Abstract
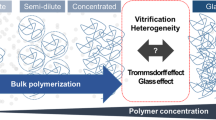
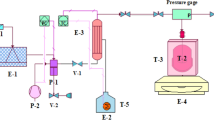
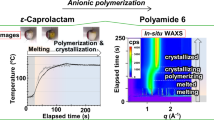
The applicability of the thermodynamic considerations of Self-Heating High-Temperature Synthesis (SHS) for frontal polymerization of acrylamide and triethylene glycol dimethacrylate is reported. The basic concept of our work is based on certain analogies existing between frontal polymerization and SHS processes. The measurements involve the determination of the heat capacity of the starting materials, as well as the product’s. The apparent activation energy of the reaction has also been studied. On the basis of the results, further new models are also set up. For instance, the role of convective flow in the low temperature initial phase might be supposed.
Similar content being viewed by others
Article PDF
References
J. P. Lebrat and A. Varma, Chem. Eng. Sci., 47, 2179 (1992).
B. S. Massey, “Units, Dimensional Analysis and Physical Similarity,” Van Nostrand-Reinhold, Princeton, New Jersey, 1971.
J. B. Holt and S. D. Dunmead, Annu. Rev. Mater. Sci., 21, 305 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeszenszky, A., Szabó, S. & Nagy, I. Comparative Review on SHS Thermodynamics Applied for Frontal Polymerization of Acrylamide and Triethylene Glycol Dimethacrylate. Polym J 38, 364–368 (2006). https://doi.org/10.1295/polymj.38.364
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.38.364