Abstract
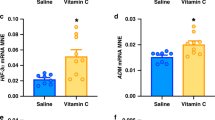
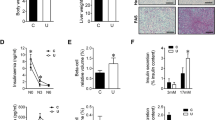
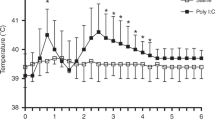
Summary: The control of glycogen metabolism was studied in the lung of the developing rat or rabbit fetus by means of hormonal suppression (in utero decapitation, or thyroidectomy, or drug administration) and hormone administration to intact or hormone-deprived fetuses. In the rat fetus, in utero decapitation plus maternal adrenalectomy did not impair lung glycogen accumulation between days 17.5 and 19.5. On the contrary, glycogen breakdown, which occurs after day 20.5 in the rat and day 27 in the rabbit, was impaired, at least partly, by decapitation of the 17.5-day-old rat fetus and of the 23-day-old rabbit fetus. On the average, on day 21.5 the glycogen content of the lungs of the decapitated rat fetus was 323 ± 33 mg/g proteins versus 195 ± 14 mg/g proteins in the lungs of their controls. Thyroid hormone deprivation did not reproduce the effect of decapitation. Phosphorylase activity, which increases near term, still increased after decapitation; however, on the average, it was reduced by approximately 25%. Cortisol injection to decapitated fetuses restored normal glycogen breakdown without restoring phosphorylase activity. Growth hormone administered to decapitated fetuses restored a normal phosphorylase activity, but the lung glycogen content remained higher than in controls.
These results led us to search for a second mechanism of glycogen degradation. Lysosomal amyloglucosidase, which increased by 40% through days 17.5 and 21.5 in the rat, was unaffected by either hormone deprivation or by cortisol administration. Autophagic activity, estimated by β-N-acetyl-glucosaminidase (NAGase) that was liberated by homogeneisation and osmotic shock, was reduced by hormone deprivation (in fetal rat lung, free NAGase after homogeneisation: 27.2 ± 0.6% of total activity in decapitates versus 35.1 ± 1.3% in controls; free NAGase after osmotic shock: 54.9 ± 1.1% of total activity in decapitates versus 60.2 ± 1.5% in controls). Cortisol administration restored the autophagic activity of the tissue. In intact rat fetuses, growth hormone induced an anticipated rise of phosphorylase activity (16.8 ± 1.2 U/g proteins in 19.5-day-old treated rat fetuses versus 9.7 ± 0.6 U/g proteins in controls). Cortisol increased autophagic activity. It is concluded that: (1) glycogen accumulation in the fetal lung is probably not dependent upon fetal hormones, (2) fetal (and possibly maternal) hormones influence glycogen breakdown, (3) the rise of phosphorylase activity observed at the end of pregnancy partially occurs in pituitary deprived fetuses but it is stimulated by growth hormone which may be necessary for a complete development of the enzyme activity and (4) the effects of corticosteroids on glycogenolysis might be exerted through the control of autophagy.
Speculation: The present observations suggest that phosphorylase is insufficient by itself to fulfill glycogen breakdown in fetal lung and that hydrolysis in lysosomal structures is necessary for glycogen utilization and lung maturation. Taking into account the lysosomerelated nature of lamellar bodies, the presence of hydrolases in these structures and their spatial association with glycogen areas, the assumption of a link between glycogen utilization and phospholipid synthesis in the developing lung is reinforced.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bourbon, J., Jost, A. Control of Glycogen Metabolism in the Developing Fetal Lung. Pediatr Res 16 (Suppl 1), 50–56 (1982). https://doi.org/10.1203/00006450-198201001-00010
Issue Date:
DOI: https://doi.org/10.1203/00006450-198201001-00010
This article is cited by
-
Optimization of fetal lung organ culture for surfractant biosynthesis
In Vitro Cellular & Developmental Biology (1987)
-
Pre- and postnatal carbohydrate metabolism of rat lung tissue
Archives of Toxicology (1986)