Abstract
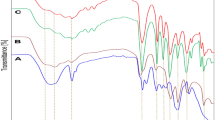
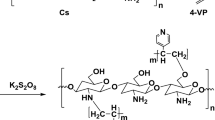
Chitosan-graft-poly(vinyl acetate) copolymers were prepared by a graft copolymerization with a redox initiator, cerium ammonium nitrate. Chitosan molecules not only participated in the reaction, but also served as a role of surfactant. As a result, a stable dispersion solution was observed during the reaction. After drying, particulate membranes were formed because of the rigid chitosan chains at particles’ surface. When the membranes were soaked in the water, the hydrophilic chitosan absorbed a great amount of water, but not the hydrophobic poly(vinyl acetate) chains mainly constituted in the core of particles. A two-stage behavior was observed during the adsorption of copper ion in the copolymer membrane. The first stage was governed by both the diffusion and reaction; whereas only diffusion dominated the second stage. The diffusion rate increased with an increase in the amount of chitosan. In order to increase its mechanical strength and acid resistance, the copolymer membrane was cross-linked with a glutaraldehyde. However, the specific adsorption quantity of copper ion decreased as the extent of cross-linking was increased.
Similar content being viewed by others
Article PDF
References
K. Aoi, A. Takasu, and M. Okada, Macromolecules, 30, 6134 (1997).
K. Inoue, K. Yoshizuka, and K. Ohto, Anal. Chim. Acta, 388, 209 (1999).
Y. Kawamura, M. Mitsuhashi, and H. Tanibe, Ind. Eng. Chem. Res., 32, 386 (1993).
E. Guibal, C. Milot, and J. M. Tobin, Ind. Eng. Chem. Res., 37, 1454 (1998).
K. Inoue, Y. Baba, and K. Yoshiguza, Bull. Chem. Soc. Jpn., 66, 2915 (1993).
G. L. Rorrer, T. Y. Hsien, and J. D. Way, Ind. Eng. Chem. Res., 32, 2170 (1993).
T.-M. Don, C.-F. King, and W.-Y. Chiu, The Synthesis and Properties of Chitosan-Modified Poly(vinyl acetate), J. Appl. Polym. Sci., (in press).
G. Odian and J. H. T. Kho, J. Macromol. Sci. Chem., A4, 317 (1970).
K. H. Kim, K. S. Kim, and J. S. Shin, Polymer (Korea), 11, 133 (1987).
T. Graczyk and V. Hornof, J. Polym. Sci., Part A: Polym. Chem., 26, 2019 (1988).
T. Graczyk, J. Macromol. Sci. Chem., A27, 23 (1990).
R. F. Storey and L. J. Goff, Macromolecules, 22, 1058 (1989).
M. J. Fernandez, I. Casinos, and J. M. Guzman, J. Polym. Sci., Part A: Polym. Chem., 28, 2275 (1990).
Y.-R. Chen, Master Thesis in Chemical Engineering, National Taiwan University, Taipei, Taiwan, 1997. p 35.
K. Toei and T. Kohara, Anal. Chim. Acta, 83, 59 (1975).
M.-L. Tsai, Ph. D. Dissertation in Department of Food Science, National Taiwan Ocean University, Keelung, Taiwan. 1997, p 17.
E. Onsoyen and Q. Skaugrud, J. Chem. Technol. Biotechnol., 49, 396 (1990).
C.-F. King, Master Thesis in Materials Science and Engineering, National Taiwan University, Taipei, Taiwan, 2000, p 62.
G. M. Barrow, “Physical Chemistry”, 6th ed, McGraw-Hill, World Scientific Publishing Co. Pte. Ltd., Singapore, chapt. 17, 1996, pp 839–840.
T.-Y. Hsien and G. L. Rorrer, Ind. Eng. Chem. Res., 36, 3631 (1997).
T.-Y. Hsien and G. L. Rorrer, Sep. Sci. Technol., 30, 2455 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Don, TM., King, CF. & Chiu, WY. Preparation of Chitosan-graft-poly(vinyl acetate) Copolymers and Their Adsorption of Copper Ion. Polym J 34, 418–425 (2002). https://doi.org/10.1295/polymj.34.418
Issue Date:
DOI: https://doi.org/10.1295/polymj.34.418
Keywords
This article is cited by
-
Investigation of poly(methyl acrylate) grafted chitosan as a polymeric drug carrier
Polymer Bulletin (2014)
-
Synthesis and Characterization of Chitosan-graft-Poly(N-Allyl Maleamic Acid) Hydrogel Membrane
Water, Air, & Soil Pollution (2013)
-
Grafting of acryloyl cyanoacetohydrazide onto chitosan
Journal of Polymer Research (2011)
-
Drug–polymer interaction in the all-trans retinoic acid release from chitosan microparticles
Journal of Thermal Analysis and Calorimetry (2007)