodamer O, et al. A FGF3 mutation associated with differential inner ear malformation, microtia, and microdontia. Laryngoscope. 2010;120:359–64.
Riazuddin S, Ahmed ZM, Hegde RS, Khan SN, Nasir I, Shaukat U, et al. Variable expressivity of FGF3 mutations associated with deafness and LAMM syndrome. BMC Med Genet. 2011;12:1–11.
Tekin M, Hişmi BÖ, Fitoz S, Özdağ H, Cengiz FB, Sırmacı A, et al. Homozygous mutations in fibroblast growth factor 3 are associated with a new form of syndromic deafness characterized by inner ear agenesis, microtia, and microdontia. Am J Hum Genet. 2007;80:338–44.
Tekin M, Öztürkmen Akay H, Fitoz S, Birnbaum S, Cengiz F, Sennaroğlu L, et al. Homozygous FGF3 mutations result in congenital deafness with inner ear agenesis, microtia, and microdontia. Clin Genet. 2008;73:554–65.
Cabanillas R, Diñeiro M, Cifuentes GA, Castillo D, Pruneda PC, Álvarez R, et al. Comprehensive genomic diagnosis of non-syndromic and syndromic hereditary hearing loss in Spanish patients. BMC Med Genom. 2018;11:1–17.
Baux D, Vaché C, Blanchet C, Willems M, Baudoin C, Moclyn M, et al. Combined genetic approaches yield a 48% diagnostic rate in a large cohort of French hearing-impaired patients. Sci Rep. 2017;7:1–10.
Van Heurck R, Carminho-Rodrigues MT, Ranza E, Stafuzza C, Quteineh L, Gehrig C, et al. Benefits of exome sequencing in children with suspected isolated hearing loss. Genes 2021;12:1277.
Shearer AE, Smith RJ. Massively parallel sequencing for genetic diagnosis of hearing loss: the new standard of care. Otolaryngol–Head Neck Surg. 2015;153:175–82.
Sloan-Heggen CM, Bierer AO, Shearer AE, Kolbe DL, Nishimura CJ, Frees KL, et al. Comprehensive genetic testing in the clinical evaluation of 1119 patients with hearing loss. Hum Genet. 2016;135:441–50.
Najmabadi H, Kahrizi K. Genetics of non-syndromic hearing loss in the Middle East. Int J Pediatr Otorhinolaryngol. 2014;78:2026–36.
Murthy T, Paul KV, Minella AC, Pillai MM. The development and use of scalable systems for studying aberrant splicing in SF3B1-mutant CLL. Chronic Lymphocytic Leukemia: Springer; 2019. 83–99.
Wang Z, Rolish ME, Yeo G, Tung V, Mawson M, Burge CB. Systematic identification and analysis of exonic splicing silencers. Cell. 2004;119:831–45.
Acknowledgements
We are grateful to the participating families included in this study. We thank all physicians, genetic counselors, and audiologists for providing all the relevant information. We are grateful to the DNA-Neurogenetics Bank of the Instituto Nacional de Ciencias Neurológicas for supporting the collection of DNA samples and associated data used in this publication. The content of this publication does not reflect the opinion of the DNA-Neurogenetics Bank. This work was supported by the NIH R01DC009645 grant to MT.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramzan, M., Duman, D., Hendricks, L.C.P. et al. Genome sequencing identifies coding and non-coding variants for non-syndromic hearing loss. J Hum Genet 68, 657–669 (2023). https://doi.org/10.1038/s10038-023-01159-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-023-01159-9
This article is cited by
-
Genetic heterogeneity in hereditary hearing loss: Potential role of kinociliary protein TOGARAM2
European Journal of Human Genetics (2024)