Abstract
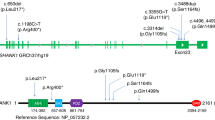
We report a 9-year-old Spanish boy with severe psychomotor developmental delay, short stature, microcephaly and abnormalities of the brain morphology, including cerebellar atrophy. Whole-exome sequencing (WES) uncovered two novel de novo variants, a hemizygous variant in CASK (Calcium/Calmodulin Dependent Serine Protein Kinase) and a heterozygous variant in EEF2 (Eukaryotic Translation Elongation Factor 2). CASK gene encodes the peripheral plasma membrane protein CASK that is a scaffold protein located at the synapses in the brain. The c.2506‐6 A > G CASK variant induced two alternative splicing events that account for the 80% of the total transcripts, which are likely to be degraded by NMD. Pathogenic variants in CASK have been associated with severe neurological disorders such as mental retardation with or without nystagmus also called FG syndrome 4 (FGS4), and intellectual developmental disorder with microcephaly and pontine and cerebellar hypoplasia (MICPCH). Heterozygous variants in EEF2, which encodes the elongation factor 2 (eEF2), have been associated to Spinocerebellar ataxia 26 (SCA26) and more recently to a childhood-onset neurodevelopmental disorder with benign external hydrocephalus. The yeast model system used to investigate the functional consequences of the c.34 A > G EEF2 variant supported its pathogenicity by demonstrating it affects translational fidelity. In conclusion, the phenotype associated with the CASK variant is more severe and masks the milder phenotype of EEF2 variant.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data of the study (variant and phenotype) have been submitted to LOVD database (patient ID 00411450).
Change history
26 May 2023
A Correction to this paper has been published: https://doi.org/10.1038/s10038-023-01164-y
References
Hata Y, Butz S, Sudhof TC. CASK: a novel dlg/PSD95 homolog with an N-terminal calmodulin-dependent protein kinase domain identified by interaction with neurexins. J Neurosci. 1996;16:2488–94.
Najm J, Horn D, Wimplinger I, Golden JA, Chizhikov VV, Sudi J, et al. Mutations of CASK cause an X-linked brain malformation phenotype with microcephaly and hypoplasia of the brainstem and cerebellum. Nat Genet. 2008;40:1065–7.
Piluso G, D’Amico F, Saccone V, Bismuto E, Rotundo IL, Di Domenico M, et al. A missense mutation in CASK causes FG syndrome in an Italian family. Am J Hum Genet. 2009;84:162–77.
Tarpey PS, Smith R, Pleasance E, Whibley A, Edkins S, Hardy C, et al. A systematic, large-scale resequencing screen of X-chromosome coding exons in mental retardation. Nat Genet. 2009;41:535–43.
Giacomini T, Nuovo S, Zanni G, Mancardi MM, Cusmai R, Pepi C, et al. CASK related disorder: Epilepsy and developmental outcome. Eur J Paediatr Neurol. 2021;31:61–9.
Zhang Y, Nie Y, Mu Y, Zheng J, Xu X, Zhang F, et al. A de novo variant in CASK gene causing intellectual disability and brain hypoplasia: a case report and literature review. Ital J Pediatr. 2022;48:73.
Kaneda Y, Yoshida MC, Kohno K, Uchida T, Okada Y. Chromosomal assignment of the gene for human elongation factor 2. Proc Natl Acad Sci USA. 1984;81:3158–62.
Kohno K, Uchida T, Ohkubo H, Nakanishi S, Nakanishi T, Fukui T, et al. Amino acid sequence of mammalian elongation factor 2 deduced from the cDNA sequence: homology with GTP-binding proteins. Proc Natl Acad Sci USA. 1986;83:4978–82.
Dever TE, Dinman JD, Green R. Translation elongation and recoding in eukaryotes. Cold Spring Harb Perspect Biol. 2018;10:a032649.
Hekman KE, Yu GY, Brown CD, Zhu H, Du X, Gervin K, et al. A conserved eEF2 coding variant in SCA26 leads to loss of translational fidelity and increased susceptibility to proteostatic insult. Hum Mol Genet. 2012;21:5472–83.
Nabais SaMJ, Olson AN, Yoon G, Nimmo GAM, Gomez CM, Willemsen MA, et al. De Novo variants in EEF2 cause a neurodevelopmental disorder with benign external hydrocephalus. Hum Mol Genet. 2021;29:3892–9.
Jorgensen R, Carr-Schmid A, Ortiz PA, Kinzy TG, Andersen GR. Purification and crystallization of the yeast elongation factor eEF2. Acta Crystallogr D Biol Crystallogr. 2002;58:712–5.
Harger JW, Dinman JD. An in vivo dual-luciferase assay system for studying translational recoding in the yeast Saccharomyces cerevisiae. RNA. 2003;9:1019–24.
Jacks T, Power MD, Masiarz FR, Luciw PA, Barr PJ, Varmus HE. Characterization of ribosomal frameshifting in HIV-1 gag-pol expression. Nature. 1988;331:280–3.
Belcourt MF, Farabaugh PJ. Ribosomal frameshifting in the yeast retrotransposon Ty: tRNAs induce slippage on a 7 nucleotide minimal site. Cell. 1990;62:339–52.
Dabrowski M, Bukowy-Bieryllo Z, Zietkiewicz E. Translational readthrough potential of natural termination codons in eucaryotes-The impact of RNA sequence. RNA Biol. 2015;12:950–8.
Pan YE, Tibbe D, Harms FL, Reissner C, Becker K, Dingmann B, et al. Missense mutations in CASK, coding for the calcium-/calmodulin-dependent serine protein kinase, interfere with neurexin binding and neurexin-induced oligomerization. J Neurochem. 2021;157:1331–50.
Biederer T, Sara Y, Mozhayeva M, Atasoy D, Liu X, Kavalali ET, et al. SynCAM, a synaptic adhesion molecule that drives synapse assembly. Science. 2002;297:1525–31.
Butz S, Okamoto M, Sudhof TC. A tripartite protein complex with the potential to couple synaptic vesicle exocytosis to cell adhesion in brain. Cell. 1998;94:773–82.
Hsueh YP, Wang TF, Yang FC, Sheng M. Nuclear translocation and transcription regulation by the membrane-associated guanylate kinase CASK/LIN-2. Nature. 2000;404:298–302.
Kuo TY, Hong CJ, Chien HL, Hsueh YP. X-linked mental retardation gene CASK interacts with Bcl11A/CTIP1 and regulates axon branching and outgrowth. J Neurosci Res. 2010;88:2364–73.
Jeyifous O, Waites CL, Specht CG, Fujisawa S, Schubert M, Lin EI, et al. SAP97 and CASK mediate sorting of NMDA receptors through a previously unknown secretory pathway. Nat Neurosci. 2009;12:1011–9.
Liu X, Sun P, Yuan Q, Xie J, Xiao T, Zhang K, et al. Specific deletion of CASK in pancreatic beta cells affects glucose homeostasis and improves insulin sensitivity in obese mice by reducing hyperinsulinemia running title: beta cell CASK deletion reduces hyperinsulinemia. Diabetes 2022;71:104–15.
Lejeune F. Nonsense-mediated mRNA decay, a finely regulated mechanism. Biomedicines. 2022;10:141.
Backwell L, Marsh JA. Diverse molecular mechanisms underlying pathogenic protein mutations: beyond the loss-of-function paradigm. Annu Rev Genomics Hum Genet. 2022;23:475–98.
Wang GS, Hong CJ, Yen TY, Huang HY, Ou Y, Huang TN, et al. Transcriptional modification by a CASK-interacting nucleosome assembly protein. Neuron. 2004;42:113–28.
Nix SL, Chishti AH, Anderson JM, Walther Z. hCASK and hDlg associate in epithelia, and their src homology 3 and guanylate kinase domains participate in both intramolecular and intermolecular interactions. J Biol Chem. 2000;275:41192–41200.
Knight JRP, Garland G, Poyry T, Mead E, Vlahov N, Sfakianos A, et al. Control of translation elongation in health and disease. Dis Model Mech. 2020;13:dmm043208.
Acknowledgements
We thank the patient and his family for their contribution.
Funding
This project was supported by the Spanish Instituto de Salud Carlos III (ISCIII) and European Regional Development Fund (ERDF) (grant PI17/00487 and PI20/00150 to FM-A). FJC-V and MER-G were supported by ISCIII-ERDF (PI17/00487 and PI20/00150).
Author information
Authors and Affiliations
Contributions
MER-G, FJC-V and MTS-C performed the experimental work on CASK; JDD, BBAdV and MJNS performed and evaluated functional assays on EEF2; AMA, RSdlH and PQ-F examined the patients and characterized the clinical features of the disease; FM-A supervised the study, interpreted data and wrote the original draft of the manuscript. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The Ethic Committee of the Instituto de Investigación Hospital 12 de Octubre (i + 12) approved the study, and written informed consent was obtained from the patient’s parents.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Dr. Alexandra N. Olson was missing.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rodríguez-García, M.E., Cotrina-Vinagre, F.J., Olson, A.N. et al. A novel de novo variant in CASK causes a severe neurodevelopmental disorder that masks the phenotype of a novel de novo variant in EEF2. J Hum Genet 68, 543–550 (2023). https://doi.org/10.1038/s10038-023-01150-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-023-01150-4