Abstract
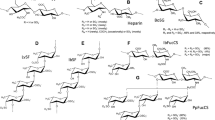
Anti-AIDS virus activity of synthetic and natural dextran sulfates was assayed in vitro by using a MT-4 cell line. These dextran sulfates with the number average molecular weight (M̅n) of 1.8×104−10.7×104 showed potent anti-AIDS virus activity in the concentration as low as 3.3 or 10 μg ml−1, respectively. This high activity was almost the same as that of curdlan sulfates which was reported previously. The degree of sulfation per glucose residue in polymer backbone was 1.0–1.9 (the highest theoretical value is 3.0) which was calculated from the elemental analysis. The relationship between the structure and anti-AIDS virus activity is discussed based on high resolution NMR spectroscopy.
Similar content being viewed by others
Article PDF
References
C. R. Ricketts, Biochemistry, 51, 129 (1951).
G. Oshima, T. Nagai, and K. Nagasawa, Thrombosis Res., 35, 601 (1984).
T. Uryu, in S. Penczek, Ed., “Models of Biopolymers by Ring-Opening Polymerization,” CRC Press, Boca Raton, Florida, 1990, p. 133.
E. R. Ruckel and C. Schuerch, J. Org. Chem., 31, 2233 (1966).
T. Uryu and C. Schuerch, Macromolecules, 4, 342 (1971).
T. Uryu and M. Yamanaka, M. Henmi, K. Hatanaka, and K. Matsuzaki, Carbohydr. Res., 157, 157 (1986).
K. Hatanaka, T. Hirobe, T. Yoshida, M. Yamanaka, and T. Uryu, Polym. J., 22, 435 (1990).
M. Torii, S. Ogawa, K. Watanabe, T. Koshikawa, M. Yamazoe, T. Uryu, and C. Schuerch, J. Biochem., 99, 263 (1986).
B. Ehlers, R. Rudolph, and H. Diringer, J. Gen. Virol., 65, 423 (1984).
H. Diringer, Jpn. Patent (Tokkai Sho) 62-215529 (1987).
R. Ueno and S. Kuno, Lancet, 1379 (1987).
H. Nakashima, O. Yoshida, T. Tochikura, T. Yoshida, T. Mimura, Y. Kido, Y. Motoki, Y. Kaneko, T. Uryu, and N. Yamamoto, Jpn. J. Cancer Res., 78, 1164 (1987).
H. Mitsuya, D. J. Looney, S. Kuno, R. Ueno, F. Ueno, F. W. Stall, and S. Broder, Science, 240, 646 (1988).
M. Baba, R. Pauwels, J. Balzarini, J. Arnout, J. Desmyter, and E. De Clercq, Proc. Natl. Acad. Sci. U.S.A., 85, 6132 (1988).
H. Nakashima, O. Yoshida, M. Baba, E. De Clercq, and N. Yamamoto, Antiviral Res., 11, 233 (1989).
N. R. Hartman, D. G. Johns, and H. Mitsuya, AIDS Res. Hum. Retroviruses, 6, 805 (1990).
L. N. Callahan, M. Phelan, M. Mallinson, and M. A. Norcross, J. Virol., 65, 1543 (1991).
M. Witvrouw, D. Schols, G. Andrei, R. Snoeck, M. Hosoya, R. Pauwels, J. Balzarini, and E. De Clercq, Antiviral Chem. Chemother., 2, 171 (1991).
J. Zachoval and C. Schuerch, J. Am. Chem. Soc., 91, 1165 (1969).
C. Schuerch and T. Uryu, Macromolecular Synth., 4, 151 (1972).
G. H. Hakimelahi, Z. A. Proba, and K. K. Ogilvie, Tetrahedron Lett., 4775 (1981).
D. Gagnaire and M. Vignon, Makromol. Chem., 178, 2321 (1977).
U. S. Pharmacopeia National Formulary, USP XXI, 1985.
Y. Kaneko, O. Yoshida, R. Nakagawa, T. Yoshida, M. Date, S. Ogihara, S. Shioya, Y. Matsuzawa, N. Nagashima, Y. Irie, T. Mimura, H. Shinkai, N. Yasuda, K. Matsuzaki, T. Uryu, and N. Yamamoto, Biochem. Pharmacol., 39, 793 (1990).
T. Yoshida, K. Hatanaka, T. Uryu, Y. Kaneko, E. Suzuki, H. Miyano, T. Mimura, O. Yoshida, and N. Yamamoto, Macromolecules, 23, 3717 (1990).
K. J. Lorentsen, C. W. Hendrix, J. M. Collins, D. M. Kornhauser, B. G. Petty, R. W. Kecker, C. Flexner, and R. H. Eckel, Ann. Intern. Med., 111, 561 (1989).
K. Hatanaka, T. Yoshida, T. Uryu, O. Yoshida, H. Nakashima, N. Yamamoto, T. Mimura, and Y. Kaneko, Jpn. J. Cancer Res., 80, 95 (1989).
T. Moriya, H. Kurita, K. Matsumoto, T. Otake, H. Mori, M. Moromoto, N. Ueba, and N. Kunita, J. Med. Chem., 34, 2301 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshida, T., Nakashima, H., Yamamoto, N. et al. Anti-AIDS Virus Activity in vitro of Dextran Sulfates Obtained by Sulfation of Synthetic and Natural Dextrans. Polym J 25, 1069–1077 (1993). https://doi.org/10.1295/polymj.25.1069
Issue Date:
DOI: https://doi.org/10.1295/polymj.25.1069
Keywords
This article is cited by
-
Conversion of levoglucosan into glucose by the coordination of four enzymes through oxidation, elimination, hydration, and reduction
Scientific Reports (2020)
-
Microwave pyrolysis of cellulosic materials for the production of anhydrosugars
Journal of Wood Science (2001)