Abstract
Summary
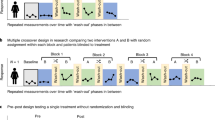
Translating the growing evidence base on COPD management into practice can be challenging and understanding the strengths and weakness of published studies is crucial. Studies should conform to the standards of CONSORT statement; they should be sufficiently powered, participants should be randomised, there should be assignment concealment, and the outcome measures and analyses should be decided in advance.
The interpretation of the results may be affected by age and severity inclusion criteria for the study and the exclusion of patients with co-morbid illnesses. Whether previous medication is continued or stopped can affect the interpretation of the results. Secondary analyses in sub-groups should be viewed with caution unless pre-specified and accommodated in the trial design and power calculations. Real world observational studies may be confounded by non-randomisation of participants but can sometimes yield valuable insights.
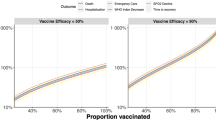
The way in which the results are presented can influence their interpretation and their magnitude with respect to minimal important differences as well as statistical significance is important.
Research studies help formulate management algorithms but often the questions they address are too specific to allow evidence-based sequencing of therapies.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Halpin has received sponsorship to attend international meetings, and honoraria for lecturing, attending advisory boards and preparing educational materials from, AstraZeneca, Boehringer Ingelheim, Chiesis, Nycomed, GlaxoSmithKline, and Pfizer. His department has received research funding from AstraZeneca.
Rights and permissions
About this article
Cite this article
Halpina, D. Lessons from the major studies in COPD: problems and pitfalls in translating research evidence into practice. Prim Care Respir J 19, 170–179 (2010). https://doi.org/10.4104/pcrj.2010.00015
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.4104/pcrj.2010.00015
This article is cited by
-
Exclusion rates in randomized controlled trials of treatments for physical conditions: a systematic review
Trials (2020)
-
Comparative Causal Analysis of the Effects of Long-Acting Muscarinic Antagonist Versus No Long-Acting Bronchodilator Use on Readmission or Mortality After Hospitalization for Chronic Obstructive Pulmonary Disease
Drugs - Real World Outcomes (2020)
-
Follow-up interviews from The Salford Lung Study (COPD) and analyses per treatment and exacerbations
npj Primary Care Respiratory Medicine (2019)
-
Eligibility of real-life patients with COPD for inclusion in trials of inhaled long-acting bronchodilator therapy
Respiratory Research (2016)
-
The prevalence of clinically-relevant comorbid conditions in patients with physician-diagnosed COPD: a cross-sectional study using data from NHANES 1999–2008
BMC Pulmonary Medicine (2012)