Abstract
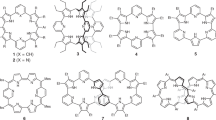
3-{4-[(3,5-Di-tert-butyl-4-acetoxyphenyl)(3,5-di-tert-butyl-4-oxocyclohexa-2,5-diene-1-ylidene)methyl]phenyl} thiophene (2a) was synthesized and oxidatively polymerized with ferric chloride at low temperature to yield poly(3-{4-[(3,5-di-tert-butyl-4-acetoxyphenyl)(3,5-di-tert-butyl-4-oxocyclohexa-2,5-diene-1-ylidene)methyl]phenyl} thiophene) (1a). Head-to-tail content of 1a with a molecular weight of >104 reached 92%. This polymer has a quinoid chromophore, and it reversibly showed a dark blue and red brownish color ascribed to an anion and a radical form, respectively. The polyradical 1 was almost quantitatively generated from 1b (a spin concentration of 0.9 spin/galvinoxyl unit), which was persistent at room temperature. The SQUID measurement at low temperature gave a spin quantum number (S) of 1/2 for 1, which indicated a paramagnetic ground state. 1 doped with iodine showed an electrical conductivity of 10-5 S cm-1. The polyradical derivatives, 7 and 8, displayed S of 1/2–2/2 and 1/2, respectively, and an electrical conductivity of 10-4–10-3 S cm-1 after doping.
Similar content being viewed by others
Article PDF
References
P. M. Lahti, “Magnetic Properties of Organic Materials,” Marcel Dekker, Inc., New York, N.Y., 1999.
K. Ito and M. Kinoshita, “Molecular Magnetism - New Magnetic Materials,” Kodansha and Gordon and Breach Science Publishers, Tokyo and Amsterdam, 2000.
N. Yoshioka, H. Nishide, T. Kaneko, H. Yoshiki, and E. Tsuchida, Macromolecules, 25, 3838 (1992).
H. Nishide, T. Kaneko, N. Yoshioka, H. Akiyama, M. Igarashi, and E. Tsuchida, Macromolecules, 26, 4567 (1993).
H. Nishide, T. Kaneko, M. Igarashi, E. Tsuchida, N. Yoshioka, and P. M. Lahti, Macromolecules, 27, 3082 (1994).
H. Nishide, T. Kaneko, T. Nii, K. Katoh, E. Tsuchida, and P. M. Lahti, J. Am. Chem. Soc., 118, 9695 (1996).
H. Nishide, T. Kaneko, S. Toriu, Y. Kuzumaki, and E. Tsuchida, Bull. Chem. Soc. Jpn., 69, 499 (1996).
H. Nishide, Y. Hozumi, T. Nii, and E. Tsuchida, Macromolecules, 30, 3986 (1997).
Y. Miura, Y. Ushitani, K. Inui, Y. Teki, T. Takui, and K. Itoh, Macromolecules, 26, 3698 (1993).
H. Oka, T. Tamura, Y. Miura, and Y. Teki, J. Mater. Chem., 9, 1227 (1999).
H. Oka, T. Tamura, Y. Miura, and Y. Teki, Polym. J., 31, 979 (1999).
T. Yamamoto and H. Hayashi, J. Polym. Sci., Polym. Chem. Ed., 35, 463 (1997).
H. Hayashi and T. Yamamoto, Macromolecules, 30, 330 (1997).
H. Hayashi and T. Yamamoto, Macromolecules, 31, 6063 (1998).
C. Xie and P. M. Lahti, J. Polym. Sci., Part A: Polym. Chem., 37, 779 (1999).
D. Fichou, “Handbook of Oligo- and Polythiophene,” John Wiley & Sons, Inc., New York, N.Y., 1999.
R. D. McCullough and R. D. Lowe, J. Chem. Soc., Chem. Comm., 70 (1992).
T.-A. Chen, X. Wu, and R. D. Rieke, J. Am. Chem. Soc., 117, 233 (1995).
S. Guillerez and G. Bidan, Synthetic Metals, 93, 123 (1998).
M. R. Andersson, D. Selse, M. Berggren, H. Jarvinen, T. Hjertberg, O. Inganas, O. Wennerstrom, and J.-E. Osterhorm, Macromolecules, 27, 6503 (1994).
S. Amou, O. Haba, K. Shirato, T. Hayakawa, M. Ueda, K. Takeuchi, and M. Asai, J. Polym. Sci., Part A: Polym Chem., 37, 1943 (1999).
M. Miyasaka, T. Yamazaki, E. Tsuchida, and H. Nishide, Macromolecules, 33, 8211 (2000).
M. Miyasaka, T. Yamazaki, E. Tsuchida, and H. Nishide, Polyhedron, 20, 1157 (2001).
W. Harrer, H. Kurreck, J. Reusch, and W. Gierke, Tetrahedron, 31, 625 (1975).
K. Mukai, Bull. Chem. Soc. Jpn., 48, 2405 (1975).
K. Awaga, T. Sugano, and M. Kinoshita, J. Chem. Phys., 85, 2211 (1986).
D. F. Evans, J. Chem. Soc., 2003 (1959).
S. K. Sur, J. Magn. Reson., 82, 169 (1989).
K. R. Stickley, T. D. Selby, and S. C. Blackstock, J. Org. Chem., 62, 448 (1997).
N. Miyaura and A. Suzuki, Chem. Rev., 95, 2457 (1995).
X. Bi, Q. Ying, and R. Qian, Makromol. Chem., 193, 2905 (1992).
T. Yamamoto, D. Oguro, and K. Kubota, Macromolecules, 29, 1833 (1996).
R. L. Carlin, “Magnetochemistry,” Springer-Verlag GmbH & Co., Berlin, 1986.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miyasaka, M., Yamazaki, T. & Nishide, H. Poly(3-phenylgalvinoxylthiophene). A New Conjugated Polyradical with High Spin Concentration. Polym J 33, 849–856 (2001). https://doi.org/10.1295/polymj.33.849
Issue Date:
DOI: https://doi.org/10.1295/polymj.33.849