Abstract
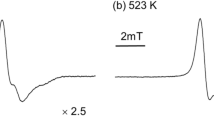
The ESR spectra of poly(2,6-dimethylphenylene oxide) (PPO) were recorded before and after irradiation both in the solid state and in solution. The PPO powder sample gave an apparent eight-line spectrum with a separation of about 5.5 gauss, which is due to substituted phenoxy radical (I).

Signal intensity increased after irradiation at room temperature. Irradiation at −196°C gave an apparent singlet spectrum with some shoulders.When a benzene solution of PPO was irradiated at room temperature in the presence of air, there was observed at an earlier stage an apparent seven-line spectrum with a coupling constant of 5.60 gauss, and after a longer time of irradiation a multi-component spectrum. Both spectra could be attributed to the free radical (I). On the other hand, irradiation in the presence of nitrogen or under vacuum gave only the multi-component spectrum.In all cases, the signal intensity was larger in the presence of air than under vacuum or in the presence of nitrogen.ESR spectra observed at higher temperature showed line width alternation. The mechanism of radical formation and the possible reactions during irradiation are discussed.
Similar content being viewed by others
Article PDF
References
K. Tsuji and T. Seiki, J. Polym. Sci., Part B, 7, 839 (1969).
K. Tsuji and T. Seiki, J. Polym. Sci., Part A-1, 9, 3063 (1971).
K. Tsuji and T. Seiki, Polym. J., 2, 606 (1971).
T. Takeshita, K. Tsuji, and T. Seiki, J. Polym. Sci., Part A-1, 10, 2315 (1972).
K. Tsuji, T. Seiki, and T. Takeshita, J. Polym. Sci., Part A-1, 10, 3119 (1972).
K. Tsuji and S. Okamura, Nobel Symposium No. 22, Stockholm, June 20–23, 1972.
K. Tsuji and T. Seiki, Polym. J., 1, 133 (1970).
K. Tsuji and T. Seiki, J. Polym. Sci., Part B, 8, 817 (1970).
K. Tsuji and T. Seiki, J. Polym. Sci., Part B, 10, 139 (1972).
K. Tsuji, K. Hayashi, and S. Okamura, J. Polym. Sci., Part A-1, 8, 583 (1970).
K. Tsuji and T. Seiki, J. Polym. Sci., Part A-1, 10, 123 (1972).
A. S. Hay, Adv. Polym. Sci., 4, 496 (1967).
W. G. B. Huysmans and W. A. Water, J. Chem. Soc., Part B, 1163 (1967).
P. G. Kelleher, L. B. Jassie, and B. D. Gasner, J. Appl. Polym. Sci., 11, 137 (1967).
M. C. R. Symons and J. K. Yandell, J. Chem. Soc., Part A, 1995 (1970).
E. R. Altwicker, Chem. Rev., 67, 475 (1967).
A. R. Forrester, J. M. Hay, and R. H. Thomson, “Organic Chemistry of Stable Free Radicals,” Academic Press, London, 1968, p 287.
R. T. Conley, J. Macromol. Sci., Chem., A1, 81 (1967).
A. Factor, J. Polym. Sci., Part A-1, 7, 363 (1969).
R. A. Jerussi, J. Polym. Sci., Part A-1, 9, 2009 (1971).
C. Steelink, J. D. Fitzpatrick, L. D. Kispert, and J. S. Hyde, J. Am. Chem. Soc., 90, 4354 (1968).
E. J. Land, G. Porter, and E. Strachen, Trans. Faraday Soc., 57, 1885 (1961).
G. Porter and E. Strachen, Trans. Faraday Soc., 54, 1595 (1958).
J. L. Roebber, J. Chem. Phys., 37, 1974 (1962).
H. Tsubomura, K. Kimura, H. Yamada, and M. Kato, Tetrahedron Lett., No. 47, 4217 (1965).
K. Kimura, K. Yoshinaga, and H. Tsubomura, J. Phys. Chem., 71, 4485 (1967).
J. E. Bennett and B. Mile, Trans. Faraday Soc., 67, 1587 (1971).
H. Tsubomura and R. S. Mulliken, J. Am. Chem. Soc., 82, 5966 (1960).
A. U. Munck and J. F. Scott, Nature, 177, 587 (1956).
V. I. Stenberg, R. D. Olson, C. T. Wang, and N. Kulevsky, J. Org. Chem., 32, 3227 (1968).
D. R. Snelling, Chem. Phys. Lett., 2, 346 (1968).
H. Kautsky and H. de Bruijn, Naturwissenschaften, 19, 1043 (1931).
H. Kautsky, H. de Bruijn, R. Neuwirth, and W. Baumeister, Ber. Deut. Chem. Ges., 66, 1588 (1933).
H. Kautsky, Biochem. Z., 291, 271 (1937).
D. R. Kearns and A. U. Khan, Photochem. Photobiol., 10, 193 (1969).
T. Matsuura, K. Omura, and R. Nakashima, Bull. Chem. Soc. Jpn., 38, 1358 (1965).
ref 17, p 294.
R. G. Caldwell and J. L. Ihring, J. Am. Chem. Soc., 84, 2878 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsuji, K., Seiki, T. Electron Spin Resonance Study of Poly(2,6-dimethylphenylene oxide) Irradiated with Ultraviolet Light. Polym J 4, 589–600 (1973). https://doi.org/10.1295/polymj.4.589
Issue Date:
DOI: https://doi.org/10.1295/polymj.4.589