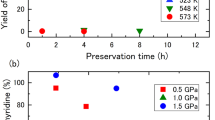
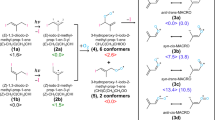
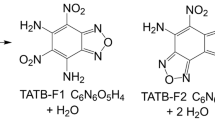
Abstract
Modes of thermal decomposition reactions of several azo-type initiators were studied in the presence or absence of a transition-metal complex. Some metal complexes examined cause dramatic influence upon the relative ratio of decomposition products. Particularly, a cobalt complex prevents the coupling reaction of radicals formed from azo compounds, with increased formation of noncoupling products being observed. Futhermore no difference was observed in the mole ratios of an unsymmetrical coupling product to the sum of symmetrical products in the decomposition of an unsymmetrical azo compound with or without a complex, Co(II)(sal)2((R)-CHXDA). This phenomenon was satisfactorily explained by assuming that primary radicals immediately formed interact with the metal complex and are stabilized prior to subsequent reactions.
Similar content being viewed by others
Article PDF
References
J. Thiele and K. Heuser, Ann., 290, 1 (1896).
C. G. Overberger, M. T. O’Shaughnessy, and H. Shalit, J. Am. Chem. Soc., 71, 2661 (1949).
C. G. Overberger and M. B. Berenbaum, J. Am. Chem. Soc., 73, 2618 (1950).
J. E. Falk, “Porphyrins and Metalloporphyrins,” Elsevier, Amsterdam, 1964.
P. Pothemund and A. R. Menotti, J. Am. Chem. Soc., 70, 1808 (1948).
H. Aoi, M. Ishimori, S. Yoshikawa, and T. Tsuruta, J. Organometal. Chem., 85, 241 (1975).
T. Hirano, T. Miki, and T. Tsuruta, Makromol. Chem., 104, 230 (1967).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshino, K., Ohkatsu, Y. & Tsuruta, T. Study on the Modes of Thermal Decomposition of Several Azo-Type Initiators. Polym J 9, 275–282 (1977). https://doi.org/10.1295/polymj.9.275
Issue Date:
DOI: https://doi.org/10.1295/polymj.9.275