Abstract
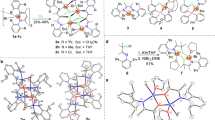
We have achieved the synthesis of neutral nickel catalyst with the modified O^N-chelating ligand for the ethylene polymerization. The activity of the catalyst, and the molecular weight and the branching structure of the polymer obtained strongly depend on the ligand structure as well as the presence of Ni(COD)2 which is used as an activator. The crystal structure of the catalyst was determined and the long nickel–phosphine bond length seems to play an important role for increasing the polymerization activity.
Similar content being viewed by others
Article PDF
References
S. D. Ittel, L. K. Johnson, and M. Brookhart, Chem. Rev., 100, 1169 (2000).
R. Soula, J. P. Broyer, M. F. Llauro, A. Tomov, R. Spitz, J. Claverie, X. Drujon, J. Malinge, and T. Saudemont, Macromolecules, 34, 2438 (2001).
V. C. Gibson and S. K. Spitzmesser, Chem. Rev., 103, 283 (2003).
C. Wang, S. Friedrich, T. R. Younkin, R. T. Li, R. H. Grubbs, D. A. Bansleben, and M. W. Day, Organometallics, 17, 3149 (1998).
L. K. Johnson, A. M. A. Bennett, S. D. Ittel, L. Wang, A. Parthasarathy, E. Hauptman, R. D. Simpson, J. Feldman, and E. B. Coughlin, WO Patent Application 9830609 to DuPont.
T. R. Younkin, E. F. Conner, J. I. Henderson, S. K. Friedrich, R. H. Grubbs, and D. A. Bnasleben, Science, 287, 460 (2000).
F. A. Hicks and M. Brookhart, Organometallics, 20, 3217 (2001).
F. M. Bauers and S. Mecking, Macromolecules, 34, 1165 (2001).
R. Soula, C. Novat, A. Tomov, R. Spitz, J. Claverie, X. Drujon, J. Malinge, and T. Saudemont, Macromolecules, 34, 2022 (2001).
R. Soula, B. Saillard, R. Spitz, J. Claverie, M. F. Llaurro, and C. Monnet, Macromolecules, 35, 1513 (2002).
S. Mecking, Angew. Chem. Int. Ed., 40, 534 (2001).
S. Mecking, A. Held, and F. M. Bauers, Angew. Chem., Int. Ed., 41, 544 (2002).
G. Bartoli, G. Palmieri, M. Bosco, and R. Dalpozzo, Tetrahedron Lett., 30, 2129 (1989).
D. R. Dobson, J. Gilmore, and D. A. Long, SYNLETT, 79 (1992).
R. A. Betlett, H. V. R. Dias, and P. P. Power, J. Organomet. Chem., 341, 1 (1988).
J. S. Baum, M. Condon, and D. A. Shook, J. Org. Chem., 52, 2983 (1987).
M. Hidai, T. Kashiwagi, T. Ikeuchi, and Y. Uchida, J. Organomet. Chem., 30, 279 (1971).
A. Altomare, G. Cascarno, C. Giacovazzo, and A. Gualardi, J. Appl. Crystallogr., 26, 343 (1993).
Bruker Analytical X-Ray Systems, Madison, WI (1998).
A. L. Spek, Acta Crystallogr., Sect. A, 46, C34 (1990).
PLATON, A Multipurpose Crystallographic Tool, Utrecht University, Utrecht, The Netherlands, A. L. Spek (2000).
B. M. Novak, G. Tian, M. Nodono, and P. Boyle, PMSE-Prepr., 223rd ACS Natl. Meet., Orland, FL, April 7–11, 2002; Am. Chem. Soc., Washington D.C., 2002, Vol. 86. p. 326.
Crystal data for 1: C45H43N2NiP, M = 701.49, monoclinic, temperature = 173(2) K, crystal size 0.40×0.40×0.22 mm3, a=16.832(3), b=11.7136(18), c=19.943(3) Å, V=3672.8(10) Å3, T=173(2) K, space group P21/c, Z=4, μ(Mo-Kα) = 0.606 mm−1, 26281 reflections collected, indipendent reflections = 6511 (Rint=0.065), final wR2=0.0812. Crystal data for 2: C47H50NNiOP, M = 734.56, orthorhombic, temperature = 173(2) K, crystal size 0.38×0.23×0.10 mm3, a=11.3284(12), b=18.380(2), c=18.663(2) Å, V=3885.9(7) Å3, T=173(2) K, space group P212121, Z=4, μ(Mo-Kα) = 0.577 mm−1, 34364 reflections collected, indipendent reflections = 8866 (Rint=0.0311), final wR2=0.1093.
The polymerization by 2 with B(C6F5)3 (2eq.) or MAO (2eq.) was conducted under the same condition of entry 4 in Table II.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nodono, M., Novak, B. & Boyle, P. Ethylene Polymerization Catalyzed by Neutral Nickel(II) Complex with O^N-Chelating Ligand. Polym J 36, 140–145 (2004). https://doi.org/10.1295/polymj.36.140
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.36.140