Abstract
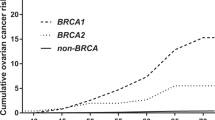
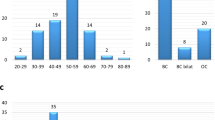
We previously reported that L63X and Q934X are BRCA1 common founder variants in Japan. So far, there have been no reports of a correlation between such BRCA common variants and the risk of BRCA-related cancers. In this analysis, we investigated the correlation between the risk of ovarian cancer (OC) and BRCA recurrent pathogenic variants. We examined the database of the Japanese organization of hereditary breast and ovarian cancer. The database contained 3517 probands who underwent BRCA genetic testing. Among them, 11.1% (392/3517) had germline BRCA1 pathogenic variant, and 8.3% (293/3517) had BRCA2 pathogenic variant. We calculated the OC prevalence, breast cancer (BC) prevalence, and the ratio of OC to BC within second-degree relatives. The ratio of OC to BC in Q934X family members was significantly higher than that in the overall BRCA1 family members (0.80 vs.0.52: p = 0.038), and the ratio in STOP799 was 0.42, which was relatively lower than the overall BRCA1 value. Both Q934X and STOP799 are located in the ovarian cancer cluster region (OCCR), however there seems to be a difference in the risk of OC. R2318X family members had a significant higher ratio of OC to BC at 0.32 than the overall BRCA2 value of 0.13 (p = 0.012). R2318X is known to be located in the OCCR. This is the first report to investigate the correlation between BRCA recurrent variants and the risk of OC in Japan. The family members of probands with Q934X or R2318X have a higher risk of OC than that with other BRCA variants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S, et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science. 1994;266:66–71.
Wooster R, Bignell G, Lancaster J, Swift S, Seal S, Mangion J, et al. Identification of the breast cancer susceptibility gene BRCA2. Nature. 1995;378:789–92.
Sekine M, Nishino K, Enomoto T. BRCA genetic test and risk-reducing salpingo-oophorectomy for hereditary breast and ovarian cancer: state-of-the-art. Cancers. 2021;13:2562.
Sekine M, Enomoto T. Precision medicine for hereditary tumors in gynecologic malignancies. J Obstet Gynaecol Res. 2021;47::2597–606.
Shanmughapriya S, Nachiappan V, Natarajaseenivasan K. BRCA1 and BRCA2 mutations in the ovarian cancer population across race and ethnicity: special reference to Asia. Oncology. 2013;84:226–32.
Satagopan JM, Boyd J, Kauff ND, Robson M, Scheuer L, Narod S, et al. Ovarian cancer risk in Ashkenazi Jewish carriers of BRCA1 and BRCA2 mutations. Clin Cancer Res. 2002;8:3776–81.
Menkiszak J, Chudecka-Głaz A, Gronwald J, Cymbaluk-Płoska A, Celewicz A, Świniarska M, et al. Prophylactic salpingo-oophorectomy in BRCA1 mutation carriers and postoperative incidence of peritoneal and breast cancers. J Ovarian Res. 2016;9:11.
Rebbeck TR, Mitra N, Wan F, Sinilnikova OM, Healey S, McGuffog L, et al. Association of type and location of BRCA1 and BRCA2 mutations with risk of breast and ovarian cancer. JAMA. 2015;313:1347–61.
Sugiyama T, Kamura T, Kigawa J, Terakawa N, Kikuchi Y, Kita T, et al. Clinical characteristics of clear cell carcinoma of the ovary: a distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer. 2000;88:2584–9.
Konstantinopoulos PA, Brady WE, Farley J, Armstrong A, Uyar DS, Gershenson DM. Phase II study of single-agent cabozantinib in patients with recurrent clear cell ovarian, primary peritoneal or fallopian tube cancer (NRG-GY001). Gynecol Oncol. 2018;150:9–13.
Sekine M, Nishino K, Enomoto T. Differences in ovarian and other cancers risks by population and BRCA mutation location. Genes. 2021;12:1050.
Anglian breast cancer study group. Prevalence and penetrance of BRCA1 and BRCA2 mutations in a population-based series of breast cancer cases. Br J Cancer. 2000;83:1301–8.
Weiderpass E, Sandin S, Inoue M, Shimazu T, Iwasaki M, Sasazuki S, et al. Risk factors for epithelial ovarian cancer in Japan - results from the Japan Public Health Center-based Prospective Study cohort. Int J Oncol. 2012;40:21–30.
National Cancer Center Cancer Control Information Center: Projected Cancer Statistics. https://ganjoho.jp/reg_stat/statistics/stat/summary.html.
Sekine M, Nagata H, Tsuji S, Hirai Y, Fujimoto S, Hatae M, et al. Mutational analysis of BRCA1 and BRCA2 and clinicopathologic analysis of ovarian cancer in 82 ovarian cancer families: two common founder mutations of BRCA1 in Japanese population. Clin Cancer Res. 2001;7:3144–50.
Sekine M, Nagata H, Tsuji S, Hirai Y, Fujimoto S, Hatae M, et al. Localization of a novel susceptibility gene for familial ovarian cancer to chromosome 3p22-p25. Hum Mol Genet. 2001;10:1421–9.
Nomura H, Sekine M, Yokoyama S, Arai M, Enomoto T, Takeshima N, et al. Clinical background and outcomes of risk-reducing salpingo-oophorectomy for hereditary breast and ovarian cancers in Japan. Int J Clin Oncol. 2019;24:1105–10.
Arai M, Yokoyama S, Watanabe C, Yoshida R, Kita M, Okawa M, et al. Genetic and clinical characteristics in Japanese hereditary breast and ovarian cancer: first report after establishment of HBOC registration system in Japan. J Hum Genet. 2018;63:447–57.
Spurdle AB, Lakhani SR, Healey S, Parry S, Da Silva LM, Brinkworth R, et al. Clinical classification of BRCA1 and BRCA2 DNA sequence variants: the value of cytokeratin profiles and evolutionary analysis–a report from the kConFab Investigators. J Clin Oncol. 2008;26:1657–63.
Williams RS, Chasman DI, Hau DD, Hui B, Lau AY, Glover JN. Detection of protein folding defects caused by BRCA1-BRCT truncation and missense mutations. J Biol Chem. 2003;278:53007–16.
Lee MS, Green R, Marsillac SM, Coquelle N, Williams RS, Yeung T, et al. Comprehensive analysis of missense variations in the BRCT domain of BRCA1 by structural and functional assays. Cancer Res. 2010;70:4880–90.
Kawaku S, Sato R, Song H, Bando Y, Arinami T, Noguchi E. Functional analysis of BRCA1 missense variants of uncertain significance in Japanese breast cancer families. J Hum Genet. 2013;58:618–21.
Bonatti F, Pepe C, Tancredi M, Lombardi G, Aretini P, Sensi E, et al. RNA-based analysis of BRCA1 and BRCA2 gene alterations. Cancer Genet Cytogenetics. 2006;170:93–101.
Hirotsu Y, Nakagomi H, Sakamoto I, Amemiya K, Mochizuki H, Omata M. Detection of BRCA1 and BRCA2 germline mutations in Japanese population using next-generation sequencing. Mol Genet Genom Med. 2015;3:121–9.
Tokunaga H, Iida K, Hozawa A, Ogishima S, Watanabe Y, Shigeta S, et al. Novel candidates of pathogenic variants of the BRCA1 and BRCA2 genes from a dataset of 3,552 Japanese whole genomes (3.5KJPNv2). PLoS One. 2021;16:e0236907.
Hasmad HN, Lai KN, Wen WX, Park DJ, Nguyen-Dumont T, Kang PCE, et al. Evaluation of germline BRCA1 and BRCA2 mutations in a multi-ethnic Asian cohort of ovarian cancer patients. Gynecol Oncol. 2016;141:318–22.
Palmero EI, Carraro DM, Alemar B, Moreira MAM, Ribeiro-Dos-Santos Â, Abe-Sandes K, et al. The germline mutational landscape of BRCA1 and BRCA2 in Brazil. Sci Rep. 2018;8:9188.
Meindl A. Comprehensive analysis of 989 patients with breast or ovarian cancer provides BRCA1 and BRCA2 mutation profiles and frequencies for the German population. Int J Cancer. 2002;97:472–80.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24.
Felix GES, Zheng Y, Olopade OI. Mutations in context: implications of BRCA testing in diverse populations. Fam Cancer. 2018;17:471–83.
Lubinski J, Phelan CM, Ghadirian P, Lynch HT, Garber J, Weber B, et al. Cancer variation associated with the position of the mutation in the BRCA2 gene. Fam Cancer. 2004;3:1–10.
Antoniou AC, Pharoah PD, Narod S, Risch HA, Eyfjord JE, Hopper JL, et al. Breast and ovarian cancer risks to carriers of the BRCA1 5382insC and 185delAG and BRCA2 6174delT mutations: a combined analysis of 22 population based studies. J Med Genet. 2005;42:602–3.
Yoshida R, Watanabe C, Yokoyama S, Inuzuka M, Yotsumoto J, Arai M, et al. Analysis of clinical characteristics of breast cancer patients with the Japanese founder mutation BRCA1 L63X. Oncotarget. 2019;10:3276–84.
Gayther SA, Warren W, Mazoyer S, Russell PA, Harrington PA, Chiano M, et al. Germline mutations of the BRCA1 gene in breast and ovarian cancer families provide evidence for a genotype-phenotype correlation. Nat Genet. 1995;11:428–33.
Thompson D, Easton D. Variation in BRCA1 cancer risks by mutation position. Cancer Epidemiol Biomark Prev. 2002;11:329–36.
Gayther SA, Mangion J, Russell P, Seal S, Barfoot R, Ponder BA, et al. Variation of risks of breast and ovarian cancer associated with different germline mutations of the BRCA2 gene. Nat Genet. 1997;15:103–5.
Momozawa Y, Iwasaki Y, Parsons MT, Kamatani Y, Takahashi A, Tamura C, et al. Germline pathogenic variants of 11 breast cancer genes in 7,051 Japanese patients and 11,241 controls. Nat Commun. 2018;9:4083.
Yoshihara K, Enomoto T, Aoki D, Watanabe Y, Kigawa J, Takeshima N, et al. Association of gBRCA1/2 mutation locations with ovarian cancer risk in Japanese patients from the CHARLOTTE study. Cancer Sci. 2020;111:3350–58.
Acknowledgements
JOHBOC Registration Committee [Committee]: Tadashi Nomizu (Hoshi General Hospital), Akihiro Sakurai (Sapporo Medical University), Megumi Okawa (St. Luke’s International Hospital), Junko Yotsumoto (International University of Health and Welfare), Hiraku Kumamaru (The University of Tokyo), [Secretariat of JOHBOC] Miyuki Shimoda. Participating Facilities of Nationwide registration projects: Aichi Cancer Center Hospital, Asahikawa Medical University, Cancer Institute Hospital, Chiba University, Ehime University, Fujita Medical University, Fukui University, Fukushima Medical University, Gifu University, Gunma Prefectural Cancer Center, Hiroshima Prefectural Hospital, Hiroshima University, Hokkaido Cancer Center, Hokkaido University, Hoshi General Hospital, Ibaraki Prefectural Hospital, Ishikawa Prefectural Central Hospital, Iwate Medical University, JA Hiroshima General Hospital, Japanese Red Cross Aichi Medical Center Nagoya Daini Hospital, Japanese Red Cross Ishinomaki Hospital, Japanese Red Cross Medical Center, Japanese Red Cross Nagoya Daiichi Hospital, Japanese Red Cross Saitama Hospital, Japanese Red Cross Yamaguchi Hospital, JCHO Saitama Medical Center, Jisenkai Aizawa Hospital, Juntendo University, Kanagawa Cancer Center, Kanazawa University, Kansai Medical University, Kansai Rosai Hospital, Kawasaki Medical School, Keio University, Kitakyushu Municipal Medical Center, Kitano Hospital, Kitazato University, Kochi Medical Center, Kochi University, Kokura Medical Center, Konan Kosei Hospital, Kyoto University, Nagasaki University, Nagoya City University, Nagoya University, Nara Medical University, National Hospital Organization Kyoto Medical Center, National Hospital Organization Osaka National Hospital, Nihonkai General Hospital, Niigata Cancer Center Hospital, Niigata University, Nippon Medical School, Nippon Medical School Musashi Kosugi Hospital, Okayama University, Osaka University, Sagara Hospital Breast Center, Saitama Cancer Center, Saitama Medical Center, Saitama Medical University International Medical Center, Saku Medical Center, Sapporo Medical University, Seirei Hamamatsu General Hospital, Shiga University of Medical Science, Shikoku Cancer Center, Shinshu University, Shiroyama Hospital, Shizuoka Cancer Center, Shizuoka General Hospital, Shonan Memorial Hospital, Showa Medical University Hospital, St. Lukes Hospital, St. Marianna University School of Medicine, Tawara IVF Clinic, The Jikei University, Tokuyama Central Hospital., Tokyo Medical Center, Tokyo Metropolitan Komagome Hospital, Tosei General Hospital, Tottori University, University of Tsukuba, Yamagata Prefectural Central Hospital, Yamaguchi University, Yamanashi Prefectural Central Hospital, and Yokohama City University.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
M.S. received lecture fees from AstraZeneca. T.E. received lecture fees from AstraZeneca and Chugai Pharmaceutical. S.N. received lecture fees from AstraZeneca and KONICA MINOLTA. Other authors declare no competing interests.
Ethics approval
The ethics review board of the Japanese HBOC Consortium approved the establishment of the database to investigate Japanese HBOC patients’ characteristics. This registration has been begun after obtaining further approval from the institutional review board of each medical institution where BRCA genetic testing and genetic counseling were available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Sekine, M., Enomoto, T., Arai, M. et al. Correlation between the risk of ovarian cancer and BRCA recurrent pathogenic variants in Japan. J Hum Genet 67, 267–272 (2022). https://doi.org/10.1038/s10038-021-01002-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-021-01002-z
This article is cited by
-
The frequency and pathogenicity of BRCA1 and BRCA2 variants in the general Japanese population
Journal of Human Genetics (2024)
-
Genetic medicine in companion diagnostics of germline BRCA testing of Japanese pancreatic cancer patients
Journal of Human Genetics (2023)