Abstract
Imprinted cyclodextrin polymers are prepared and applied to stationary phases of high performance liquid chromatography (HPLC) for the selective recognition of steroids and other hydrophobic guest molecules. Molecular-imprinting efficiently promotes the binding-affinity and substrate-selectivity towards the template molecules compared with the control polymers prepared in their absence. When rigid and hydrophobic molecules such as cholesterol are imprinted, retention times towards the template molecules are notably enhanced. Thus, imprinting of cyclodextrins is effective for the preparation of synthetic tailor-made receptors for various kinds of hydrophobic guest molecules, and the polymers are potentially applicable to selective recognition of biologically important molecules or removal of toxic molecules from aqueous media.
Similar content being viewed by others
Article PDF
References
J.-M. Lehn, “Supramolecular Chemistry”, VCH, Weinheim, 1995.
M. M. Conn and J. Rebek, Jr., Chem. Rev., 97, 1647 (1997).
J. Vicens and V. Bohmer, Ed., “Calixarenes: A Versatile Class of Macrocyclic Compounds”, Kluwer Academic Publishers, Dordrecht, 1991.
J. Vicens, Z. Asfari, and J.M. Harrowfield, Ed., “Calixarenes 50th Anniversary: Commemorative Volume”, Kluwer Academic Publishers, Dordrecht, 1994.
H. S. Park, Q. Lin, and A. D. Hamilton, J. Am. Chem. Soc., 121, 8 (1999).
P. Wallimann, T. Marti, A. Furer, and F. Diederich, Chem. Rev., 97, 1567 (1997).
M. L. Bender and M. Komiyama, “Cyclodextrin Chemistry”, Springer-Verlag GmbH & Co, Berlin 1978.
J. Szèjtlí, “Cyclodextrin Technology”, Kluwer Academic Publishers, Budapest (1988).
K. A. Connors, Chem. Rev., 97, 1325 (1997).
A. Harada, Adv. Polym. Sci., 133, 141 (1997).
M. R. de Jong, J. F. J. Engbersen, J. Huskens, and D. N. Reinhoudt, Chem. Eur. J., 6, 4034 (2000).
Z. Yang and R. Breslow, Tetrahedron Lett., 38, 6171 (1997).
R. Breslow and B. Zhang, J. Am. Chem. Soc., 118, 8495 (1996).
R. Breslow, Z. W. Yang, R. Ching, G. Trojandt, and F. Odobel, J. Am. Chem. Soc., 120, 3536 (1998).
D. K. Leung, Z. W. Yang, and R. Breslow, Proc. Natl. Acad. Sci. U.S.A., 97, 5050 (2000).
K. Haupt and K. Mosbach, Trends Biotechnol., 16, 468 (1998).
K. Mosbach, Trends Biochem. Sci., 19, 9 (1994).
D. A. Spivak and K. J. Shea, Macromolecules, 31, 2160 (1998).
K. J. Shea and D. Y. Sasaki, J. Am. Chem. Soc., 113, 4109 (1991).
M. J. Whitcombe, M. E. Rodriguez, P. Villar, and E. N. Vulfson, J. Am. Chem. Soc., 117, 7105 (1995).
B. Sellergren, J. Wieschemeyer, K. S. Boos, and D. Seidel, Chem. Mater., 10, 4037 (1998).
B. Sellergren, Angew. Chem., Int. Ed. Eng., 39, 1031 (2000).
M. E. Davis, A. Katz, and W. R. Ahmad, Chem. Mater., 8, 1820 (1996).
O. Ramström and K. Mosbach, Curr. Opin. Chem. Biol., 3, 759 (1999).
G. Wülff, Angew. Chem., Int. Ed. Eng., 34, 1812 (1995).
T. Takeuchi and J. Haginaka, J. Chromatogr. B, 728, 1 (1999).
J. Matsui, Y. Miyoshi, O. Doblhoff-Dier, and T. Takeuchi, Anal. Chem., 67, 4404 (1995).
C. J. Allender, K. R. Brain, and C. M. Heard, “Progress in Medicinal Chemistry”, 2nd ed, F. D. King and A. W. Oxford, Ed., Elsevier Science Publishers B. V., Amsterdam, 1999. vol. 36, chapt. 6, p 235.
J. U. Klein, M. J. Whitcombe, F. Mulholland, and E. N. Vulfson, Angew. Chem., Int. Ed. Eng., 38, 2057 (1999).
V. T. Remcho and Z. J. Tan, Anal. Chem., 71, 248 A (1999).
H. Asanuma, T. Hishiya, and M. Komiyama, Adv. Mater., 12, 1019 (2000).
H. Asanuma, T. Akiyama, K. Kajiya, T. Hishiya, and M. Komiyama, Anal. Chim. Acta, 435, 25 (2001).
H. Asanuma, K. Kajiya, T. Hishiya, and M. Komiyama, Chem. Lett., 665 (1999).
S. A. Piletsky, H. S. Andersson, and I. A. Nicholls, Macromolecules, 32, 633 (1999).
T. Hishiya, M. Shibata, M. Kakazu, H. Asanuma, and M. Komiyama, Macromolecules, 32, 2265 (1999).
T. Hishiya, H. Asanuma, and M. Komiyama, J. Am. Chem. Soc., 124, 570 (2002).
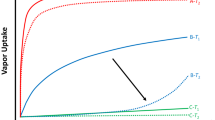
At a higher ratio of water in the eluent, peaks were too broadened to determine retention times precisely.
Present imprinting made the peak broadened as observed in the conventional imprinting. But, it did not affect the measurement of retention times for the guests at all.
High hydrophobicity of cholesterol compared with other steroids also contributed to this remarkable imprinting effect.
NOE was observed between CyD molecules and the guest (p-terphenyl) in DMSO, indicating that complex was formed during polymerization. UV spectrum was also changed (peak shift was observed) on addition of β-CyD in DMSO.
This method has an advantage in preparing artificial receptors for the chemicals sparingly soluble in water.
Phenoxathiin is similar in structure to dioxin compounds except for the lack of halogen atoms.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hishiya, T., Asanuma, H. & Komiyama, M. Molecularly Imprinted Cyclodextrin Polymers as Stationary Phases of High Performance Liquid Chromatography. Polym J 35, 440–445 (2003). https://doi.org/10.1295/polymj.35.440
Issue Date:
DOI: https://doi.org/10.1295/polymj.35.440
Keywords
This article is cited by
-
Development of a new chemically modified carbon paste electrode based on nano-sized molecular imprinted polymer for selective and sensitive determination of naproxen
Journal of Materials Science: Materials in Electronics (2016)
-
Photochemical molecular imprinting of cholesterol
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2008)