Abstract
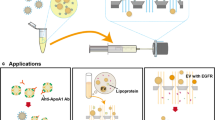
Amphiphilic poly(ethylene glycol)-b-polylactide (PEG/PLA) block macromonomers with an aldehyde group at the PEG chain end and a methacryloyl group at the PLA chain end were quantitatively synthesized by anionic polymerization. PEGylated latex particles were then prepared by dispersion radical copolymerization of the PEG/PLA block macromonomer with styrene in water. PEG/PLA macromonomer itself works as a surfactant to stabilize styrene droplets in the copolymerization process. Acetal groups at the PEG chain end on the latex particle were quantitatively converted to aldehyde groups by an acid treatment. In this way, polystyrene latex particles covered with PEG tethered chains possessing an aldehyde end-group were obtained. The size of the latex was quantitatively controlled ranging in size from 56 nm to 235 nm by the amount of the block macromonomer. The number of aldehyde groups on the latex particle was estimated to be approximately 0.029 molecules nm-2 by electric spin resonance (ESR) using 4-amino-2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) as a probe. Regardless of the size of the latex particles synthesized in this study, the occupied area of 4-amino-TEMPO on the particle was constant. Biotin was then introduced in a distal end of PEG-strands as a model ligand. The biotin-modified PEGylated latex was coagulated in a facile manner by the addition of avidin. From an inhibitory coagulation test using free biotin, it was found that more than 5.8 × 108 times of free biotin was required for the prevention of coagulation. Such a high complex formation ability of the biotin on the latex surface to avidin may be explained by the hypervalency effect. The core-shell type polystyrene latex particles, with reactive PEG brushes thus prepared, show a stable dispersity and a lowered non-specific interaction with proteinous compounds, and may have a promising potential in the diagnostic field.
Similar content being viewed by others
Article PDF
References
E. S. Daniels, E. D. Sudol, and M. S. El-Aasser, Ed., “Polymer Latexes”, American Chemical Society, Washington, D.C., 1992.
J. M. Singer and C. M. Plotz, Am. J. Med., 21, 888 (1956).
S. Kawaguchi, M. A. Winnik, and K. Ito, Macromolecules, 28, 1159 (1995).
Y. J. Kim, Y. Nagasaki, K. Kataoka, M. Kato, M. Yokoyama, T. Okano, and Y. Sakurai, Polym. Bull., 33, 1 (1994).
S. Cammas, Y. Nagasaki, and K. Kataoka, Bioconjugate Chem., 6, 226 (1995).
Y. Nagasaki, T. Kutsuna, M. Iijima, M. Kato, and K. Kataoka, Bioconjugate Chem., 6, 231 (1995).
Y. Nagasaki, M. Iijima, M. Kato, and K. Kataoka, Bioconjugate Chem., 6, 702 (1995).
Y. Nagasaki, R. Ogawa, S. Yamamoto, M. Kato, and K. Kataoka, Macromolecules, 30, 6489 (1997).
D. D. Perrin, W. L. F., F. W. Moor, Ed., “Purification of Laboratory Chemicals”, 2nd ed, Pergamom Press, Oxford, 1980.
M. Maniruzzaman, S. Kawaguchi, and K. Ito, Macromolecules, 33, 1583 (2000).
M. Akashi, D. Chao, E. Yashima, and N. Miyauchi, J. Appl. Polym. Sci., 39, 2027 (1990).
J. Liu, C. H. Chew, and L. M. Gan, J. macromol. Sci., Pure Appl. Chem., A33, 337 (1996).
Y. Wang and J. Huang, Macromolecules, 31, 13 (1998).
J. S. Shay, R. J. English, R. J. Spontak, C. M. Balik, and S. A. Khan, Macromolecules, 33, 6664 (2000).
A. Brindley, S. S. Davis, M. C. Davies, and J. F. Watts, J. Colloid Interface Sci., 171, 150 (1995).
I. Capec, “Macromolecular Reports”, A32(Suppls. 5&6), 1995, pp 749–758.
Y. Nagasaki, T. Okada, C. Scholz, M. Iijima, M. Kato, and K. Kataoka, Macromolecules, 31, 1473 (1998).
J. Chlebicki, J. Collod Interface Sci., 206, 77 (1998).
C. Wu, M. Akashi, and M.-Q. Chen, Macromolecules, 30, 2187 (1997).
M. Wilchek and E. A. Bayer, Ed., “Methods in Enzymology, Volume 184”, Academic Press, Inc., New York, N.Y., 1990.
M. Mammen, S.-K. Choi, and G. M. Whitesides, Angew. Chem. Int. Ed., 37, 2754 (1998).
H. Otsuka, Y. Nagasaki, and K. Kataoka, Biomacromolecules, 1, 39 (2000).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ogawa, R., Nagasaki, Y., Shibata, N. et al. Core–Shell Type Polystyrene Latex Possessing Reactive Poly(ethylene glycol) Brushes on the Surface for High Performance Immunodiagnostics. Polym J 34, 868–875 (2002). https://doi.org/10.1295/polymj.34.868
Issue Date:
DOI: https://doi.org/10.1295/polymj.34.868
Keywords
This article is cited by
-
A new time-resolved fluorometric microarray detection system using core–shell-type fluorescent nanosphere and its application to allergen microarray
Analytical and Bioanalytical Chemistry (2006)