Abstract
Sgt1/Sugt1, a cochaperone of Hsp90, is involved in several cellular activities including Cullin E3 ubiqutin ligase activity. The high level of Sgt1 expression in colorectal and gastric tumors suggests that Sgt1 is involved in tumorigenesis. Here, we report that Sgt1 is overexpressed in colon, breast and lung tumor tissues and in Ewing sarcoma and rhabdomyosarcoma xenografts. We also found that Sgt1 heterozygous knockout resulted in suppressed Hras-mediated transformation in vitro and tumor formation in p53−/− mouse embryonic fibroblast cells and significantly increased survival of p53−/− mice. Moreover, depletion of Sgt1 inhibited the growth of Ewing sarcoma and rhabdomyosarcoma cells and destabilized EWS-FLI1 and PAX3-FOXO1 oncogenic fusion proteins, respectively, which are required for cellular growth. Our results suggest that Sgt1 contributes to cancer development by stabilizing oncoproteins and that Sgt1 is a potential therapeutic target.
Similar content being viewed by others
Introduction
Dysregulation of oncogenes and tumor suppressor genes is a common feature of different types of cancer. Because aberrant oncoproteins are unstable, cancer cells utilize Hsp90 as a chaperone to promote folding and function of mutated or overexpressed oncoproteins.1 The proliferation and survival of cancer cells can often be suppressed by the inhibition of one or more oncoproteins.2 Therefore, Hsp90 inhibitors have been developed and are under investigation as cancer therapy in clinical trials.1
Sgt1/Sugt1 (suppressor of G2 allele of skp1) is a highly conserved protein that functions as a cochaperone of Hsp90.3, 4, 5, 6, 7 Cochaperones, which interact with and are required for Hsp90 function, regulate the ATPase activity of Hsp90, recruit client proteins to Hsp90 and have been proposed as potential targets for cancer therapy.1 Presumably as a cochaperone, Sgt1 is involved in several specific cellular functions including ubiquitination,8 cyclic AMP pathway,9, 10 centrosome maturation,11 kinetochore assembly8, 12 and immune response.13, 14, 15, 16 Sgt1 and Hsp90 are required for kinetochore assembly because of their enhancement of the localization of kinetochore proteins.12, 17, 18 In addition, Sgt1 and Hsp90 participate in kinetochore-microtuble attachment by stabilizing the Mis12 complex at kinetochores.17, 19 Therefore, knockdown of Sgt1 expression induces misalignment of chromosomes, activation of a weakened spindle checkpoint and, possibly, the occurrence of aneuploidy.12, 17, 18, 19 In addition to these functions, Sgt1 and Hsp90 play a role in neuroblast cortical polarity through localization of Par and Pins complexes20 and in HGF-mediated epithelial morphogenesis through stabilization of Scribble.21
Overexpression of Sgt1 in tumor tissues has been reported. Sgt1 mRNA levels are elevated in colorectal cancer, and this increased expression is linked to an increased rate of recurrence and poorer prognosis.22 In gastric tumor cells, overexpression of Sgt1 upregulates Akt phosphorylation through the degradation of the phosphatase PHLPP1 by enhancing the interaction of PHLPP1 with SCF-β-TrCP.23 These findings suggest that overexpression of Sgt1 is involved in tumorigenesis. However, the function of Sgt1 in cancer development remains obscure.
Here, we report the findings of our investigation of Sgt1 protein levels in tumor tissues and pediatric tumor xenografts. Sgt1 was highly expressed in colon, breast and lung tumor tissues and in Ewing sarcoma and rhabdomyosarcoma xenografts. Reduction of Sgt1 altered the ability of mouse embryonic fibroblast (MEF) cells to undergo transformation and form tumors and the Sgt1 reduction resulted in increased survival of p53 knockout mice. These effects appeared to be independent of mitotic defects. Knockdown of Sgt1 expression inhibited the proliferation of cancer cells and destabilized oncoproteins that are required for the growth of Ewing sarcoma and rhabdomyosarcoma cells. Our results suggest that Sgt1 is involved in cancer development, possibly by stabilizing oncoproteins, and highlight Sgt1 as a potential therapeutic target.
Results
Sgt1 protein levels are elevated in tumor tissues and in Ewing sarcoma and rhabdomyosarcoma xenografts
To check protein levels of Sgt1 in tumor tissues, we performed immunoblotting analysis of tumor tissues (colon adenocarcinoma, breast ductal carcinoma, lung adenocarcinoma and lung squamous cell carcinoma) and normal adjacent tissues from the same patient. Levels of Sgt1 protein were greater in most tumor tissues than in normal adjacent tissues (Supplementary Figure S1). In additional immunoblotting studies, we also evaluated Sgt1 protein levels in 50 solid tumor xenografts obtained from the Pediatric Preclinical Testing Program (PPTP). Overexpression of Sgt1 protein was observed in xenografts, especially those of Ewing sarcoma and rhabdomyosarcoma (Figure 1).
Furthermore, the expression of Sgt1 protein was evaluated by immunohistochemistry of resected breast carcinoma, lung carcinoma, Ewing sarcoma and rhabdomyosarcoma specimens using an anti-Sgt1 antibody. Sgt1 appears to be more abundant in cancer tissues than in normal tissues (Supplementary Figure S5).
These results indicate that the Sgt1 protein is overexpressed in a broad range of tumor tissues.
Sgt1 heterozygous knockout decreases transformation and tumorigenicity of MEF cells
To investigate the role of Sgt1 in tumorigenesis, we first produced Sgt1 knockout mice by using the BayGenomics ES cell line RRS405. The gene trap vector was inserted into intron 2 of the Sgt1 gene (Supplementary Figure S2A), and this insertion resulted in the expression of a truncated Sgt1 protein fused with a β-geo marker. We crossed Sgt1 heterozygous knockout female and male mice; however, Sgt1 homozygous knockout mice were not obtained (Supplementary Figure S2B). Sgt1 homozygous knockout embryos at embryonic day (E) 3.5 were obtained by in vitro fertilization, whereas Sgt1 homozygous knockout embryos from E8.5 to E14.5 were not obtained (Supplementary Figure S2C). These results indicate that the truncated Sgt1 protein is not functional and that Sgt1 homozygous knockout results in early embryonic lethality in mice.
To evaluate the effect of decreased Sgt1 protein, we established Sgt1+/+ p53−/− and Sgt1+/− p53−/− MEF cells by crossing Sgt1 knockout and p53 knockout mice. Hras was expressed in these MEF cells as a result of retrovirus transduction, and focus formation assays were performed. Both types of cells that expressed Hras formed foci; however, the number of foci formed by Sgt1+/− p53−/− MEF cells was significantly less than that formed by Sgt1+/+ p53−/− MEF cells (Figure 2a). No proliferation defects in Sgt1+/− p53−/− MEF cells were observed (data not shown). To test the effect of Sgt1 reduction on tumorigenicity, we conducted an allograft experiment in which Hras-transduced Sgt1+/+ p53−/− and Sgt1+/− p53−/− MEF cells were injected into immunodeficient mice and tumor formation was monitored. In vivo tumor formation of Hras-transduced Sgt1+/− p53−/− MEF cells was significantly slower than that of Hras-transduced Sgt1+/+ p53−/− MEF cells (Figure 2b).
Sgt1 heterozygous knockout suppresses Hras-mediated transformation and tumorigenicity of p53−/− MEF cells and increases survival of p53−/− mice. (a) Sgt1+/+ p53−/− and Sgt1+/− p53−/− MEF cells transduced with Hras were used for focus-formation assays. After 2 weeks of incubation, cells were stained and the number of foci was counted. The experiments were repeated three times, and similar results were obtained. Average values±s.d. are shown (∗∗P<0.01; unpaired t test). (b) Hras-transduced MEF cells were injected into immunodeficient CB17SC-F scid−/− female mice. Tumor volumes were measured at indicated time points. Average values±s.d. are shown (∗∗∗∗P<0.0001; unpaired t-test). Mice in the blue curve were killed owing to dehydration on the twenty-first day or the twenty-fourth day. (c) The survival estimates of Sgt1+/+ and Sgt1+/− mice are shown. (d) The survival curve of Sgt1+/+ p53−/− and Sgt1+/− p53−/− mice are shown (P<0.05; log-rank test).
Sgt1 heterozygous knockout increases the survival of p53−/− mice
To test the effect of Sgt1 heterozygous knockout in vivo, the survival of Sgt1+/+ and Sgt1+/− mice was monitored. No significant difference in the survival between Sgt1+/+ and Sgt1+/− mice was observed (Figure 2c). Sgt1+/− mice and Sgt1+/− MEF cells did not show any obvious mutant phenotypes (data not shown), a result that indicated that one copy of Sgt1 is sufficient for viability.
To analyze the in vivo effect of the Sgt1 heterozygous knockout in mice that are deficient in a tumor suppressor gene, Sgt1+/+ p53−/− and Sgt1+/− p53−/− mice were produced, and the survival of these mice were monitored. Sgt1 heterozygous knockout resulted in significantly longer survival of p53−/− mice than Sgt1 wild-type mice (Figure 2d).
Taken together, our results indicate that Sgt1 reduction suppresses Hras-mediated transformation and tumorigenicity of p53−/− MEF cells, and this suppression may result in the prolonged survival of p53−/− mice.
Kinetochore formation, spindle checkpoint and ploidy are normal in Sgt1+/− MEF cells
Aneuploidy (that is, the state of having an abnormal number of chromosomes) that results from CENP-E (a kinetochore motor protein) heterozygous knockout appears to inhibit tumorigenesis in p19/ARF−/− mice.24, 25 Sgt1 depletion by short interfering RNA (siRNA) in HeLa cells causes delocalization of kinetochore proteins and activation of the weakened spindle checkpoint, which may result in aneuploidy.12, 17, 19 Therefore, we hypothesized that aneuploidy is the mechanism of tumor suppression in Sgt1 heterozygous knockout mice. Our phenotype analysis of MEF cells found that the level of Sgt1 protein in Sgt1+/− MEF cells was reduced to about 30% of the level in Sgt1+/+ MEF cells (Supplementary Figure S3A). Indirect immunofluorescence microscopy of kinetochore localization showed that CENP-H signals at kinetochores in Sgt1+/− MEF cells were indistinguishable from those in Sgt1+/+ MEF cells (Supplementary Figure S3B). Also, the mitotic index in response to paclitaxel was indistinguishable between Sgt1+/+ p53−/− MEF cells and Sgt1+/− p53−/− MEF cells (Supplementary Figure S4A). Furthermore, chromosome numbers were similar between Sgt1+/+ MEF cells and Sgt1+/− MEF cells (Supplementary Figure S3C). These results indicate that kinetochores are assembled properly and that the spindle checkpoint is normal in Sgt1+/− MEF cells and that there is no difference in ploidy between Sgt1+/+ MEF cells and Sgt1+/− MEF cells. Therefore, we concluded that aneuploidy does not seem to be the mechanism of the tumor suppression in Sgt1+/− mice.
Sgt1 heterozygous knockout does not increase senescent and apoptotic cells
Cellular senescence and apoptosis are major mechanisms of tumor suppression.26 Cellular senescence was observed in Skp2-deficient MEF cells in an ARF-p53-independent manner.27 Sgt1 interacts with Skp1,8, 18 which interacts with Skp2.28 Our analysis found no significant difference in HRas-induced senescence and apoptosis between Sgt1+/− p53−/− MEF cells and Sgt1+/+ p53−/− MEF cells (Supplementary Figures S4B and S4C). These results suggest that senescence and apoptosis are not the mechanism of tumor suppression.
Knockdown of Sgt1 expression inhibits the proliferation of cancer cells
Because the reduced expression of Sgt1 protein that resulted from Sgt1 heterozygous knockout suppressed Hras-mediated transformation and tumorigenicity of p53−/− MEF cells and prolonged the survival of p53−/− mice (Figure 2), we hypothesized that Sgt1 is a potential target for cancer therapy. To test this hypothesis, we examined whether depletion of Sgt1 affects the proliferation of Sgt1-overexpressing cancer cells (that is, Ewing sarcoma cell lines EW8 and TC-71 and rhabdomyosarcoma cell line Rh41). To knock down Sgt1 expression, we incubated cancer cells transfected with two independent siRNAs against Sgt112, 18 (Figure 4a and Supplementary Figure S4D) and recorded the extent of cell growth at 4-h intervals. Knockdown of Sgt1 expression significantly inhibited the proliferation of cells when compared with that of cells subjected to a luciferase control (Figure 3).
Knockdown of Sgt1 expression inhibits the growth of cancer cells. (a) EW8, TC-71, Rh41, HCT 116, MDA-MB-231 and HCC1806 cells were transfected with luciferase siRNA (Luc) and two independent Sgt1 siRNAs (Sgt1-1 and Sgt1-2). Proliferation of the cells was monitored at 4-h intervals, and data were analyzed by IncuCyte software. The experiments were repeated three times, and similar results were obtained. Average values±s.d. are shown. (b) Representative images of cells transfected with siRNAs are shown. Bars represent a distance of 100 μm.
Sgt1 and Hsp90 stabilize the oncoproteins EWS-FLI1 and PAX3-FOXO1
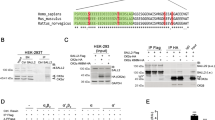
Sgt1 interacts with Hsp90 and functions as a cochaperone of Hsp90.3, 6, 11, 17, 21 Sgt1 is involved in stability of Polo, Mis12 complex and Scribble that are required for centrosome maturation, proper kinetochore assembly or HGF-mediated epithelial morphogenesis, respectively.11, 17, 21 Therefore, we hypothesized that Sgt1 may be involved in stability of the oncofusion proteins that are essential for the proliferation of cancer cells. To test this hypothesis, we evaluated the levels of the oncogenic fusion proteins EWS-FLI1 and PAX3-FOXO1, which are required for the proliferation of Ewing sarcoma and alveolar rhabdomyosarcoma cell lines, respectively.29, 30 Protein levels of EWS-FLI in EW8 cells and PAX3-FOXO1 in Rh41 cells were reduced by Sgt1 depletion or ganetespib (an Hsp90 inhibitor) treatment (Figures 4a and b).
EWS-FLI1 and PAX3-FOXO1 oncoproteins were destabilized after knockdown of Sgt1 expression or inhibition of Hsp90. (a and b) Indicated proteins in EW8 or Rh41 were detected by immunoblotting at 72 h after siRNA treatment (a) or at 24 h after ganetespib treatment (b). Levels of EWS-FLI1, PAX3-FOXO1 and Sgt1 were normalized to β-tubulin, and the protein level of control cells was established as a value of 1.
These may imply that Sgt1 and Hsp90 are required for the proliferation of cancer cells by stabilizing proteins essential to that growth.
Discussion
In our current study and in previous reports, overexpression of Sgt1 in tumor tissues and xenografts was observed.22, 23 Sgt1 heterozygous knockout decreased Hras-mediated transformation and tumorigenicity of p53−/− MEF cells and extended the survival period of p53−/− mice. These results prompted us to hypothesize that Sgt1 is an oncogene. To test this hypothesis, we conducted focus-formation assays using p53−/− and ARF−/− MEF cells that overexpressed mouse Sgt1 and wild-type MEF cells that coexpressed Sgt1 and the immortalizing oncogene c-Myc. However, the results under these conditions were negative (data not shown). Next, we tested the effect of overexpression of Sgt1 on Hras-mediated transformation of MEF cells in focus-formation assays. Overexpression of Sgt1 and Hras in p53−/− MEF cells had no effect on Hras-mediated transformation (data not shown). These findings suggest that Sgt1 may not be an authentic oncogene. Like Sgt1, Cdc37 is a co-chaperone of Hsp90, is overexpressed in different types of cancer cells, and is thought to be an oncogene.31 Although overexpression of Cdc37 in mice causes tumors after long latency,32 there has been no report that describes cellular transformation induced by Cdc37 overexpression. It will be interesting to test whether overexpression of Sgt1 affects the long latency of tumors in Sgt1 transgenic mice.
What is the molecular mechanism of tumor suppression caused by the Sgt1 heterozygous knockout? The results of experiments described in this article indicate that mitotic defects, senescence, and apoptosis are not the mechanisms. Recently, Gao et al.23 reported that overexpression of Sgt1 upregulates Akt phosphorylation through enhancement of SCF-β-TrCP-dependent degradation of the phosphatase PHLPP1 in gastric cancer cells. However, we found no difference in Akt phosphorylation and PHLPP1 protein levels among p53−/− Sgt1+/−, p53−/− Sgt1+/+ or Sgt1-overexpressing MEF cells (data not shown). In light of our finding that levels of the EWS-FLI1 protein in EW8 cells or PAX3-FOXO1 protein in Rh41 cells were decreased by the knockdown of Sgt1 expression, it is plausible that destabilization of the onco-proteins is a mechanism of tumor suppression that results from the Sgt1 heterozygous knockout. Such a destabilization mechanism functions in Sgt1-depleted cancer cells.
An Hsp90 cochaperone that is overexpressed in cancers is a potential target for cancer therapy, because Hsp90 requires cochaperones to function.33 Knockdown of Cdc37 expression inhibits the growth of cancer cells and xenografts.34 Given that Sgt1 is a cochaperone of Hsp90, that Sgt1 is overexpressed in tumor cells and that decreased Sgt1 protein suppresses cellular transformation and allograft growth, we believe that Sgt1 could be a potential target for cancer therapy. Consistent with this possibility is the previous finding that knockdown of Sgt1 expression inhibits the growth of cancer cells.23 Sgt1 plays a role in various cellular functions by stabilizing the proteins together with Hsp90. A mutation in Sgt1 decreases levels of Polo.11 Knockdown of Sgt1 or Hsp90 expression or inhibition of Hsp90 by 17-AAG decreases levels of the Mis12 complex17 or Scribble.21 Reduction of these proteins causes defects in centrosome maturation, kinetochore formation and epithelial morphogenesis. Therefore, it is possible that Sgt1 is involved in cancer development by stabilizing proteins that are required for the growth of cancer cells. The oncogenic fusion proteins EWS-FLI1 and PAX3-FOXO1 are required for the growth of Ewing sarcoma and rhabdomyosarcoma cells, respectively.29, 30 Levels of the EWS-FLI1 protein in EW8 cells or the PAX3-FOXO1 protein in Rh41 cells were decreased by knockdown of Sgt1 expression. These results corroborate the potential of Sgt1 for cancer therapy. Further study is needed to identify client proteins of Sgt1 in other types of cancer cells.
Materials and methods
Reagents
The following antibodies were used in our experiments: mouse anti-Sgt1 (BD Biosciences, San Jose, CA, USA), rabbit anti-GAPDH (Abcam, Cambridge, MA, USA), rabbit anti-Fli1, rabbit anti-FKHR (Santa Cruz Biotechnology, Dallas, TX, USA), mouse anti-β-tubulin (MP Biomedicals, Santa Ana, CA, USA), mouse anti-CENP-H (BD Biosciences), human anti-centromere autoimmune serum (a generous gift from Dr. William R. Brinkley, Baylor College of Medicine), rabbit anti-phospho-histone H3 (Ser10) (EMD Millipore, Billerica, MA, USA), goat anti-mouse IgG-HRP, goat anti-rabbit IgG-HRP (Santa Cruz Biotechnology), IRDye 680RD goat anti-mouse IgG (H+L), IRDye 800CW goat anti-mouse IgG (H+L), IRDye 800CW goat anti-rabbit IgG (H+L) (LI-COR), Alexa Fluor 488 goat anti-mouse IgG (H+L), Alexa Fluor 488 goat anti-rabbit IgG (H+L) and Alexa Fluor 594 goat anti-human IgG (H+L) (Life Technologies, Grand Island, NY, USA).
The ready-to-use membranes purchased from Novus Biologicals (Littleton, CO, USA) were INSTA-Blot Male Lung Tissue Oncopair (catalog number IMB-127a), INSTA-Blot Female Lung Tissue Oncopair (catalog number IMB-128a), INSTA-Blot Breast Tissue Oncopair (catalog number IMB-130a), INSTA-Blot Breast Tissue Oncopair (catalog number IMB-130e) and INSTA-Blot Colon Tissue Oncopair (catalog number IMB-131a).
Immunoblotting was performed by standard procedure, and immunolabeled proteins were detected by ECL Plus Western Blotting Detection Reagents (GE-Healthcare Life Sciences, Pittsburgh, PA, USA) or ODYSSEY CLx (LI-COR, Lincoln, NE, USA).
The siRNAs against luciferase and Sgt1 were previously described.11, 18 Luciferase siRNA (5′-CUUACGCUGAGUACUUCGATT), Sgt1-1 siRNA (5′-GCUAGAGGGGCAAGGAGAUTT) and Sgt1-2 siRNA (5′-AAGGCUUUGGAACAGAAACCA) were synthesized by the Hartwell Center for Bioinformatics and Biotechnology at St. Jude Children’s Research Hospital.
Cell lines, primary cells and cell culture
HCT 116, MDA-MB-231 and HCC1806 cells were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA). EW8, Rh41 and HCC1806 cells were maintained in RPMI 1640 (Lonza, Portsmouth, NH, USA) supplemented with 10% fetal bovine serum (FBS; Life Technologies), TC-71 was maintained in IMDM (Lonza) supplemented with 10% FBS and ITS (Life Technologies), HCT 116 cells were maintained in McCoy’s 5A (ATCC) supplemented with 10% FBS, and MDA-MB-231 cells were maintained in DMEM (Lonza) supplemented with 10% FBS at 37 °C with 5% CO2.
Primary MEF cells were derived from E13.5 or E14.5 embryos. The p53−/− and ARF−/− MEF cells are generous gifts from Dr. Martine F. Roussel (St. Jude Children’s Research Hospital). MEF cells were maintained in DMEM (Lonza) that contained 10% FBS, 1 × MEM non-essential amino acid, 55 μM 2-mercaptoethanol and 10 μg/ml gentamicin (Life Technologies) at 37 °C with 5% CO2. Early passage (P2-P5) MEF cells were used for the experiments. Exceptions were p53−/− and ARF−/− MEF cells.
Retrovirus production and focus-formation assay
Plat-E cells (Cell Biolabs, San Diego, CA, USA) were used to produce retrovirus according to the manufacturer’s instructions. Plat-E cells were transfected with pBABE-puro and pBABE-puro Hras V12 (a generous gift from Martine F. Roussel/Scott Lowe) by using FuGENE 6 (Promega, Fitchburg, WI, USA). Retroviral supernatant was filtered and mixed with 4 μg/ml polybrene (EMD Millipore). MEF cells were infected with retroviral supernatant twice and selected in the presence of 2 μg/ml of puromycin (Sigma-Aldrich, St. Louis, MO, USA) for 4 days.
Focus-formation assays were performed as previously described.35 Briefly, 103 MEF cells infected with retrovirus were mixed with 3 × 105 uninfected MEF cells, and the mixture was cultured in a 100-mm dish in triplicate. Medium was changed every 2 or 3 days. After 2 weeks incubation, foci were stained with giemsa stain (Sigma-Aldrich), and the number of foci was counted.
Mouse strains
BayGenomics ES cell line RRS405 (129P2/OlaHsd) was purchased from Mutant Mouse Regional Resource Centers (http://www.mmrrc.org/index.php). This ES cell line was used to produce Sgt1 knockout mice by standard procedures at the St. Jude Transgenic Core Facility. The p53 knockout mice (C57BL/6J) were purchased from The Jackson Laboratory (stock number 002101) (Bar Harbor, ME, USA). All mice have mixed 129P2/OlaHsd and C57BL/6J genetic background. All mice, including those used in xenograft and allograft experiments, were maintained under barrier conditions, and experiments were conducted according to protocols and conditions approved by the institutional animal care and use committee.
Solid tumor xenograft and allograft
Immunodeficient CB17SC-F scid−/− female mice (Taconic Farms, Germantown, NY, USA)36 were used to propagate subcutaneously implanted tumors. Tumors were excised, rapidly frozen in liquid nitrogen and pulverized under liquid nitrogen. Total proteins were extracted with cell lysis buffer (Cell Signaling Technology, Danvers, MA, USA) supplemented with protease inhibitors and protein phosphatase inhibitors (Roche, Indianapolis, IN, USA). Thirty-five micrograms of total proteins were used in immunoblotting experiments to detect Sgt1 and GAPDH.
The tumorigenic potential of MEF cells was evaluated by injecting 106 cells (volume, 0.1 ml) into the left flank of immunodeficient CB17SC-F scid−/− female mice. Tumor volume (cm3) was measured with calipers and determined as previously described.37 Tumor volume was measured biweekly at the initial observation of the tumor growth until endpoint criteria were achieved. Tumor volume was calculated according to the following formula: (π/6) · d3, where d represents the mean diameter.37 Mice were humanely killed at the endpoint. The endpoint criteria met by this study were follows: a tumor volume of 2.24 cm3, ulceration of the tumor and dehydration or poor condition of the animal.
Transfection and proliferation assay
Cells underwent reverse transfection with 50 nM siRNA by using Lipofectamine RNAiMAX Transfection Reagent (Life Technologies) according to the manufacturer’s instruction. For immunoblotting, cells were cultured in a 6-well plate. For the proliferation assay, cells were cultured in triplicate in a 96-well plate and incubated in IncuCyte (Essen BioScience, Ann Arbor, MI, USA). Proliferation of cells was monitored at 4-h intervals, and confluence (%) was calculated by IncuCyte software.
Immunofluorescence, chromosome spreads, senescence assay and apoptosis assay
Indirect immunofluorescence and quantitation of kinetochore signals were performed as previously described.38 Chromosome spreads were achieved as previously described.25 Senescence assays were performed as previously described.39 Briefly, 104 MEF cells were cultured in a six-well plate in triplicate for 4 days. Cells were fixed with 2% formaldehyde (Fisher Scientific, Pittsburgh, PA, USA) and 0.2% glutaraldehyde (Sigma-Aldrich) in phosphate-buffered saline at room temperature for 5 min. SA-β-gal activity was quantified by counting 200 cells per well. For apoptosis assays, 1.3 × 105 MEF cells were cultured in a 6-well plate in duplicate for 2 days. Cells were harvested and stained with annexin V-FITC and propidium iodide by using the Annexin V-FITC Apoptosis Detection Kit (Abcam). FACS analysis was performed with the BD LSR II (BD Biosciences).
Clinical samples
The tissues from patients with breast cancer, lung cancer, Ewing sarcoma and rhabdomyosarcoma were obtained from Cooperative Human Tissue Network, University of Pennsylvania, Philadelphia, PA, USA and Nationwide Children’s Hospital.
Immunohistochemistry of Sgt1
Immunohistochemistry study to determine the distribution of Sgt1/Sugt1 protein in tissue sections from patients with breast cancer, lung cancer, Ewing sarcoma and rhabdomyosarcoma were performed as described previously with a minor modification.22 Briefly, deparaffinized tissue sections were heat-retrieved in 0.01M citrate buffer and immunohistochemically stained with the primary rabbit polyclonal antibodies against Sgt1/Sugt1 (Protein Tech Group Inc., Chicago, IL, USA) at a dilution of 1:250–1:500. Detection reagents were MACH 2 rabbit HRP polymer (Biocare Medical, Concord, CA, USA) followed by Liquid DAB Substrate and Chromagen system (DAKO, Carpinteria, CA, USA). The sections were counterstained with Mayer’s hematoxylin. The images of the stained tissues were acquired on a Zeiss AxioScope microscope system (Zeiss, Dublin, CA, USA) equipped with Plan-NEOFLUAR objective lenses and the AxioCam HRc high resolution digital camera (Zeiss) using AxioVision software (Zeiss).
References
Trepel J, Mollapour M, Giaccone G, Neckers L . Targeting the dynamic HSP90 complex in cancer. Nat Rev Cancer 2010; 10: 537–549.
Weinstein IB, Joe A . Oncogene addiction. Cancer Res 2008; 68: 3077–3080 discussion 3080.
Bansal PK, Abdulle R, Kitagawa K . Sgt1 associates with Hsp90: an initial step of assembly of the core kinetochore complex. Mol Cell Biol 2004; 24: 8069–8079.
Hubert DA, Tornero P, Belkhadir Y, Krishna P, Takahashi A, Shirasu K et al. Cytosolic HSP90 associates with and modulates the Arabidopsis RPM1 disease resistance protein. EMBO J 2003; 22: 5679–5689.
Lee YT, Jacob J, Michowski W, Nowotny M, Kuznicki J, Chazin WJ . Human Sgt1 binds HSP90 through the CHORD-Sgt1 domain and not the tetratricopeptide repeat domain. J Biol Chem 2004; 279: 16511–16517.
Lingelbach LB, Kaplan KB . The interaction between Sgt1p and Skp1p is regulated by HSP90 chaperones and is required for proper CBF3 assembly. Mol Cell Biol 2004; 24: 8938–8950.
Takahashi A, Casais C, Ichimura K, Shirasu K . HSP90 interacts with RAR1 and SGT1 and is essential for RPS2-mediated disease resistance in Arabidopsis. Proc Natl Acad Sci USA 2003; 100: 11777–11782.
Kitagawa K, Skowyra D, Elledge SJ, Harper JW, Hieter P . SGT1 encodes an essential component of the yeast kinetochore assembly pathway and a novel subunit of the SCF ubiquitin ligase complex. Mol Cell 1999; 4: 21–33.
Dubacq C, Guerois R, Courbeyrette R, Kitagawa K, Mann C . Sgt1p contributes to cyclic AMP pathway activity and physically interacts with the adenylyl cyclase Cyr1p/Cdc35p in budding yeast. Eukaryot Cell 2002; 1: 568–582.
Schadick K, Fourcade HM, Boumenot P, Seitz JJ, Morrell JL, Chang L et al. Schizosaccharomyces pombe Git7p, a member of the Saccharomyces cerevisiae Sgtlp family, is required for glucose and cyclic AMP signaling, cell wall integrity, and septation. Eukaryot Cell 2002; 1: 558–567.
Martins T, Maia AF, Steffensen S, Sunkel CE . Sgt1 a co-chaperone of Hsp90 stabilizes Polo and is required for centrosome organization. EMBO J 2009; 28: 234–247.
Steensgaard P, Garre M, Muradore I, Transidico P, Nigg EA, Kitagawa K et al. Sgt1 is required for human kinetochore assembly. EMBO Rep 2004; 5: 626–631.
Austin MJ, Muskett P, Kahn K, Feys BJ, Jones JD, Parker JE . Regulatory role of SGT1 in early R gene-mediated plant defenses. Science 2002; 295: 2077–2080.
Azevedo C, Sadanandom A, Kitagawa K, Freialdenhoven A, Shirasu K, Schulze-Lefert P . The RAR1 interactor SGT1, an essential component of R gene-triggered disease resistance. Science 2002; 295: 2073–2076.
da Silva Correia J, Miranda Y, Leonard N, Ulevitch R . SGT1 is essential for Nod1 activation. Proc Natl Acad Sci USA 2007; 104: 6764–6769.
Mayor A, Martinon F, De Smedt T, Petrilli V, Tschopp J . A crucial function of SGT1 and HSP90 in inflammasome activity links mammalian and plant innate immune responses. Nat Immunol 2007; 8: 497–503.
Davies AE, Kaplan KB . Hsp90-Sgt1 and Skp1 target human Mis12 complexes to ensure efficient formation of kinetochore-microtubule binding sites. J Cell Biol 2010; 189: 261–274.
Niikura Y, Ohta S, Vandenbeldt KJ, Abdulle R, McEwen BF, Kitagawa K . 17-AAG, an Hsp90 inhibitor, causes kinetochore defects: a novel mechanism by which 17-AAG inhibits cell proliferation. Oncogene 2006; 25: 4133–4146.
Liu XS, Song B, Tang J, Liu W, Kuang S, Liu X . Plk1 phosphorylates Sgt1 at the kinetochores to promote timely kinetochore-microtubule attachment. Mol Cell Biol 2012; 32: 4053–4067.
Andersen RO, Turnbull DW, Johnson EA, Doe CQ . Sgt1 acts via an LKB1/AMPK pathway to establish cortical polarity in larval neuroblasts. Dev Biol 2012; 363: 258–265.
Eastburn DJ, Zegers MM, Mostov KE . Scrib regulates HGF-mediated epithelial morphogenesis and is stabilized by Sgt1-HSP90. J Cell Sci 2012; 125: 4147–4157.
Iwatsuki M, Mimori K, Sato T, Toh H, Yokobori T, Tanaka F et al. Overexpression of SUGT1 in human colorectal cancer and its clinicopathological significance. Int J Oncol 2010; 36: 569–575.
Gao G, Kun T, Sheng Y, Qian M, Kong F, Liu X et al. SGT1 regulates Akt signaling by promoting beta-TrCP-dependent PHLPP1 degradation in gastric cancer cells. Mol Biol Rep 2013; 40: 2947–2953.
Silk AD, Zasadil LM, Holland AJ, Vitre B, Cleveland DW, Weaver BA . Chromosome missegregation rate predicts whether aneuploidy will promote or suppress tumors. Proc Natl Acad Sci USA 2013; 110: E4134–E4141.
Weaver BA, Silk AD, Montagna C, Verdier-Pinard P, Cleveland DW . Aneuploidy acts both oncogenically and as a tumor suppressor. Cancer Cell 2007; 11: 25–36.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Lin HK, Chen Z, Wang G, Nardella C, Lee SW, Chan CH et al. Skp2 targeting suppresses tumorigenesis by Arf-p53-independent cellular senescence. Nature 2010; 464: 374–379.
Zhang H, Kobayashi R, Galaktionov K, Beach D . p19Skp1 and p45Skp2 are essential elements of the cyclin A-CDK2 S phase kinase. Cell 1995; 82: 915–925.
Dohjima T, Lee NS, Li H, Ohno T, Rossi JJ . Small interfering RNAs expressed from a Pol III promoter suppress the EWS/Fli-1 transcript in an Ewing sarcoma cell line. Mol Ther 2003; 7: 811–816.
Kikuchi K, Tsuchiya K, Otabe O, Gotoh T, Tamura S, Katsumi Y et al. Effects of PAX3-FKHR on malignant phenotypes in alveolar rhabdomyosarcoma. Biochem Biophys Res Commun 2008; 365: 568–574.
Gray PJ Jr ., Prince T, Cheng J, Stevenson MA, Calderwood SK . Targeting the oncogene and kinome chaperone CDC37. Nat Rev Cancer 2008; 8: 491–495.
Stepanova L, Finegold M, DeMayo F, Schmidt EV, Harper JW . The oncoprotein kinase chaperone CDC37 functions as an oncogene in mice and collaborates with both c-myc and cyclin D1 in transformation of multiple tissues. Mol Cell Biol 2000; 20: 4462–4473.
Calderwood SK . Molecular cochaperones: tumor growth and cancer treatment. Scientifica (Cairo) 2013; 2013: 217513.
Marino-Enriquez A, Ou WB, Cowley G, Luo B, Jonker AH, Mayeda M et al. Genome-wide functional screening identifies CDC37 as a crucial HSP90-cofactor for KIT oncogenic expression in gastrointestinal stromal tumors. Oncogene 2013; 33: 1872–1876.
Sun H, Taneja R . Analysis of transformation and tumorigenicity using mouse embryonic fibroblast cells. Methods Mol Biol 2007; 383: 303–310.
Houghton PJ, Morton CL, Tucker C, Payne D, Favours E, Cole C et al. The pediatric preclinical testing program: description of models and early testing results. Pediatr Blood Cancer 2007; 49: 928–940.
Morton CL, Houghton PJ . Establishment of human tumor xenografts in immunodeficient mice. Nat Protoc 2007; 2: 247–250.
Niikura Y, Ogi H, Kikuchi K, Kitagawa K . BUB3 that dissociates from BUB1 activates caspase-independent mitotic death (CIMD). Cell Death Differ 2010; 17: 1011–1024.
Itahana K, Campisi J, Dimri GP . Methods to detect biomarkers of cellular senescence: the senescence-associated beta-galactosidase assay. Methods Mol Biol 2007; 371: 21–31.
Acknowledgements
We thank G Grosveld for mouse production; J Swift for his help in performing in vitro fertilization experiments; A Inoue for his help in collecting embryos; and WR Brinkley, MF Roussel and S Lowe for their generous gifts of reagents. Financial Support: CA165995 from the National Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogenesis website .
Supplementary information
Rights and permissions
Oncogenesis is an open-access journal published by Nature Publishing Group. This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ogi, H., Sakuraba, Y., Kitagawa, R. et al. The oncogenic role of the cochaperone Sgt1. Oncogenesis 4, e149 (2015). https://doi.org/10.1038/oncsis.2015.12
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/oncsis.2015.12