Abstract
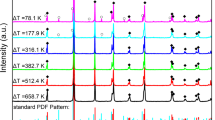
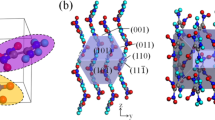
The morphology and crystal growth kinetics of isotactic polybutene-1 (it-PB1) trigonal phase in molten thin films have been studied with transmission electron microscopy, electron diffraction and optical microscopy. The growth rate of trigonal crystals was determined by in situ optical microscopy. It is one hundredth that of it-PB1 tetragonal crystals. The growth rate of trigonal crystals, as well as that of tetragonal crystals, shows the supercooling dependence derived from the nucleation theory. Trigonal crystals grown at 75 and 90.1 °C possessed well facetted morphology, which suggests the existence of flat growth faces required for nucleation theory between 75 and 90.1 °C. This is consistent with the observed temperature dependence of trigonal crystal growth rate in accordance with the nucleation theory. Lateral surface free energy σ of the trigonal phase determined from the observed growth kinetics is about 7 times as large as the value σHoff calculated according to Hoffman’s equation, while that of the tetragonal phase is roughly in agreement with the estimation. The discrepancy between the values of σ and σHoff for the trigonal phase can be attributed to the loss of conformational entropy of the ethyl side chains of it-PB1.
Similar content being viewed by others
Article PDF
References
R. L. Miller and V. F. Holland, J. Polym. Sci., Part C: Polym. Lett., 2, 519 (1964).
F. Danusso and G. Gianotti, Macromol. Chem., 61, 139 (1963).
A. Turner-Jones, Polymer, 7, 23 (1976).
A. Turner-Jones, J. Polym. Sci., Part C: Polym. Lett., 1, 455 (1963).
G. Natta, P. Corradini, and I. W. Bassi, Nuovo Cimento Suppl., 15, 52 (1960).
V. F. Holland and R. L. Miller, J. Appl. Phys., 35, 3241 (1964).
C. Nakafuku and T. Miyaki, Polymer, 24, 141 (1984).
J. Powers, J. D. Hoffman, J. J. Weeks, and F. A. Quinn Jr., J. Res. Natl. Bur. Stand. (U.S.), 69A, 335 (1965).
S. Kopp, J. C. Wittmann, and B. Lotz, Polymer, 35, 916 (1994).
B. Zhang, D. Yang, and S. Yan, J. Polym. Sci., Part B: Polym. Phys., 40, 2641 (2002).
M. Yamashita, A. Hoshino, and M. Kato, J. Polym. Sci., Polym. Phys. Ed., 45, 684 (2007).
M. Yamashita, J. Cryst. Growth., 310, 1739 (2008).
M. Yamashita and T. Takahashi, Kobunshi Ronbunshu, 65, 218 (2008).
M. Yamashita and S. Ueno, Cryst. Res. Technol., 42, 1222 (2007).
M. Yamashita, H. Miyaji, K. Izumi, and A. Hoshino, Polym. J., 36, 226 (2004).
H. Miyaji, Y. Miyamoto, K. Taguchi, A. Hoshino, M. Yamashita, O. Sawanobori, and A. Toda, J. Macromol. Sci., B42, 867 (2003).
M. Yamashita and T. Takahashi, in “Modern Research and Educational Topics in Microscopy,” A. Méndez-Vilas, Ed., Formatex Research Center, Badajoz, Spain, 2007, Vol. 2, p 713.
M. Yamashita and M. Kato, J. Appl. Crystallogr., 40, s650 (2007).
U. Leute and W. Dollhopf, Colloid Polym. Sci., 261, 299 (1983).
M. Yamashita and M. Kato, J. Appl. Crystallogr., 40, s558 (2007).
J. D. Hoffman and R. L. Miller, Polymer, 38, 3151 (1997).
J. D. Hoffman, G. T. Davis, and J. I. Lauritzen Jr., in “Treatise on Solid State Chemistry,” N. B. Hannay, Ed., Plenum, New York, 1976, chap. 7, p. 497.
K. Tashiro, A. Saiani, S. Miyashita, Y. Chatani, and H. Tadokoro, Polym. Prepr., Jpn., 47, 3869 (1998).
A. Toda, J. Chem. Phys., 118, 8446 (2003).
M. Kurata and Y. Tsunashima, in “Polymer Handbook” 4th ed., J. Brandrup, E. H. Immergut, and E. A. Grulke, Ed., Interscience Publishers, New York, 1999, chap. 6, pp VI/48–49.
S. Spaepen, Acta. Metall. Mater., 23, 729 (1975).
D. Turnbull and F. Spaepen, J. Polym. Sci., Polym. Symp., 63, 237 (1963).
J. D. Hoffman, Polymer, 33, 2643 (1992).
J. D. Hoffman, R. L. Miller, H. Marrand, and D. B. Roitman, Macromolcules, 25, 2221 (1992).
D. Maring, B. Meurer, and G. Weill, J. Polym. Sci., Part B: Polym. Phys., 33, 1235 (1995).
T. Miyoshi, S. Hayashi, F. Imashiro, and A. Kaito, Macromolecules, 35, 6060 (2002).
T. Miyoshi, S. Hayashi, F. Imashiro, and A. Kaito, Macromolecules, 35, 2624 (2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamashita, M., Takahashi, T. Melt Crystallization of isotactic Polybutene-1Trigonal Form: the Effect of Side Chain Entropy on Crystal Growth Kinetics. Polym J 40, 996–1004 (2008). https://doi.org/10.1295/polymj.PJ2007196
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2007196
Keywords
This article is cited by
-
The enhanced II–I transition behaviors of an isotactic polybutene-1 alloy by a TAB-3
Journal of Materials Science (2021)
-
Synchrotron microbeam X-ray scattering study of the crystallite orientation in the spherulites of isotactic poly(butene-1) crystallized isothermally at different temperatures
Polymer Journal (2019)
-
Kinetic roughening transition and missing regime transition of melt crystallized polybutene-1 tetragonal phase: growth kinetics analysis
Frontiers of Chemical Engineering in China (2009)
-
Lamellar thickness transition of melt-crystallized polybuten-1 tetragonal phase: configurational change in chain folding directions
Frontiers of Chemical Engineering in China (2009)