Abstract
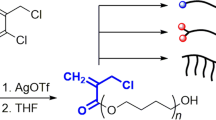
A specially designed N,N-dialkylmethacrylamide having a benzoyl chloride moiety in the N-alkyl chain (T) was prepared as a terminator for anionic polymerization of N,N-diethylacrylamide (DEA) to synthesize an end-functionalized polymer. Living poly(DEA) with narrow molecular weight distribution, which was produced by means of anionic polymerization of DEA with Ph2CHK in the presence of diethylzinc at 0 °C, reacted with T to afford an end-functionalized poly(DEA) carrying a methacrylamide group at the chain end. The end-group functionality of the polymer obtained was estimated to be up to 98% by 1H and 13C NMR and MALDI-TOF-MS analysis. However, the sufficient functionalization was not achieved because of side reactions when the living polymer generated with an organolithium initiator was used. Although the end-functionalized poly(DEA) is a macromonomer, neither polymerization nor oligomerization took place with any anionic initiator, but only a monoaddition product between them was obtained because N,N-disubstituted methacylamides are difficult to be polymerized. The end-functionalized poly(DEA) reacted with Ph2CHK equivalently to afford a well-defined anionic macroinitiator that initiates another anionic polymerization of DEA to yield a postpolymer consisting of two poly(DEA) blocks.
Similar content being viewed by others
Article PDF
References
“Anionic Polymerization. Principles and Practical Applications,” H. L. Hsieh, R. P. Quirk, Ed, Marcel Dekker, Inc., N.Y., Basel, Hong Kong., 1996, pp 133–135.
A. G. Evans and D. B. George, J. Chem. Soc., 4653 (1961).
A. G. Evans and D. B. George, J. Chem. Soc., 141 (1962).
B. C. Anderson, G. D. Andres, P. Arthur, Jr., H. W. Jacobson, L. R. Melby, A. J. Playtis, and W. H. Sharkey, Macromolecules, 14, 1599 (1981).
“Anionic Polymerization. Principles and Practical Applications,” H. L. Hsieh, R. P. Quirk, Ed, Marcel Dekker, Inc., N.Y., Basel, Hong Kong., 1996, pp 279–291 and pp 353–365.
A. Hirao, M. Hayashi, and N. Haraguchi, Macromol. Rapid Commun., 21, 1171 (2000).
X. Xie and T. E. Hogen-Esch, Macromolecules, 29, 1746 (1996).
T. Kodaita, H. Tanahashi, and K. Hara, Polym. J., 22, 649 (1990).
Y. Okamoto and H. Yuki, J. Polym. Sci., Polym. Chem. Ed., 19, 2647 (1981).
D. Xie, S. Tomczak, and T. E. Hogen-Esch, J. Polym. Sci., Part A: Polym. Chem., 39, 1403 (2001).
M. Kobayashi, S. Okuyama, T. Ishizone, and S. Nakahama, Macromolecules, 32, 6466 (1999).
M. Kobayashi, T. Ishizone, and S. Nakahama, Macromolecules, 33, 4411 (2000).
T. Ishizone, A. Hirao, and S. Nakahama, Macromolecules, 24, 625 (1991).
A. Hirao, K. Takenaka, S. Packirisamy, K. Yamaguchi, and S. Nakahama, Makromol. Chem., 186, 1157 (1985).
D. L. Tuleen and B. A. Hess Jr., J. Chem. Educ., 48, 476 (1971).
R. P. Woodbery and M. W. Rathke, J. Org. Chem., 42, 1688 (1977).
R. P. Woodbery and M. W. Rathke, J. Org. Chem., 43, 881 (1978).
D. Xie, S. Tomczak, and T. E. Hogen-Esch, J. Polym. Sci., Part A: Polym. Chem., 39, 1403 (2001).
B. I. Nakhmanovich, T. N. Prudskova, A. A. A-Yakubovich, and A. H. E. Müller, Macromol. Rapid Commun., 22, 1243 (2001).
Our private data. The end-functionalities of poly(DEA) obtained by reactions of the living polymer with a (2,2-dimethyl-1,3-dioxolane-4-yl)methyl tosylate, 4-iodomethyl-2,2-dimethyl-1,3-dioxolane, N-(2-methoxycarbonylethyl)-N-propylmethacrylamide, and ethylene oxide were 23%, 36%, 25%, and 3%, respectively.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kobayashi, M., Hirata, M. & Takeishi, M. Synthesis of Well-Defined Poly(N,N-diethylacrylamide)s End-Functionalized with Methacrylamide Structure by Means of Anionic Polymerization. Polym J 36, 238–247 (2004). https://doi.org/10.1295/polymj.36.238
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.36.238