Abstract
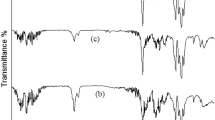
Chain scission of nylon 66 films due to exposure to nitrogen dioxide at a pressure of 0.5 mmHg has been studied as a function of temperature, film thickness, and polymer morphology. The rate of random chain scission in cast polymer films is very sensitive to changes in morphology caused by small variations in solvent composition for film casting and in mode of preparation. Decrease of chain scission with film thickness indicates that diffusion of NO2 into the polymer is rate-determining. The magnitude of the energy of activation is in agreement with this finding. It is assumed that amide links in the chain folds of polymer molecules located in the interfacial region between amorphous and crystalline polymer portions are especially susceptible to chain scission, due to their strain energy.
Similar content being viewed by others
Article PDF
References
H. H. G. Jellinek and A. Chaudhuri, J. Polym. Sci., Part A-1, 10, 1773 (1972).
A. C. G. Egerton, J. Chem. Soc., 105, 647 (1914).
see also Gmelin ’s Handbuch, Der Anorganischan Chemie, System 4, Lieferung 1–4, Verlag Chemie Leipzig, Berlin 1936 (1955) p. 748.
M. Dole and B. Wunderlich, Makromol. Chem., 34, 29 (1959).
H. H. G. Jellinek and S. Igarashi, J. Phys. Chem., 74, 1409 (1970).
P. Hrdlovic, J. Pavlinee, and H. H. G. Jellinek, J. Polym. Sci., Part A-1, 9, 1235 (1971).
see also D. C. Bassett, Polymer, 5, 457 (1964).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jellinek, H., Yokota, R. & Itoh, Y. Reaction of Nitrogen Dioxide with Nylon 66. Polym J 4, 601–606 (1973). https://doi.org/10.1295/polymj.4.601
Issue Date:
DOI: https://doi.org/10.1295/polymj.4.601