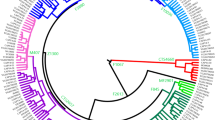
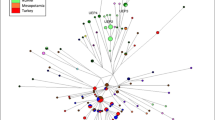
Abstract
Western Kazakhstan is populated by three clans totaling 2 million people. Since the clans are patrilineal, the Y-chromosome is the most informative genetic system for tracing their origin. We genotyped 40 Y-SNP and 17 Y-STR markers in 330 Western Kazakhs. High phylogenetic resolution within haplogroup C2a1a2-M48 was achieved by using additional SNPs. Three lines of evidence indicate that the Alimuly and Baiuly clans (but not the Zhetiru clan) have a common founder placed 700 ± 200 years back by the STR data and 500 ± 200 years back by the sequencing data. This supports traditional genealogy claims about the descent of these clans from Emir Alau, who lived 650 years ago and whose lineage might be carried by two-thirds of Western Kazakhs. There is accumulation of specific haplogroups in the subclans representing other lineages, confirming that the clan structure corresponds with the paternal genetic structure of the steppe population.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zerjal T, Xue YL, Bertorelle G, Wells RS, Bao WD, Zhu SL, et al. The genetic legacy of the Mongols. Am J Hum Genet. 2003;72:717–21.
Xue Y, Zerjal T, Bao W, Zhu S, Lim SK, Shu Q, et al. Recent spread of a Y-chromosomal lineage in northern China and Mongolia. Am J Hum Genet. 2005;77:1112–6.
Balaresque P, Poulet N, Cussat-Blanc S, Gerard P, Quintana-Murci L, Heyer E, et al. Y-chromosome descent clusters and male differential reproductive success: young lineage expansions dominate Asian pastoral nomadic populations. Eur J Hum Genet. 2015;23:1413–22.
Balanovsky O, Zhabagin M, Agdzhoyan A, Chukhryaeva M, Zaporozhchenko V, Utevska O, et al. Deep phylogenetic analysis of haplogroup G1 provides estimates of SNP and STR mutation rates on the human Y-chromosome and reveals migrations of Iranic speakers. PLoS One. 2015;10:e0122968.
Zhabagin M, Sabitov Zh, Tarlykov P, Tazhigulova I, Junnisova Z, Yerezhepov D, et al. The medieval Mongolian roots of Y-chromosomal lineages from South Kazakhstan. BMC Genetics. 2020;21(Suppl 1):87.
Wen SQ, Yao HB, Du PX, Wei LH, Tong XZ, Wang LX, et al. Molecular genealogy of Tusi Lu’s family reveals their paternal relationship with Jochi, Genghis Khan’s eldest son. J Hum Genet. 2019;64:815–20.
Larmuseau MHD, van den Berg P, Claerhout S, Calafell F, Boattini A, Gruyters L, et al. A historical-genetic reconstruction of human extra-pair paternity. Curr Biol. 2019;29:4102–7.e7.
Zhabagin M, Balanovsky О, Sabitov ZH, Temirgaliyev A, Agdzhoyan A, Koshel S, et al. Reconstructing the genetic structure of the Kazakh from clan distribution data. Vavilovskii Zh Genetiki i Selektsii. 2018;22:895–904.
Massanov N. Nomadic Civilization of Kazakhs: the basics migratory habits of life of society. Almaty: Nurbolat Masanov Fund; 2011.
Rakishev R. Placement of the main clans of the Kazakhs by region and their approximate number. Rep. Acad Sci Repub Kazakhstan. 2015;3:193–8.
Usenbaev T. Alshin Shezhiresi (Genealogy of Alshyn). Kyzylorda: Tumar Publ.; 2003.
Sabitov ZH. Ethnogenesis of Kazakhs in terms of population genetics. Russian J Genet Geneal. 2013;5:29–47.
Rashid al-Din. Collection of Histories. Volume 1. Book 1. Moscow-Leningrad: Publishing House of the Academy of Sciences of the USSR; 1952.
Tyurin A. Ethnic roots of the Kazakhs of the Alimuly tribal alliance according to population genetics data. Astrakhan Petrovsky Readings. 2020;4:59–64.
Sultanov T. Nomadic tribes of the Aral Sea region in the XV – XVII centuries. Moscow: Nauka; 1982.
Zhabagin M, Balanovska E, Sabitov Z, Kuznetsova M, Agdzhoyan A, Balaganskaya O, et al. The Connection of the Genetic, Cultural and Geographic Landscapes of Transoxiana. Sci Rep. 2017;7:3085.
Zhabagin M, Sarkytbayeva A, Tazhigulova I, Yerezhepov D, Li S, Akilzhanov R, et al. Development of the Kazakhstan Y-chromosome haplotype reference database: analysis of 27 Y-STR in Kazakh population. Int J Leg Med. 2019;133:1029–32.
Baimukhanov N, Baimbetov G. Genetic subclade ZQ5 of haplogroup C-Y15552 tribal union “Alimuly”. J Hist. 2018;2:172–80.
Baimbetov G, Baimukhanov N. Haplogroup C-L1370 of the Y-chromosome DNA or “Steppe branch” of haplogroup C-M86, its sub-branches in phylogenetic tree and variety of haplotypes in Kazakh and other ethnic populations. J Hist. 2018;4:50–58.
Lell JT, Sukernik RI, Starikovskaya YB, Su B, Jin L, Schurr TG, et al. The dual origin and Siberian affinities of Native American Y chromosomes. Am J Hum Genet. 2002;70:192–206.
Zerjal T, Wells RS, Yuldasheva N, Ruzibakiev R, Tyler-Smith C. A genetic landscape reshaped by recent events: Y-chromosomal insights into Central Asia. Am J Hum Genet. 2002;71:466–82.
Pakendorf B, Novgorodov IN, Osakovskij VL, Danilova AP, Protod’jakonov AP, Stoneking M. Investigating the effects of prehistoric migrations in Siberia: genetic variation and the origins of Yakuts. Hum Genet. 2006;120:334–53.
Pakendorf B, Novgorodov IN, Osakovskij VL, Stoneking M. Mating patterns amongst Siberian reindeer herders: inferences from mtDNA and Y-chromosomal analyses. Am J Phys Anthropol. 2007;133:1013–27.
Malyarchuk B, Derenko M, Denisova G, Wozniak M, Grzybowski T, Dambueva I, et al. Phylogeography of the Y-chromosome haplogroup C in northern Eurasia. Ann Hum Genet. 2010;74:539–46.
Balanovska E, Bogunov Y, Kamenshikova E, Balaganskaya O, Agdzhoyan A, Bogunova A, et al. Demographic and genetic portraits of the Ulchi population. Russian J Genet. 2018;54:1245.
Katoh T, Munkhbat B, Tounai K, Mano S, Ando H, Oyungerel G, et al. Genetic features of Mongolian ethnic groups revealed by Y-chromosomal analysis. Gene. 2005;346:63–70.
Dulik MC, Zhadanov SI, Osipova LP, Askapuli A, Gau L, Gokcumen O, et al. Mitochondrial DNA and Y chromosome variation provides evidence for a recent common ancestry between Native Americans and Indigenous Altaians. Am J Hum Genet. 2012;90:229–46.
Karmin M, Saag L, Vicente M, Wilson Sayres MA, Järve M, Talas UG, et al. A recent bottleneck of Y chromosome diversity coincides with a global change in culture. Genome Res. 2015;25:459–66.
Karafet TM, Osipova LP, Gubina MA, Posukh OL, Zegura SL, Hammer MF. High levels of Y-chromosome differentiation among native Siberian populations and the genetic signature of a boreal hunter-gatherer way of life. Hum Biol. 2002;74:761–89.
Liu BL, Ma PC, Wang CZ, Shi Y, Yao HB, et al. Paternal origin of Tungusic-speaking populations: Insights from the updated phylogenetic tree of Y-chromosome haplogroup C2a-M86. Am J Hum Biol. 2020;e23462
Balanovska E, Zhabagin M, Agdzhoyan A, Chukhryaeva M, Markina N, Balaganskaya O, et al. Population biobanks: organizational models and prospects of application in gene geography and personalized medicine. Russian J Genet. 2016;52:1227–43.
Balanovsky O, Rootsi S, Pshenichnov A, Kivisild T, Churnosov M, Evseeva I, et al. Two sources of the Russian patrilineal heritage in their Eurasian context. Am J Hum Genet. 2008;82:236–50.
Nei M. Molecular evolutionary genetics. New York: Columbia University Press; 1987.
Bandelt H-J, Forster P, RohL A. Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol. 1999;16:37–48.
Bandelt HJ, Forster P, Sykes BC, Richards MB. Mitochondrial portraits of human populations using median networks. Genetics 1995;141:743–53.
Muzzio M, Muzzio JC, Bravi CM, Bailliet G. Technical note: a method for assignment of the weight of characters. Am J Phys Anthropol. 2010;143:488–92.
Willuweit S, Roewer L. The new Y chromosome haplotype reference database. Forensic Sci Int Genet. 2015;15:43–8.
Roewer L, Krüger C, Willuweit S, Nagy M, Rodig H, Kokshunova L, et al. Y-chromosomal STR haplotypes in Kalmyk population samples. Forensic Sci Int. 2007;173:204–9.
Dulik MC, Osipova LP, Schurr TG. Y-chromosome variation in Altaian Kazakhs reveals a common paternal gene pool for Kazakhs and the influence of Mongolian expansions. PLoS One. 2011;6:e17548.
Di Cristofaro J, Pennarun E, Mazieres S, Myres NM, Lin AA, Temori SA, et al. Afghan Hindu Kush: where Eurasian sub-continent gene flows converge. PLoS One. 2013;8:e76748.
Balinova N, Post H, Kushniarevich A, Flores R, Karmin M, Sahakyan H, et al. Y-chromosomal analysis of clan structure of Kalmyks, the only European Mongol people, and their relationship to Oirat-Mongols of Inner Asia. Eur J Hum Genet. 2019;27:1466–74.
Forster P, Harding R, Torroni A, Bandelt HJ. Origin and evolution of native American mtDNA variation: a reappraisal. Am J Hum Genet. 1996;59:935–45.
Saillard J, Forster P, Lynnerup N, Bandelt HJ, Norby S. mtDNA variation among Greenland Eskimos: the edge of the Beringian expansion. Am J Hum Genet. 2000;67:718–26.
Macaulay V, Soares P, Richards MB. Rectifying long-standing misconceptions about the ρ statistic for molecular dating. PLoS One. 2019;14:e0212311.
Ge J, Budowle B, Aranda XG, Planz JV, Eisenberg AJ, Chakraborty R. Mutation rates at Y chromosome short tandem repeats in Texas populations. Forensic Sci Int Genet. 2009;3:179–84.
Fenner JN. Cross-cultural estimation of the human generation interval for use in genetics-based population divergence studies. Am J Phys Anthropol. 2005;128:415–23.
Mustakimov I. On a list of Daftar-i Genghis-name. Medieval Turk-Tatar S. 2009;1:122–31.
Sikaliev A. Nogai heroic epic. Cherkessk: KChIGI Publ.; 1994.
Sengirbekulı M. Forty heroes of Crimea. In: Kosan S, editor. Ancestral Heritage (Babalar sozi): 51 volume. Astana: Foliant Publ.; 2008.
Guillot EG, Cox MP. High frequency haplotypes are expected events, not historical figures. F1000Res. 2015;4:666.
Sobakin A. Atameken Atlas. Almaty: Brif Research Group; 1999.
Acknowledgements
We are grateful for the trust of the sample donors. This work was funded by the Ministry of Education and Science of Republic Kazakhstan (Grants No. AP05134955). EB was funded by the State assignments of Russian Ministry of Science and Higher Education for the Research Centre for Medical Genetics. OB was funded by the Russian Scientific Fund project 17-14-01345 (statistical analysis of the Central Asian influence). MZ and HL are supported by Scientific and Technology Committee of Shanghai Municipality (19410740700). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: MZ, EB; performed the experiments: MZ, IT, IA; analyzed the data: MZ, OB, VU; contributed reagents/materials/analysis tools: ZS, IT, AAg, LW, VU, SK, KM, AAk, HL, EB; MZ drafted the manuscript; MZ, OB contributed in writing the paper; study initiation: MZ, AAk; read and approved the final version of the paper: all co-authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
Ethical approval and consent to participate were received from the Ethics Committee of the Research Centre for Medical Genetics (Moscow, Russia), National Center for Biotechnology and the National Laboratory Astana (Nur-Sultan, Kazakhstan).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhabagin, M., Sabitov, Z., Tazhigulova, I. et al. Medieval Super-Grandfather founder of Western Kazakh Clans from Haplogroup C2a1a2-M48. J Hum Genet 66, 707–716 (2021). https://doi.org/10.1038/s10038-021-00901-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-021-00901-5