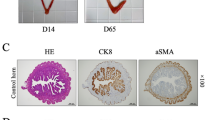
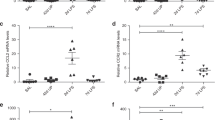
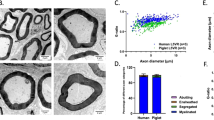
Abstract
ABSTRACT: Replication-deficient adenoviruses have been used to transfer various genes of interest to mammalian tissues in vivo. Effective gene therapy for inborn genetic defects presenting with significant morbidity and mortality at birth will require correction of the defect prenatally. To test the hypothesis that intraamniotically administered adenovirus transfers gene expression to fetal tissues, replication-deficient human type 5 adenovirus carrying the lacZ gene which encodes nuclear-targeted bacterial β-galactosidase (Av1LacZ4) was instilled into the amniotic cavity of fetal sheep (1010 to 1.5 X 1011 pfu) and fetal mice (109 pfu) at 0.8 term gestation. Amniotic membranes and gastrointestinal and respiratory tract tissues were harvested after 3 d, bacterial β-ga-lactosidase activity was determined by 5-bromo-4-chloro-3-indoyl-β-D-galactopyranoside (X-gal) enzyme-histochemistry, and tissue integrity was assessed in sections stained with hematoxylin and eosin. Bacterial β-galactosidase activity was abundant in amniotic membranes and present in lower levels in esophagus, stomach, and small intestine as well as in conducting airways and pulmonary alveoli. To determine whether gene transfer by intraamniotic injection of adenovirus was dose-dependent, Av1Lucl, an adenoviral vector carrying the gene for luciferase (105-109 pfu), was injected intraamniotically into fetal mice at 0.8 term gestation. Luciferase activity measured after 3 d in tissue homogenates of Av1Luc1 -treated fetal mice revealed a linear dose response in amniotic membranes and gastrointestinal and respiratory tract organs. Intraamniotic administration of an adenoviral gene vector leads to expression of the transferred gene in amniotic membranes as well as in fetal gastrointestinal and respiratory tract tissues in a dose-dependent manner.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Holzinger, A., Trapnell, B., Weaver, T. et al. Intraamniotic Administration of an Adenoviral Vector for Gene Transfer to Fetal Sheep and Mouse Tissues. Pediatr Res 38, 844–850 (1995). https://doi.org/10.1203/00006450-199512000-00004
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199512000-00004
This article is cited by
-
Mechanistic Insights into Factor VIII Immune Tolerance Induction via Prenatal Cell Therapy in Hemophilia A
Current Stem Cell Reports (2019)
-
In utero stem cell transplantation and gene therapy: rationale, history, and recent advances toward clinical application
Molecular Therapy - Methods & Clinical Development (2016)
-
Intra-Amniotic rAAV-Mediated Microdystrophin Gene Transfer Improves Canine X-Linked Muscular Dystrophy and May Induce Immune Tolerance
Molecular Therapy (2015)
-
Rapid functional dissection of genetic networks via tissue-specific transduction and RNAi in mouse embryos
Nature Medicine (2010)
-
The developmental stage determines the distribution and duration of gene expression after early intra-amniotic gene transfer using lentiviral vectors
Gene Therapy (2010)