Abstract
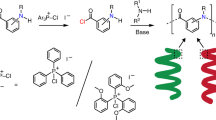
Palladium-mediated polymerization of diazoacetamides bearing various substituents 1–4 is described. Homopolymerization of monomers with n-hexyl or 4-n-octylphenyl group on the nitrogen afforded polymers [poly(N-substituted carbamoylmethylene)], whose all the main chain carbons have N-substituted carbamoly groups. Elemental analysis of the products indicated incorporation of azo group in the main chain. Copolymerization of diazoacetamides 1–4 with a diazoketone, (E)-1-diazo-4-phenyl-3-buten-2-one 5, proceeded efficiently, giving poly(substituted methylene)s with a variety of N-substituted carbamoyl groups in their side chains.
Similar content being viewed by others
Article PDF
References
J. Bai, L. D. Burke, and K. D. Shea, J. Am. Chem. Soc., 129, 4981 (2007).
E. Ihara, M. Kida, T. Itoh, and K. Inoue, J. Polym. Sci., Part A: Polym. Chem., 45, 5209 (2007).
L. Liu, Y. Song, and H. Li, Polym. Int., 51, 1047 (2002).
E. Ihara, N. Haida, M. Iio, and K. Inoue, Macromolecules, 36, 36 (2003).
E. Ihara, M. Fujioka, N. Haida, T. Itoh, and K. Inoue, Macromolecules, 38, 2101 (2005).
E. Ihara, A. Nakada, T. Itoh, and K. Inoue, Macromolecules, 39, 6440 (2006).
E. Ihara, M. Kida, M. Fujioka, N. Haida, T. Itoh, and K. Inoue, J. Polym. Sci., Part A: Polym. Chem., 45, 1536 (2007).
E. Ihara, T. Hiraren, T. Itoh, and K. Inoue, J. Polym. Sci., Part A: Polym. Chem., 46, 1638 (2008).
D. G. H. Hetterscheid, C. Hendriksen, W. I. Dzik, J. M. M. Smits, E. R. H. van Eck, A. E. Rowan, V. Busico, M. Vacatello, V. V. A. Castelli, A. Segre, E. Jellema, T. G. Bloemberg, and B. de Bruin, J. Am. Chem. Soc., 128, 9746 (2006).
E. Jellema, P. H. M. Budzelaar, J. N. H. Reek, and B. de Bruin, J. Am. Chem. Soc., 129, 11631 (2007).
T. Oyama, T. Fukui, K. Naka, and Y. Chujo, Polym. Bull., 43, 183 (1999).
A. Ouihia, L. Rene, J. Guilhem, C. Pascard, and B. Badet, J. Org. Chem., 58, 1641 (1993).
A. Ouihia, L. Rene, and B. Badet, Tetrahedron Lett., 33, 5509 (1992).
Although the signal in the aromatic region derived from 5′ apparently overlaps with the residual CHCl3; signal, the integral ratio for the latter was excluded for the calculation of the composition by using the paper weight of the peak areas throughout this study.
T. Maeda, K. Yamamoto, and T. Aoyagi, J. Colloid Interface Sci., 302, 467 (2006).
H. Wakamatsu, K. Yamamoto, A. Nakao, and T. Aoyagi, J. Magn. Magn. Mater., 302, 327 (2006).
T. Maeda, M. Takenouchi, K. Yamamoto, and T. Aoyagi, Biomacromolecules, 7, 2230 (2006).
D. Matsukuma, K. Yamamoto, and T. Aoyagi, Langmuir, 22, 5911 (2006).
T. Maeda, T. Kanda, Y. Yonekura, K. Yamamoto, and T. Aoyagi, Biomacromolecules, 7, 545 (2006).
R. L. Danheiser, R. F. Miller, R. G. Brisbois, and S. Z. Park, J. Org. Chem., 55, 1959 (1990).
C. J. Blankley, F. J. Sauter, and H. O. House, “Organic Synthesis,” Wiley, New York, 1973, Collect. Vol. V, p 258.
L. S. Hegedus, “Organometallics in Synthesis. A Manual,” M. Schlosser, Ed., John Wiley and Sons, West Sussex, 2002, p 1126.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ihara, E., Hiraren, T., Itoh, T. et al. Palladium-mediated Polymerization of Diazoacetamides. Polym J 40, 1094–1098 (2008). https://doi.org/10.1295/polymj.PJ2008145
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2008145