Abstract
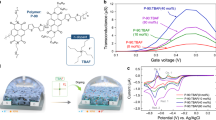
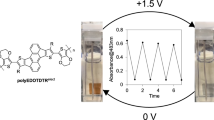
In this study, we synthesized thiophene derivatives by the protection of the carboxyl group of 3-thiophene acetic acid(monomer) with different substituted benzyl groups for hybridized electrochemical DNA sensors composed of a conductive polymer and oligo-DNA (ODN). In general, while 3-thiophene acetic acid is not suitable for direct electro-polymerization because of the electron withdrawing effect of the –COOH group in above monomer, our synthesized thiophene monomer derivatives are easily electro-oxidized to form stable electro-active polymer films by the protection of –COOH group. These polymer films showed the specific electrochemical characteristics of poly 3-alkylthiophenes with a reversible redox transition in the range of 0.8–1.6 V in CV (cyclic voltammogram) measurement. Biological recognition was monitored by comparison with the CV signal from the hybridization of single and double strands of ODN. The oxidation current of double strand ODN was less than that of the single strand, which corresponds to a decrease in electrochemical activity of the conducting polymer with an increase in stiffness of the side group of the electroactive polymer.
Similar content being viewed by others
Article PDF
References
S. Cosnier, Biosens. Bioelectron., 14, 443 (1999).
J. Wang, X. Cai, G. Rivas, H. Shiraishi, P. A. M. Farias, and N. Dontha, Anal. Chem., 68, 2629 (1996).
J. Wang, G. Liu, M. R. Jan, and Q. Zhu, Electrochem. Commun., 5, 1000 (2003).
J. Wang, Anal. Chim. Acta, 500, 247 (2003).
J. Wang, G. Liu, and A. Merkoçi, Anal. Chim. Acta, 482, 149 (2003).
G. Marrazza, I. Chianella, and M. Mascini, Biosens. Bioelectron., 14, 43 (1999).
K. Kerman, D. Ozkan, P. Kara, B. Meric, J. J. Gooding, and M. Ozsoz, Anal. Chim. Acta, 462, 39 (2002).
M. Zhao, D. B. Hibbert, and J. J. Gooding, Biosens. Bioelectron., 18, 827 (2003).
K. M. Millan and S. R. Mikkelsen, Anal. Chem., 65, 2317 (1993).
K. H. Row, Y. Polyakova, and S. K. Lee, Ind. Eng. Chem., 9, 473 (2003).
H. K.-Youssoufi, F. Garnier, P. Srivastava, P. Godillot, and A. Yassar, J. Am. Chem. Soc., 119, 7388 (1997).
P. Buerle, M. Hiller, S. Scheib, T. Msokolowski, and E. Umbach, Adv. Mater., 8, 214 (1996).
I. Mukoyama, K. Aoki, and J. Chen, J. Electroanal. Chem., 531, 133 (2002).
Y. Tezuka, K. Aoki, and T. Ishii, Electrochim. Acta, 44, 1871 (1999).
G. Li, G. Kobmehl, H. P. Welzel, G. Engelmann, W. D. Hunnius, W. Plieth, and H. Zhu, Marcromol. Chem. Phys., 199, 525 (1998).
R. L. Presdge, D. R. K. Harding, and W. S. Hancock, J. Org. Chem., 41, 2579 (1976).
P. Siebe, Helv. Chim. Acta, 60, 2711 (1977).
J. Cha, J. I. Han, Y. Choi, D. S. Yoon, K. W. Oh, and G. Lim, Biosens. Bioelectron., 18, 1241 (2003).
J. H. Lee, J. Y. Bae, and D. J. Chung, Polymer (Korea), 23, 129 (1999).
S. H. Jeong, J. Y. Bae, J. H. Kim, and D. J. Chung, Polymer (Korea), 26, 28 (2002).
G. Li, G. Kobmehl, H. P. Welzel, G. Engelmann, W. D. Hunnius, W. Plieth, and H. Zhu, Marcromol. Chem. Phys., 199, 2255 (1998).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kang, S., Kim, JH., An, J. et al. Synthesis of Polythiophene Derivatives and Their Application for Electrochemical DNA Sensor. Polym J 36, 937–942 (2004). https://doi.org/10.1295/polymj.36.937
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.36.937
Keywords
This article is cited by
-
Polythiophenes and polythiophene-based composites in amperometric sensing
Analytical and Bioanalytical Chemistry (2013)