Abstract
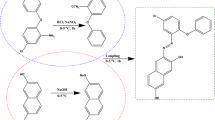
A pyrenyl-substituted chiral sulfoxide, (R)-(+)-1-pyrenyl 4-tolyl sulfoxide (R-TPYS) was synthesized and photoracemized in acetonitrile, benzene, toluene and chloroform. Quantum yields of their photoinversion reactions were 0.02–0.03 from the changes in CD spectra and the rates of decomposition measured by high-performance liquid chromatography (HPLC) during photoirradiation. Quantum yields of photoinversion for R-TPYS in poly(methyl methacrylate) (PMMA) and in polystyrene were almost the same as in solution. It is considered that photoinversion reaction of R-TPYS is not affected by the polymer matrix at room temperature. The reaction at 88K deviates from the first-order kinetics. The free volume around the sulfoxide in polymer matrix at 88K does not seem to be enough compared to the sweep volume necessary for the reaction to occur.
Similar content being viewed by others
Article PDF
References
G. Smets, Adv. Polym. Sci., 50, 17 (1983).
K. Horie and I. Mita, Adv. Polym. Sci., 88, 77 (1989).
K. Horie, M. Tsukamoto, and I. Mita, Eur. Polym. J., 21, 805 (1985).
K. Horie, K. Hirao, N. Kenmochi, and I. Mita, Makromol. Chem., Rapid Commun. 9, 267 (1988).
I. Mita, K. Horie, and K. Hirao, Macromolecules, 22, 558 (1989).
T. Naito, K. Horie, and I. Mita, Eur. Polym. J., 26, 1295 (1990).
T. Naito, K. Horie, and I. Mita, Macromolecules, 24, 2907 (1991).
T. Naito, K. Horie, and I. Mita, Polym. J., 23, 809 (1991).
T. Naito, K. Horie, and I. Mita, Polymer, 34, 4140 (1993).
M. C. Carreño, Chem. Rev., 95, 1717 (1995).
D. R. Rayner, A. J. Gordon, and K. Mislow, J. Am. Chem. Soc., 90, 4854 (1968).
A. G. Schultz, C. D. DeBoer, and R. H. Schlessinger, J. Am. Chem. Soc., 90, 5314 (1968).
A. G. Schultz and R. H. Schlessinger, J. Chem. Soc., Chem. Commun., 1483 (1969).
A. G. Schultz and R. H. Schlessinger, J. Chem. Soc., Chem. Commun., 1294 (1970).
B. S. Larsen, J. Kolc, and S. O. Lawesson, Tetrahedron, 27, 5163 (1971).
G. M. Gurria and G. H. Posner, J. Org. Chem., 38, 2419 (1973).
K. A. Muszkat, K. Praefcke, I. Khait, and R. Lüdersdorf, J. Chem. Soc., Chem. Commun., 898 (1979).
I. Khait, R. Lüdersdorf, K. A. Muszkat, and K. Praefcke, J. Chem. Soc., Perkin Trans. 2, 1417 (1981).
B. C. Gilbert, B. Gill, and M. D. Sexton, J. Chem. Soc., Chem. Commun., 78 (1978).
C. Chatgilialoglu, B. C. Gilbert, B. Gill, and M. D. Sexton, J. Chem. Soc., Perkin Trans. 2, 1141 (1980).
Y. Guo and W. S. Jenks, J. Org. Chem., 62, 857 (1997).
W. S. Jenks, W. Lee, and D. Shutters, J. Phys. Chem., 98, 2282 (1994).
G. Balavoine, S. Juge, and H. B. Kagan, Tetrahedron Lett., 42, 4159 (1973).
W. H. Pirkle and P. L. Rinaldi, J. Am. Chem. Soc., 99, 3510 (1977).
C. Eskenazi, J. F. Nicoud, and H. B. Kagan, J. Org. Chem., 44, 995 (1979).
L. Finzi, G. Maccagnani, S. Masiero, B. Samori, and P. Zani, Liq. Cryst., 6, 199 (1989).
Y. Inoue, Chem. Rev., 92, 741 (1992).
K. Mislow, M. Axelrod, D. R. Rayner, H. Gotthardt, L. M. Coyne, and G. S. Hammond, J. Am. Chem. Soc., 87, 4958 (1965).
G. S. Hammond, Hans Gotthardt, L. M. Coyne, M. Axelrod, D. R. Rayner, and K. Mislow, J. Am. Chem. Soc., 87, 4959 (1965).
R. S. Cooke and G. S. Hammond, J. Am. Chem. Soc., 90, 2958 (1968).
R. S. Cooke and G. S. Hammond, J. Am. Chem. Soc., 92, 2739 (1970).
K. K. Andersen, Tetrahedron Lett., 3, 93 (1962).
K. K. Andersen, W. Gaffield, N. E. Papanikolaou, J. W. Foley, and R. I. Perkins, J. Am. Chem. Soc., 86, 5637 (1964).
J. B. Birks, “Photophysics of Aromatic Molecules,” Wiley-Interscience, London, 1970, Chapter 1, pp 8–10.
J. B. Birks, “Photophysics of Aromatic Molecules,” Wiley-Interscience, London, 1970, Chapter 3, p 71.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsurutani, Y., Yamashita, T. & Horie, K. Photoinversion Reaction of a Pyrenyl Sulfoxide in Solution and in Polymer Matrices. Polym J 30, 11–16 (1998). https://doi.org/10.1295/polymj.30.11
Issue Date:
DOI: https://doi.org/10.1295/polymj.30.11