Abstract
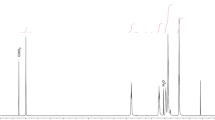
The reorientational correlation time in carbon tetrachloride of the phenyl groups of polystyrene was measured by Raman-line shape analysis. Up to the point where the molecular weight of the polymer reaches about 1.0×104, the larger the molecular weight, the longer is the reorientational correlation time of the phenyl groups. For molecular weights above 1.0×104, the reorientational correlation time of the phenyl groups levels off at about 4×10−10 sec. The phenyl groups of the polystyrene chains rotate about 130 times more slowly than the molecules of toluene in carbon tetrachloride.
Similar content being viewed by others
Article PDF
References
S. Kato, H. Kondo, I. Fujio, H. Nomura, and Y. Miyahara, J. Chem. Soc. Jpn., 1981 (1974).
F. A. Bovey, G. V. D. Ties, and G. Pillipovich, J. Polym. Sci., 38, 73 (1959).
F. A. Bovey, J. Chem. Phys. 32, 1887 (1960).
F. J. Bartoli and T. A. Litovitz, J. Chem. Phys., 56, 404 (1973).
F. J. Bartoli and T. A. Litovitz, J. Chem. Phys., 56, 413 (1973).
K. Ono, H. Shintani, O. Yano, and Y. Wada, Polym. J., 5, 164 (1973).
I. G. Mikhailov and E. B. Safina, Soviet-Phys., Acoustics, 17, 335 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nomura, H., Miyahara, Y. The Motion of Polymer Chains in Solutions. I. Reorientational Motion of Phenyl Groups of Polystyrene Chains in Solutions of Carbon Tetrachloride by Raman Line Shape Analysis. Polym J 8, 30–34 (1976). https://doi.org/10.1295/polymj.8.30
Issue Date:
DOI: https://doi.org/10.1295/polymj.8.30