Abstract
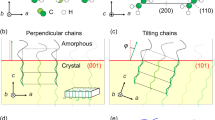
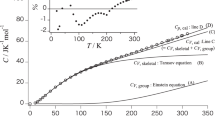
In exploring the analogy between crystalline polymers and metallic alloys it becomes necessary to allow for the differences in size and anisotropy of the crystallizing entity. As a consequence of the size and anisotropy of crystalline polymers their diffusion constant is several orders of magnitude smaller than metals. The anisotropy of the polymer molecule also gives rise to a tertiary or intramolecular nucleation process which does not occur in metals. The dispersion of molecular lengths which is common in high polymers makes it necessary to compare even the purest polymer with a multicomponent alloy. The combination of low diffusion constants and the tertiary nucleation process gives rise to a multitude of metastable equilibrium states as well as kinetically inhibited transitions which tend to complicate the phase diagrams. Nevertheless when all of these factors are considered it is possible to qualitatively account for much of the observed structural behavior of crystalline polyethylene by direct analogy to multicomponent metal alloys.
Similar content being viewed by others
Article PDF
References
F. Bueche, “Physical Properties of Polymers,” Interscience Publishers, New York, N.Y., 1962.
J. W. Rutter and B. Chalmers, Can. J. Phys., 31, 15 (1953).
H. D. Keith and F. J. Padden, J. Appl. Phys., 34, 2409 (1963).
H. D. Keith and F. J. Padden, J. Appl. Phys., 35, 1270 (1964).
H. G. Zachmann and P. Spellucci, Kolloid Z., 213, 39 (1966).
P. J. Floy and A. Vrij, J. Am. Chem. Soc., 85, 3548 (1963).
J. M. Peterson and P. H. Lindenmeyer, Makromol. Chem., 118, 343 (1968).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lindenmeyer, P. Polyethylene: A Multicomponent Alloy. Polym J 3, 507–516 (1972). https://doi.org/10.1295/polymj.3.507
Issue Date:
DOI: https://doi.org/10.1295/polymj.3.507