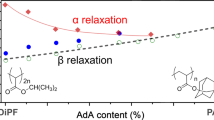
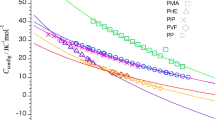
Abstract
The enthalpy relaxation of poly(cyanobipheny hexylacrylate) has been studied by differential scanning calorimetry. The experimental results of the heat capacity (cp) were compared with the prediction of a phenomenological model for the process of structural relaxation based on the configurational entropy (Sc) and the relaxation time expressed by the Adam-Gibbs equation. This model follows the evolution of the configurational entropy of the sample during the whole thermal history including an isothermal annealing. The parameters of the model were calculated by simultaneous fits to the experimental results. With the calculation result, the peak position appeared in DSC curve of cp was interpreted using the plot of temperature dependence of Sc. The plot showed that Sc rapidly approached to the configurational entropy of equilibrium (Sceq) in the narrow temperature range around Tg, which is often referred to as the recovery of enthalpy, and thus the recovery of Sc, in interpreting the enthalpy relaxation. As the measure of structural relaxation upon the isothermal ageing, the value of relaxation function (φ) was considered, which estimates the departure from the equilibrium. The correlation between φ of Sc and φ of enthalpy is also presented.
Similar content being viewed by others
Article PDF
References
Y. Tanaka, S. Takahashi, R. Kishi, and H. Ichijo, Mol. Cryst. Liq. Cryst., 339, 45 (2000).
Y. Tanaka, R. Kishi, and H. Ichijo, Mol. Cryst. Liq. Cryst., 348, 179 (2000).
Y. Tanaka, Polym. J., 39, 1030 (2007).
A. Q. Tool, J. Am. Ceram. Soc., 29, 240 (1946).
G. W. Scherer, J. Am. Ceram. Soc., 67, 504 (1984).
I. M. Hodge and A. R. Berens, Macromolecules, 14, 1598 (1981).
I. M. Hodge, Macromolecules, 20, 2897 (1987).
J. L. Gomez Ribelles and M. Monleon Pradas, Macromolecules, 28, 5867 (1995).
J. L. Gomez Ribelles, M. Monleon Pradas, A. Vidaurre Garayo, F. Romero Colomer, J. Mas Estelles, and J. M. Meseguer Duenas, Macromolecules, 28, 5878 (1995).
J. L. Gomez Ribelles, M. Monleon Pradas, A. Vidaurre Garayo, F. Romero Colomer, J. Mas Estelles, and J. M. Meseguer Duenas, Polymer, 38, 963 (1997).
J. M. Meseguer Duenas, A. Vidaurre Garayo, F. Romero Colomer, J. Mas Estelles, J. L. Gomez Ribelles, and M. Monleon Pradas, J. Polym. Sci, B: Polym. Phys., 35, 2201 (1997).
J. L. Gomez Ribelles, J. M. Meseguer Duenas, C. Torregrosa Cabanilles, and M. Monleon Pradas, J. Phys.: Condens. Matter. 15, S1149 (2003).
N. M. Alves, J. L. Gomez Ribelles, and J. F. Mano, Polymer. 46, 491 (2005).
L. Andreozzi, M. Faetti, F. Zulli, and M. Giordano, Eur. Phys. J. B, 41, 383 (2004).
L. Andreozzi, M. Faetti, F. Zulli, and M. Giordano, Macromolecules, 37, 8010 (2004).
L. Andreozzi, M. Faetti, M. Giordano, D. Palazzuoli, and F. Zulli, Macromolecules, 36, 7379 (2003).
C. T. Moynihan, A. J. Easteal, D. C. Tran, J. A. Wilder, and E. P. Donovan, J. Am. Ceram. Soc., 59, 137 (1976).
I. M. Hodge, Macromolecules, 16, 898 (1983).
T. Nagaya, H. Hotta, H. Orihara, and Y. Ishibashi, J. Phys. Soc. Jpn., 61, 3511 (1992).
C. T. Moynihan, A. J. Easteal, M. A. DeBolt, and J. Tucker, J. Am. Ceram. Soc., 59, 12 (1976).
M. A. DeBolt, A. J. Easteal, P. B. Macedo, and C. T. Moynihan, J. Am. Ceram. Soc., 59, 16 (1976).
G. Williams and D. C. Watts, Trans. Faraday Soc., 66, 80 (1970).
G. Adam and J. H. Gibbs, J. Chem. Phys., 43, 139 (1965).
O. S. Narayanaswamy, J. Am. Ceram. Soc., 54, 491 (1971).
J. H. Gibbs and E. A. DiMarzio, J. Chem. Phys., 28, 373 (1958).
T. I. Gubina, S. Kise, S. G. Kostromin, R. V. Talrose, V. P. Shivaev, and N. A. Platé, Liq. Cryst., 4, 197 (1989).
Concerning DSC apparatus, the incorrect model number was described in the previous paper of ref 3. The correct number is SEIKO DSC 200.
J. L. Gomez Ribelles, A. Ribes Greus, and R. Diaz Calleja, Polymer, 31, 223 (1990).
J. J. Tribone, J. M O'Reilly, and J. Greener, Macromolecules, 19, 1732 (1986).
I. M. Hodge and A. R. Berens, Macromolecules, 15, 762 (1982).
I. M. Hodge and G. S. Huvard, Macromolecules, 16, 371 (1983).
F. Romero Colomer and J. L. Gomez Ribelles, Polymer, 30, 849 (1989).
C. Alvarez, N. T. Correia, J. J. Moura Ramos, and A. C. Fernandes, Polymer, 41, 2907 (2000).
L. Andreozzi, M. Faetti, M. Giordano, and D. Palazzuoli, Macromolecules, 35, 9049 (2002).
M. J. Richardson and N. G. Savill, Polymer, 16, 753 (1975).
A. S. Marshall and S. E. B. Petrie, J. Appl. Phys., 46, 4223 (1975).
A. R. Berens and I. M. Hodge, Macromolecules, 15, 756 (1982).
J. M. G. Cowie and R. Ferguson, Macromolecules, 22, 2307 (1989).
D. Lacey, G. Nestor, and M. J. Richardson, Thermochim. Acta, 238, 99 (1994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tanaka, Y. Enthalpy Relaxation of Liquid Crystalline Polymer with Cyanobiphenyl Group in the Side Chain: Analysis based on Configurational Entropy Model with the AG Expression. Polym J 41, 125–131 (2009). https://doi.org/10.1295/polymj.PJ2008225
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2008225