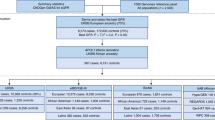
Abstract
Chronic kidney disease (CKD) is a syndrome characterized by a gradual loss of kidney function with decreased estimated glomerular filtration rate (eGFR), which may be accompanied by an increase in the urine albumin-to-creatinine ratio (UACR). Although trans-ethnic genome-wide association studies (GWASs) have been conducted for kidney-related traits, there have been few analyses in the Japanese population, especially for the UACR trait. In this study, we conducted a GWAS to identify loci related to multiple kidney-related traits in Japanese individuals. First, to detect loci associated with CKD, eGFR, and UACR, we performed separate GWASs with the following two datasets: 475 cases of CKD diagnosed at seven university hospitals and 3471 healthy subjects (dataset 1) and 3664 cases of CKD-suspected individuals with eGFR <60 ml/min/1.73 m2 or urinary protein ≥ 1+ and 5952 healthy subjects (dataset 2). Second, we performed a meta-analysis between these two datasets and detected the following associated loci: 10 loci for CKD, 9 loci for eGFR, and 22 loci for UACR. Among the loci detected, 22 have never been reported previously. Half of the significant loci for CKD were shared with those for eGFR, whereas most of the loci associated with UACR were different from those associated with CKD or eGFR. The GWAS of the Japanese population identified novel genetic components that were not previously detected. The results also suggest that the group primarily characterized by increased UACR possessed genetically different features from the group characterized by decreased eGFR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Levin A, Tonelli M, Bonventre J, Coresh J, Donner JA, Fogo AB, et al. Global kidney health 2017 and beyond: a roadmap for closing gaps in care, research, and policy. Lancet 2017;390:1888–917.
Matsushita K, Ballew SH, Astor BC, Jong PE, Gansevoort RT, Hemmelgarn BR, et al. Cohort profile: the chronic kidney disease prognosis consortium. Int J Epidemiol. 2013;42:1660–8.
Fukui A, Yokoo T, Nangaku M, Kashihara N. New measures against chronic kidney diseases in Japan since 2018. Clin Exp Nephrol. 2019;23:1263–71.
Lowrance WT, Ordoñez J, Udaltsova N, Russo P, Go AS. CKD and the risk of incident cancer. J Am Soc Nephrol. 2014;25:2327–34.
Dunnenberger HM, Khandekar JD. Value of personalized medicine. Jama 2016;315:612–3.
Joyner MJ, Paneth N. Promises, promises, and precision medicine. J Clin Invest. 2019;129:946–48.
Visscher PM, Brown MA, McCarthy MI, Yang J. Five years of GWAS discovery. Am J Hum Genet. 2012;90:7–24.
Visscher PM, Wray NR, Zhang Q, Sklar P, McCarthy MI, Brown MA, et al. 10 years of GWAS discovery: biology, function, and translation. Am J Hum Genet. 2017;101:5–22.
MacArthur J, Bowler E, Cerezo M, Gil L, Hall P, Hastings E, et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucleic Acids Res. 2017;45:D896–d901.
Zanetti D, Weale ME. Transethnic differences in GWAS signals: a simulation study. Ann Hum Genet. 2018;82:280–86.
Wuttke M, Li Y, Li M, Sieber KB, Feitosa MF, Gorski M, et al. A catalog of genetic loci associated with kidney function from analyses of a million individuals. Nat Genet. 2019;51:957–72.
Teumer A, Li Y, Ghasemi S, Prins BP, Wuttke M, Hermle T, et al. Genome-wide association meta-analyses and fine-mapping elucidate pathways influencing albuminuria. Nat Commun. 2019;10:4130.
Okada Y, Sim X, Go MJ, Wu JY, Gu D, Takeuchi F, et al. Meta-analysis identifies multiple loci associated with kidney function-related traits in east Asian populations. Nat Genet. 2012;44:904–9.
Yamada Y, Sakuma J, Takeuchi I, Yasukochi Y, Kato K, Oguri M, et al. Identification of C21orf59 and ATG2A as novel determinants of renal function-related traits in Japanese by exome-wide association studies. Oncotarget 2017;8:45259–73.
Hishida A, Nakatochi M, Akiyama M, Kamatani Y, Nishiyama T, Ito H, et al. Genome-wide association study of renal function traits: results from the Japan multi-institutional collaborative cohort study. Am J Nephrol. 2018;47:304–16.
Kuriyama S, Yaegashi N, Nagami F, Arai T, Kawaguchi Y, Osumi N, et al. The Tohoku medical megabank project: design and mission. J Epidemiol. 2016;26:493–511.
Fuse N, Sakurai-Yageta M, Katsuoka F, Danjoh I, Shimizu R, Tamiya G, et al. Establishment of integrated biobank for precision medicine and personalized healthcare: the Tohoku medical megabank project. Jma j. 2019;2:113–22.
Kashiwagi A, Kasuga M, Araki E, Oka Y, Hanafusa T, Ito H, et al. International clinical harmonization of glycated hemoglobin in Japan: From Japan Diabetes Society to National Glycohemoglobin Standardization Program values. J Diabetes Investig. 2012;3:39–40.
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39:S1–266.
Levin A, Stevens PE. Summary of KDIGO 2012 CKD Guideline: behind the scenes, need for guidance, and a framework for moving forward. Kidney Int. 2014;85:49–61.
Horio M, Imai E, Yasuda Y, Watanabe T, Matsuo S. Modification of the CKD epidemiology collaboration (CKD-EPI) equation for Japanese: accuracy and use for population estimates. Am J Kidney Dis. 2010;56:32–8.
Sumida K, Nadkarni GN, Grams ME, Sang Y, Ballew SH, Coresh J, et al. Conversion of urine protein-creatinine ratio or urine dipstick protein to urine albumin-creatinine ratio for use in chronic kidney disease screening and prognosis: an individual participant-based meta-analysis. Ann Intern Med. 2020;173:426–35.
Kawai Y, Mimori T, Kojima K, Nariai N, Danjoh I, Saito R, et al. Japonica array: improved genotype imputation by designing a population-specific SNP array with 1070 Japanese individuals. J Hum Genet. 2015;60:581–7.
Auton A, Brooks LD, Durbin RM, Garrison EP, Kang HM, Korbel JO, et al. A global reference for human genetic variation. Nature 2015;526:68–74.
Tadaka S, Katsuoka F, Ueki M, Kojima K, Makino S, Saito S, et al. 3.5KJPNv2: an allele frequency panel of 3552 Japanese individuals including the X chromosome. Hum Genome Var. 2019;6:28.
Howie BN, Donnelly P, Marchini J. A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet. 2009;5:e1000529.
Loh PR, Tucker G, Bulik-Sullivan BK, Vilhjálmsson BJ, Finucane HK, Salem RM, et al. Efficient Bayesian mixed-model analysis increases association power in large cohorts. Nat Genet. 2015;47:284–90.
Cook JP, Mahajan A, Morris AP. Guidance for the utility of linear models in meta-analysis of genetic association studies of binary phenotypes. Eur J Hum Genet. 2017;25:240–5.
Mbatchou J, Barnard L, Backman J, Marcketta A, Kosmicki JA, Ziyatdinov A, et al. Computationally efficient whole-genome regression for quantitative and binary traits. Nat Genet. 2021;53:1097–103.
Willer CJ, Li Y, Abecasis GR. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 2010;26:2190–1.
Devlin B, Roeder K. Genomic control for association studies. Biometrics 1999;55:997–1004.
de Bakker PI, Ferreira MA, Jia X, Neale BM, Raychaudhuri S, Voight BF. Practical aspects of imputation-driven meta-analysis of genome-wide association studies. Hum Mol Genet. 2008;17:R122–8.
Benner C, Havulinna AS, Järvelin MR, Salomaa V, Ripatti S, Pirinen M. Prospects of fine-mapping trait-associated genomic regions by using summary statistics from genome-wide association studies. Am J Hum Genet. 2017;101:539–51.
van Zuydam NR, Ahlqvist E, Sandholm N, Deshmukh H, Rayner NW, Abdalla M, et al. A genome-wide association study of diabetic kidney disease in subjects with type 2 diabetes. Diabetes 2018;67:1414–27.
Langefeld CD, Comeau ME, Ng MCY, Guan M, Dimitrov L, Mudgal P, et al. Genome-wide association studies suggest that APOL1-environment interactions more likely trigger kidney disease in African Americans with nondiabetic nephropathy than strong APOL1-second gene interactions. Kidney Int. 2018;94:599–607.
Kanai M, Akiyama M, Takahashi A, Matoba N, Momozawa Y, Ikeda M, et al. Genetic analysis of quantitative traits in the Japanese population links cell types to complex human diseases. Nat Genet. 2018;50:390–400.
Gorski M, Tin A, Garnaas M, McMahon GM, Chu AY, Tayo BO, et al. Genome-wide association study of kidney function decline in individuals of European descent. Kidney Int. 2015;87:1017–29.
Mahajan A, Rodan AR, Le TH, Gaulton KJ, Haessler J, Stilp AM, et al. Trans-ethnic Fine Mapping Highlights Kidney-Function Genes Linked to Salt Sensitivity. Am J Hum Genet. 2016;99:636–46.
Hellwege JN, Velez Edwards DR, Giri A, Qiu C, Park J, Torstenson ES, et al. Mapping eGFR loci to the renal transcriptome and phenome in the VA Million Veteran Program. Nat Commun. 2019;10:3842.
Graham SE, Nielsen JB, Zawistowski M, Zhou W, Fritsche LG, Gabrielsen ME, et al. Sex-specific and pleiotropic effects underlying kidney function identified from GWAS meta-analysis. Nat Commun. 2019;10:1847.
Yamada Y, Nishida T, Ichihara S, Kato K, Fujimaki T, Oguri M, et al. Identification of chromosome 3q28 and ALPK1 as susceptibility loci for chronic kidney disease in Japanese individuals by a genome-wide association study. J Med Genet. 2013;50:410–8.
Kanai M, Ulirsch JC, Karjalainen J, Kurki M, Karczewski KJ, Fauman E, et al. Insights from complex trait fine-mapping across diverse populations. medRxiv. 2021:2021.09.03.21262975.
Okuda H, Okamoto K, Abe M, Ishizawa K, Makino S, Tanabe O, et al. Genome-wide association study identifies new loci for albuminuria in the Japanese population. Clin Exp Nephrol. 2020;24:1–9.
Ioannidis JP, Patsopoulos NA, Evangelou E. Uncertainty in heterogeneity estimates in meta-analyses. Bmj 2007;335:914–6.
Kim J, Chen B, Bru JL, Huynh E, Momen M, Aswad DW. New findings on SNP variants of human protein L-isoaspartyl methyltransferase that affect catalytic activity, thermal stability, and aggregation. PLoS One. 2018;13:e0198266.
Boivin D, Bilodeau D, Béliveau R. Immunochemical characterization of L-isoaspartyl-protein carboxyl methyltransferase from mammalian tissues. Biochem J 1995;309:993–8.
Dong L, Li Y, Xue D, Liu Y. PCMT1 is an unfavorable predictor and functions as an oncogene in bladder cancer. IUBMB Life. 2018;70:291–99.
Saito H, Yamashita M, Ogasawara M, Yamada N, Niisato M, Tomoyasu M, et al. Chaperone protein L-isoaspartate (D-aspartyl) O-methyltransferase as a novel predictor of poor prognosis in lung adenocarcinoma. Hum Pathol. 2016;50:1–10.
Wägner AM, Cloos P, Bergholdt R, Boissy P, Andersen TL, Henriksen DB, et al. Post-translational protein modifications in type 1 diabetes: a role for the repair enzyme protein-L-isoaspartate (D-aspartate) O-methyltransferase? Diabetologia 2007;50:676–81.
Wägner AM, Cloos P, Bergholdt R, Eising S, Brorsson C, Stalhut M, et al. Posttranslational protein modifications in type 1 diabetes-genetic studies with PCMT1, the repair enzyme protein isoaspartate methyltransferase (PIMT) encoding gene. Rev Diabet Stud. 2008;5:225–31.
Lee J, Lee Y, Park B, Won S, Han JS, Heo NJ. Genome-wide association analysis identifies multiple loci associated with kidney disease-related traits in Korean populations. PLoS One. 2018;13:e0194044.
Lin D, Xiang T, Qiu Q, Leung J, Xu J, Zhou W, et al. Aldehyde dehydrogenase 2 regulates autophagy via the Akt-mTOR pathway to mitigate renal ischemia-reperfusion injury in hypothermic machine perfusion. Life Sci. 2020;253:117705.
Devuyst O, Pattaro C. The UMOD locus: insights into the pathogenesis and prognosis of kidney disease. J Am Soc Nephrol. 2018;29:713–26.
Köttgen A, Glazer NL, Dehghan A, Hwang SJ, Katz R, Li M, et al. Multiple loci associated with indices of renal function and chronic kidney disease. Nat Genet. 2009;41:712–7.
Gorski M, van der Most PJ, Teumer A, Chu AY, Li M, Mijatovic V, et al. 1000 Genomes-based meta-analysis identifies 10 novel loci for kidney function. Sci Rep. 2017;7:45040.
Morris AP, Le TH, Wu H, Akbarov A, van der Most PJ, Hemani G, et al. Trans-ethnic kidney function association study reveals putative causal genes and effects on kidney-specific disease aetiologies. Nat Commun. 2019;10:29.
Feingold E. Regression-based quantitative-trait-locus mapping in the 21st century. Am J Hum Genet. 2002;71:217–22.
Acknowledgements
This study was supported by AMED under Grant Number 21km0405210s. We would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Contributions
YS, YH, HN, SK, MY, and NK conceptualized the study. YS, YH, HN, AK, JW, MS, TW, HK, TN, HK, MY, SG, IN, and MN conducted SNP array study in each institution. AN and GT conducted GWAS. MY and NK supervised the study. YS and AN wrote the manuscript and the other authors revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
10038_2022_1094_MOESM9_ESM.pdf
Supplementary Table 4. Comparison of the loci significantly detected in the present study for the CKD trait with those detected in the past studies conducted in Japanese population
10038_2022_1094_MOESM10_ESM.pdf
Supplementary Table 5. Comparison of the loci significantly detected in the present study for the eGFR trait with those detected in the past studies conducted in Japanese or East Asian population
10038_2022_1094_MOESM11_ESM.pdf
Supplementary Table 6. Comparison of the loci significantly detected in the present study for the UACR trait with those included in GWAS Catalog
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sugawara, Y., Hirakawa, Y., Nagasu, H. et al. Genome-wide association study of the risk of chronic kidney disease and kidney-related traits in the Japanese population: J-Kidney-Biobank. J Hum Genet 68, 55–64 (2023). https://doi.org/10.1038/s10038-022-01094-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-022-01094-1