Abstract
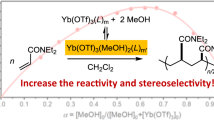
An asymmetric oxidative cross-coupling polymerization with a binary catalyst system of an achiral or chiral copper complex and a Lewis acid was investigated. Both the copper and Lewis acid catalysts significantly affected the catalyst activity, cross-coupling and stereoselectivities during the polymerization. The polymerization of methyl 3,7-dihydroxynaphthalene-2-carboxylate using a copper(I)-bisoxazoline catalyst at room temperature under an O2 atmosphere in the presence of Yb(OTf)3 proceeded with a cross-coupling selectivity of 97%.
Similar content being viewed by others
Article PDF
References
J. M. Brunel, Chem. Rev., 105, 857 (2005).
M. Putala, Enantiomer, 4, 243 (1999).
M. Hovorka, J. Günterová, and J. Závada, Tetrahedron Lett., 31, 413 (1990).
M. Hovorka, R. Šcigel, J. Günterová, M. Tichý, and J. Závada, Tetrahedron, 48, 9503 (1992).
M. Smrcina, J. Poláková, Š. Vyskocil, and P. Kocovský, J. Org. Chem., 58, 4534 (1993).
M. Smrcina, Š. Vyskocil, B. Máca, M. Polášek, T. A. Claxton, A. P. Abbott, and P. Kocovský, J. Org. Chem., 59, 2156 (1994).
T. Temma and S. Habaue, Tetrahedron Lett., 46, 5655 (2005).
T. Temma, B. Hatano, and S. Habaue, Tetrahedron, 62, 8559 (2006).
S. Habaue, Y. Takahashi, and T. Temma, Tetrahedron Lett., 48, 7301 (2007).
P. Yan, Y. Sugiyama, Y. Takahashi, H. Kinemuchi, T. Temma, and S. Habaue, Tetrahedron, 64, 4325 (2008).
T. Temma and S. Habaue, J. Polym. Sci., Part A: Polym. Chem., 43, 6287 (2005).
T. Temma and S. Habaue, J. Polym. Sci., Part A: Polym. Chem., 46, 1034 (2008).
T. Temma, Y. Takahashi, Y. Yoshii, and S. Habaue, Polym. J., 39, 524 (2007).
T. Temma, B. Hatano, and S. Habaue, Polymer, 47, 1845 (2006).
L. Pu, Chem. Rev., 104, 1687 (2004).
L. Ma, P. S. White, and W. Lin, J. Org. Chem., 67, 7577 (2002).
A. K.-Y. Jen, Y. Liu, Q.-S. Hu, and L. Pu, Appl. Phys. Lett., 75, 3745 (1999).
M. Nakajima, I. Miyoshi, K. Kanayama, S. Hashimoto, M. Noji, and K. Koga, J. Org. Chem., 64, 2264 (1999).
X. Li, J. Yang, and M. C. Kozlowski, Org. Lett., 3, 1137 (2001).
J. Gao, J. H. Reibenspies, and A. E. Martell, Angew. Chem., Int. Ed., 42, 6008 (2003).
Y. Yamamoto, S. Onuki, M. Yumoto, and N. Asao, J. Am. Chem. Soc., 116, 421 (1994).
G. S. C. Srikanth and S. L. Castle, Tetrahedron Lett., 61, 10377 (2005).
J. Zimmerman and M. P. Sibi, Top. Curr. Chem., 263, 107 (2006).
C. L. Mero and N. A. Porter, J. Org. Chem., 65, 775 (2000).
H. Baraki, S. Habaue, and Y. Okamoto, Macromolecules, 34, 4724 (2001).
Y. Isobe, D. Fujioka, S. Habaue, and Y. Okamoto, J. Am. Chem. Soc., 123, 7180 (2001).
B. Ray, Y. Isobe, K. Morioka, S. Habaue, Y. Okamoto, M. Kamigaito, and M. Sawamoto, Macromolecules, 36, 543 (2003).
S. Habaue, T. Temma, Y. Sugiyama, and P. Yan, Tetrahedron Lett., 48, 8595 (2007).
S. R. Wyatt, Q.-S. Hu, X.-L. Yan, W. B. Bare, and L. Pu, Macromolecules, 34, 7983 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, P., Temma, T. & Habaue, S. Lewis-Acid-Assisted Highly Selective Oxidative Cross-Coupling Polymerization with Copper Catalysts. Polym J 40, 710–715 (2008). https://doi.org/10.1295/polymj.PJ2008052
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2008052
Keywords
This article is cited by
-
Synthesis of regiocontrolled triarylamine-based polymer with a naphthol unit
Polymer Bulletin (2021)
-
Direct synthesis of functional novolacs and their polymer reactions
Polymer Journal (2010)