Abstract
Autophagy is a pathophysiological phenomenon in liver cirrhosis that can further progress into hepatocarcinoma. Liver cancer stem cells (CSCs) are believed to initiate hepatocarcinogenesis. To investigate the precise mechanism related to the origin of CSCs in liver cirrhosis and hepatocarcinogenesis, we labeled Axin2+ hepatic cells with EGFP in Axin2Cre;Rosa26EGFP transgenic rats, and then stratified clinical and rat liver cirrhosis samples by autophagy flux. Clinical follow-up and lineage tracing in transgenic rat liver cirrhosis revealed that while Axin2/EGFP+ hepatic cells were present in normal livers and cirrhotic livers without aberrant autophagy, hepatic Axin2/EGFP+CD90+ cells were generated exclusively in cirrhotic livers with aberrant autophagy and promoted hepatocarcinogenesis. Aberrant autophagy in liver cirrhosis resulted in hepatocyte growth factor (HGF) expression, leading to activation of Met/JNK and Met/STAT3 signaling in sorted hepatic Axin2/EGFP+ cells and their transition into Axin2/EGFP+CD90+ cells that possess CSC properties. In a transgenic rat liver cirrhosis model, induction or inhibition of autophagy in cirrhotic livers by systemic administration of rapamycin or chloroquine or transfection with Atg3- and Atg7-shRNAs significantly induced or suppressed HGF expression, which in turn increased or reduced generation of EGFP+CD90+ hepatic cells by activating or inactivating Met/JNK and Met/STAT3 signaling, thereby promoting or preventing hepatocarcinogenesis. Systemic treatment with HGF-shRNA, SP600125 or stattic also reduced generation of EGFP(Axin2)+ hepatic cell-originated CD90+ CSCs in aberrant autophagic cirrhotic livers by inactivating HGF/Met/JNK or HGF/Met/STAT3 signaling, further preventing hepatocarcinogenesis. These data suggest that activation of Met/JNK and Met/STAT3 signaling in Axin2+ hepatic cells via autophagy-dependent HGF expression and the resultant generation of Axin2+CD90+ CSCs is a major mechanism of hepatocarcinogenesis in cirrhotic livers.
Similar content being viewed by others
Introduction
Alcoholic liver disease and chronic hepatitis can progress into liver cirrhosis and then further develop into liver cancer, which is a natural process of hepatocarcinogenesis. However, the cellular and molecular mechanisms underlying the progression of liver cirrhosis to liver cancer are poorly understood.
It is well known that liver cancer stem-like cells (CSCs) have both self-renewal and tumorigenesis capacities and play a pivotal role in hepatocarcinogenesis.1, 2, 3 Although liver CSCs have been successfully isolated from human hepatocarcinoma cell lines and cancer tissues,4, 5 little is known about the origin of liver CSCs during the development and progression of liver cirrhosis into hepatocarcinoma. Axin2 is a target gene and also a negative regulator of Wnt/β-catenin signaling.6 The Wnt/Axin2 signaling cascade predominantly participates in maintaining self-renewal of normal stem cells and proliferation or differentiation of progenitor cells.7, 8, 9, 10 A recent study indicates that Wnt signals-maintained hepatic Axin2+ cells have the capacity to self-renewal and play a role of liver stem cells.11 Epigenetic dysregulation or mutation of Axin2 would promote or maintain cancer stem cell-like traits in lung cancer,12 ovarian cancer,13 osteosarcoma14 and liver cancer.15 Consequently, we hypothesized that hepatic Axin2+ cells might be responsible for the development of liver CSCs during the progression of liver cirrhosis to hepatocarcinoma.
Autophagy is an evolutionarily conserved physiological process in cell, generating intracellular nutrients, growth factors and energy to support cell survival and cellular activities during stress, such as nutrition deprivation, hypoxia or ischemia.16, 17 Such cytokines or growth factors can independently activate endogenous signals to stimulate cell reproduction and proliferation or even to promote cell stemness.18, 19 Because of the pathological changes, such as fibrosis, pseudolobar formation, reconstruction of the portal area and portal vein occlusion in liver cirrhosis, hepatocytes continuously undergo nutritional deprivation, hypoxia and ischemia,20 which may lead to aberrant autophagy. Furthermore, previous studies have shown that aberrant autophagy promotes the survival of liver CSCs in vitro.21 However, the pathophysiological role that autophagy may play in the generation of liver CSCs during the progression of liver cirrhosis to hepatocarcinoma and the responsible mechanism have not yet been investigated.
Recently, CD90 was confirmed to be a more stable marker for liver CSCs.3, 22 In this study, we identified hepatic Axin2+CD90+ cells as CSCs in liver cirrhosis, and indicated that autophagy-dependent hepatocyte growth factor (HGF)/Met/JNK and HGF/Met/STAT3 signaling activation was required for the generation of Axin2+CD90+ cells from Axin2+ cells during the progression of liver cirrhosis to hepatocarcinoma.
Results
Aberrant autophagy is associated with the transition of Axin2+ cells into Axin2+CD90+ cells in liver cirrhosis to hepatocarcinogenesis
To determine the role of autophagy in the generation of Axin2+CD90+ cells in liver cirrhosis and the further development of hepatocarcinoma, human non-cirrhotic, alcohol-related, hepatitis B virus-related and hepatitis C virus-related cirrhotic liver samples were stratified according to the autophagy status, which was determined by western blot analysis of LC3-I to LC3-II conversion and P62 downregulation (Figure 1a and Supplementary Figure SIA). Given that CD90 is a stable marker for liver CSCs,3, 22 the exhibition of hepatic Axin2+CD90+ cells in the above samples was further detected by double immunofluorescence staining. Figure 1b and Supplementary Figure SIIA show that hepatic Axin2+CD90+ cells were present in cirrhotic liver samples with aberrant autophagy, whereas other samples without aberrant autophagy only exhibited Axin2+ hepatic cells. Flow cytometry analysis also revealed higher percentages of hepatic Axin2+CD90+ cells in cirrhotic liver samples with aberrant autophagy than those in samples without aberrant autophagy (Figure 1c).
Transition of Axin2+ cells into Axin2+CD90+ cells during the progression of liver cirrhosis to hepatocarcinoma is associated with aberrant autophagy. Axin2/EGFP+CD90+ cells present in human and rat cirrhotic livers with aberrant autophagy that further progressed into hepatocarcinoma that maintained Axin2/EGFP+CD90+ cells. Four weeks after the last diethylinitrosamine injection, the biopsied Axin2Cre;Rosa26EGFP rat liver samples (n=4) and the biopsied human healthy livers (n=8) and alcohol- (n=6), hepatitis B virus- (n=10) and hepatitis C virus-related liver cirrhosis (n=6) samples were stratified according to (a) western blot analysis of the autophagy influx. These normal and cirrhotic liver tissues from human (left panel) and Axin2Cre;Rosa26EGFP rats (right panel) were then subjected to (b) double immunofluorescence staining and (c) flow cytometry to detect and quantify hepatic Axin2+CD90+ or EGFP+CD90+ cells. The dashed squares indicate higher magnification images of merged Axin2/EGFP+CD90+ cells in the lower right corner. Scale bar: 50 μm. The percentages of double-positive cells are shown in the upper right quadrants of the representative flow cytometry graphs. (d) After an 8-year follow-up and 8 weeks of additional observation, double immunofluorescence staining for hepatic Axin2/EGFP+CD90+ cells was performed in human (left panel, n=5) and rat (right panel, n=3) hepatocarcinoma tissues that progressed from liver cirrhosis with aberrant autophagy. The dashed squares indicate higher magnification images of the merged Axin2/EGFP+CD90+ cells in the lower right corner. Scale bar: 50 μm. All data are representative images from three separate experiments.
More importantly, in contrast to liver cirrhosis without aberrant autophagy and hepatic Axin2+CD90+ cells, 3/4 individuals with alcoholic liver cirrhosis, 8/10 with hepatitis B virus liver cirrhosis and 4/5 with hepatitis C virus liver cirrhosis with aberrant autophagy and hepatic Axin2+CD90+ cells developed hepatocarcinoma during the 8-year follow-up period (Supplementary Table S1). Immunofluorescence analysis showed that Axin2+CD90+ cells remained present in these hepatocarcinoma (Figure 1d).
To better trace the origin of these Axin2+CD90+ cells, a diethylnitrosamine-induced liver cirrhosis model was established in Axin2Cre;Rosa26EGFP rats whose Axin2+ cells were labeled by EGFP. In parallel with human samples, after stratification by aberrant autophagy at 4 weeks after the last diethylinitrosamine injection (Figure 1a and Supplementary Figure SIB), we also found that hepatic EGFP+CD90+ cells were significantly increased in cirrhotic livers with aberrant autophagy, whereas single EGFP+ cells were present in normal livers and cirrhotic livers without aberrant autophagy (Figures 1b and c and Supplementary Figure SIIB). Interestingly, 3/4 of these rat cirrhotic livers with aberrant autophagy and hepatic EGFP+CD90+ cells developed hepatocarcinoma within an additional 8-week observational period and hepatocarcinoma samples from these rats also contained these cells (Figure 1d). Collectively, these results indicated that aberrant autophagy was associated with the transition of Axin2+ into Axin2+CD90+ cells and the development of hepatocarcinoma from liver cirrhosis.
CD1331 and Epcam2 have been considered as biomarkers for liver cancer stem-like cells. However, low percentages of both Axin2+CD133+ and Axin2+Epcam+ cells have been detected in different types of liver cirrhosis from humans and rats, regardless of the autophagy status (Supplementary Figures SIII and SIV), suggesting that CD90 is a more reliable marker to trace liver cancer-like stem cells.
Axin2+CD90+ cells from liver cirrhosis show CSC properties
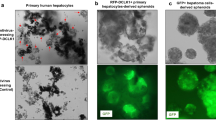
Next, the role of Axin2+CD90+ cells in hepatocarcinogenesis was investigated. Axin2+CD90+, Axin2+CD90− and Axin2−CD90− cells were sorted from the above human and rat cirrhotic liver tissues with aberrant autophagy. The expression levels of the Sox2 and Oct4 stemness genes were significantly upregulated in Axin2+CD90+ cells in contrast to Axin2+CD90− cells, and there was little Sox2 and Oct4 expression in Axin2−CD90− cells (Figure 2a). Similarly, Axin2+CD90+ cells, but not Axin2+CD90− and Axin2−CD90− cells, formed significant spheres in vitro and developed into solid tumors in nude mice (Figures 2b–d). Immunohistochemistry revealed that these tumor xenografts were AFP+ and CK19−. More importantly, Axin2+CD90+ cells were still present in human originated-tumor xenografts and EGFP(Axin2)+CD90+ cells were present in rat originated-tumor xenografts (Figure 2d), suggesting that Axin2+CD90+ cells play roles of cancer stem-like cells that are crucial for the development of hepatocarcinoma in humans and rats.
Axin2+CD90+ cells in cirrhotic liver possess CSC-like properties. Hepatic Axin2+CD90+, Axin2+CD90− and Axin2−CD90− cells were sorted from human cirrhotic livers with aberrant autophagy. EGFP+CD90+, EGFP+CD90− and EGFP−CD90− hepatic cells were sorted from rat cirrhotic livers with aberrant autophagy at 4 weeks after the last diethylinitrosamine injection. Western blot, sphere and tumor formation assays were then performed to detect Sox2 and Oct4 expression levels and cancer stem cell properties in these cells. (a) Representative western blots (lower panel) and densitometric analysis (upper panel) for Sox2 and Oct4 expression normalized to β-actin. (b) Numbers of formed spheres. (c) Sizes of formed tumor xenografts. (d) Representative tumor xenografts in nude mice (denoted by arrow heads) and corresponding immunohistochemistry staining for α-fetoprotein (AFP) and cytokeratin 19 (CK19) expression, along with double immunofluorescence staining for Axin2/EGFP+CD90+ cells in xenografts. The dashed squares indicate the higher magnification images of the merged Axin2/EGFP+CD90+ cells. Scale bar: 50 μm. All data are representative images or are expressed as the means±s.e.m. of each group (n=4) from three separate experiments. *P<0.05 vs Axin2+CD90− cells.
Induction or inhibition of autophagy increases or reduces the transition of Axin2+ cells into Axin2+CD90+ cells in cirrhotic livers
We further tested the role of autophagy in the transition of Axin2+ cells into Axin2+CD90+ cells by pharmacologically inducing or genetically and pharmacologically inhibiting aberrant autophagy in rat cirrhotic livers. Four weeks after model induction, rats with non-autophagic liver cirrhosis were intraperitoneally injected with rapamycin twice per week for 4 weeks, while rats with liver cirrhosis and aberrant autophagy were randomized and injected intravenously with shRNA-Atg3 and shRNA-Atg7 lentiviruses twice per week or were intraperitoneally injected with chloroquine weekly for 4 weeks. Rapamycin, an agonist of autophagy, significantly increased the conversion of LC3-I to LC3-II and reduced the expression of p62 in non-autophagic cirrhotic livers, indicating aberrant autophagy induction in non-autophagic cirrhotic livers (Figure 3a). However, knockdown of Atg3 and Atg7 by shRNA-Atg3 and shRNA-Atg7 lentivirus injection (Supplementary Figure SV) significantly reduced the conversion of LC3-I to LC3-II and increased the expression of p62 in aberrant autophagic cirrhotic livers (Figure 3a). Treatment with chloroquine further increased the expression of both LC3-II and p62 (Figure 3a), demonstrating autophagy inhibition in rat aberrant autophagic cirrhotic livers. More interestingly, double immunofluorescence staining and flow cytometry revealed that autophagy induction significantly increased and autophagy inhibition clearly reduced EGFP (Axin2) and CD90 double-positive cells in rat cirrhotic livers (Figures 3b and c). These results suggested that the transition of Axin2+ cells into Axin2+CD90+ cells in cirrhotic livers was dependent on autophagy.
The transition of Axin2+ cells into Axin2+CD90+ cells is dependent on aberrant autophagy in liver cirrhosis. Axin2Cre;Rosa26EGFP rats with non-autophagic or aberrant autophagic liver cirrhosis at 4 weeks after induction were administered with rapamycin or infected with vector lentiviruses or lentiviruses carrying Atg3/Atg7-shRNAs or were injected with chloroquine. Four weeks after treatment, liver autophagy influx was determined by western blot analysis. The presentation and quantitation of hepatic EGFP+CD90+ cells in liver tissues were determined by double immunofluorescence staining and flow cytometry. Induction of aberrant autophgay by rapamycin and inhibition of aberrant autophagy by shRNA interference or chloroquine, respectively, increased and reduced the numbers of hepatic EGFP+CD90+ cells in liver cirrhosis. (a) Representative western blot (left panel) and densitometric analyses (right panel) for autophagy influx in different livers tissues normalized to β-actin. (b) Double immunofluorescence staining (left panel) and quantitative percentages (right panel) for EGFP(Axin2)+CD90+ cells in different liver tissues. The dashed squares indicate the higher magnification image of merged EGFP+CD90+ cells. Scale bar: 50 μm. (c) Representative flow cytometry of hepatic EGFP+CD90+ cells (upper right quadrant) in different rat liver tissues. The percentages of double-positive cells are shown in the upper right quadrants of the representative flow cytometry graphs. All data are representative images or are expressed as the means±s.e.m. of each group (n=4) from three separate experiments. *P<0.05 vs liver cirrhosis with shRNA-vector/vehicle.
Autophagy-dependent HGF is required for the transition of Axin2+ cells into Axin2+CD90+ cells in liver cirrhosis
Previous studies have shown that HGF, interleukin (IL)-2R, IL-6, IL-16, IL-18 and CXCL12 expression are upregulated during the progression from liver cirrhosis to hepatocarcinoma.23 Enzyme-linked immunosorbent assay showed that HGF levels but not IL-2R, IL-6, IL-16, IL-18 or CXCL12 levels were significantly elevated in sera from rats and human patients with aberrant autophagic liver cirrhosis, in contrast to sera from subjects with autophagy-negative liver cirrhosis (Figure 4a). Similarly, HGF expression levels were also significantly upregulated in the above human and rat cirrhotic livers with aberrant autophagy rather than in those without aberrant autophagy (Figures 4b and c). In addition to HGF expression in aberrant autophagic cirrhotic livers, induction of aberrant autophagy by rapamycin also resulted in HGF expression in non-autophagic cirrhotic livers. Nevertheless, inhibition of autophagy by Atg3 and Atg7 silencing or by chloroquine treatment significantly reduced the expression of HGF in rat cirrhotic liver tissues with aberrant autophagy (Figure 4c and Supplementary Figures SVI and SVII). Previous study has shown that methylation of HGF gene promoter by DNA methyltransferase1 (DNMT1) blocks HGF expression in cells.24 We found that induction or inhibition of aberrant autophagy respectively reduced or increased expression of DNMT1 in cirrhotic livers, which caused increase in or decrease in HGF expression (Figure 4c and Supplementary Figure SVI). Hence, HGF expression was upregulated in liver cirrhosis in an autophagy-dependent manner via DNMT1 degradation.
Autophagy-dependent HGF expression is required for the generation of Axin2+CD90+ cells in liver cirrhosis. All samples were first stratified according to the autophagy status in liver cirrhosis. HGF production significantly increased in samples from liver cirrhosis with aberrant autophagy. (a) The concentrations of cytokines were tested by enzyme-linked immunosorbent assay in serum from humans with healthy livers (n=8) and alcohol- (n=6), hepatitis B virus- (n=10) and hepatitis C virus-related liver cirrhosis (n=6) and from control rats or rats with liver cirrhosis at 8 weeks after the last diethylinitrosamine injection (n=4). Data are expressed as the means±s.e.m. of each group from three separate experiments. *P<0.05 vs serum from subjects without aberrant autophagy in liver cirrhosis. (b) Western blot analysis (left panel) and immunofluorescence staining (right panel) for HGF expression in healthy human livers and cirrhotic liver samples. Scale bar: 100 μm. (c) Axin2Cre;Rosa26EGFP rats with non-autophagic or aberrant autophagic liver cirrhosis at 4 weeks after induction were administered with rapamycin or infected with vector lentiviruses or lentiviruses carrying Atg3/Atg7-shRNAs or HGF-shRNA or were injected with chloroquine. Four weeks after treatment, the autophagic flux and degradation of DNMT1 was analyzed by western blot. Western blotting (left panel) and immunofluorescence staining (right panel) showed that HGF expression was induced by rapamycin and inhibited by either chloroquine or transfection of Atg3/Atg7-shRNAs or HGF-shRNA. Scale bar: 100 μm. (d) Double immunofluorescence staining (left panel—dashed squares, scale bar: 50 μm; right panel—quantitative percentages) and (e) flow cytometry analysis showed that HGF knockdown by shRNA-HGF reduced hepatic EGFP+CD90+ cells in rat liver cirrhosis with aberrant autophagy. The percentages of double-positive cells are shown in the upper right quadrants of the representative flow cytometry graphs. The data are representative images or are expressed as the means±s.e.m. of each group (n=4) from three separate experiments. *P<0.05 vs liver cirrhosis with shRNA-vector.
To further evaluate the role of HGF expression in the autophagy-dependent transition of Axin2+ cells into Axin2+CD90+ cells, rats with aberrant autophagic liver cirrhosis were also randomized and treated with vector or HGF-shRNA lentiviruses for 4 weeks. Treatment with HGF-shRNA lentiviruses significantly reduced HGF expression, confirming the effectiveness of HGF silencing (Figure 4c and Supplementary Figures SVI and SVII). Further double immunofluorescence staining and flow cytometry analyses revealed that HGF knockdown significantly reduced EGFP+CD90+ cells in rat cirrhotic livers with aberrant autophagy (Figures 4d and e). Therefore, HGF is crucial for the autophagy-dependent transition of Axin2+ cells into Axin2+CD90+ cells during liver cirrhosis.
Autophagy-dependent HGF provides Met/JNK and Met/STAT3 signaling for the transition from Axin2+ cells into Axin2+CD90+ cells in liver cirrhosis
HGF binds to and activates its receptor cMet, which can further activate JNK and STAT3 signaling to regulate CSC self-renewal25, 26 and tumorigenesis in vitro.27 To further elucidate the mechanism of Axin2+CD90+ cell generation, we sorted Axin2− and Axin2+ cells from different types of human cirrhotic livers that were first stratified according to autophagy status and HGF expression. The autophagy status, degradation of DNMT1 and HGF expression in sorted cells were confirmed by western blottings (Figure 5a). We found that, compared with Axin2− cells, Met and β-catenin were significantly expressed in Axin2+ cells from cirrhotic livers in an autophagy- and HGF-independent manner. However, Met, JNK and STAT3 were only significantly phosphorylated in Axin2+ cells with aberrant autophagy and HGF expression. As a result, CD90 was expressed in these Met/JNK- and Met/STAT3-phosphorylated Axin2+ cells but not in those Axin2− cells or Axin2+ cells without significant phosphorylation of Met/JNK and Met/STAT3 (Figures 5a and b).
Autophagy provides HGF/Met/JNK and HGF/Met/STAT3 signaling for the transition of Axin2+ cells into Axin2+CD90+ cells in liver cirrhosis. Axin2− cells and Axin2+ cells were sorted from different types of human cirrhotic livers that were first stratified according to autophagy status and HGF expression. The autophagy status, degradation of DNMT1 and HGF expression of the sorted cells were confirmed by western blotting. Met and β-catenin were significantly expressed in Axin2+ cells from all types of cirrhotic livers. However, Met, JNK and STAT3 were only significantly phosphorylated, and CD90 was exclusively expressed in Axin2+ cells with aberrant autophagy and HGF expression. (a) Representative western blots (upper panel) and densitometric analysis (lower panel) for LC3-II, P62, DNMT1, HGF, β-catenin, total-Met and CD90 normalized to β-actin and phosphorylated signaling molecules normalized to total signaling molecules in Axin2− cells and Axin2+ cells from healthy human livers (n=8) and from alcohol- (n=6), hepatitis B virus- (n=10) and hepatitis C virus-related cirrhotic livers (n=6). (b) Double immunofluorescence staining for Axin2 and CD90 in the above hepatic Axin2− and Axin2+ cells. Scale bar: 50 μm. Data are expressed as the means±s.e.m. or representative images of each group from three separate experiments. *P<0.05 vs Axin2− cells from cirrhotic livers without aberrant autophagy.
Met and β-catenin were also significantly expressed in EGFP+ cells but not in EGFP− cells from transgenic rat cirrhotic livers, which was similar to that observed in human cells. CD90 was also exclusively expressed in Met/JNK- and Met/STAT3-phosphorylated EGFP+ cells with aberrant autophagy and HGF expression (Figure 6a). Moreover, inhibition of autophagy-dependent HGF by HGF-shRNAs transfection in rat liver cirrhosis significantly suppressed the phosphorylation of Met, JNK and STAT3 in EGFP+ cells and inhibited CD90 expression (Figures 6a and b). These results indicated that activation of autophagy-dependent HGF/Met/JNK and HGF/Met/STAT3 signaling was essential for the generation of hepatic Axin2+CD90+ cells from Axin2+ cells in liver cirrhosis.
Generation of EGFP(Axin2)+CD90+ cells in liver cirrhosis requires activation of Met/JNK and Met/STAT3 signaling in EGFP(Axin2)+ cells through autophagy-dependent HGF. Transgenic rats with autophagic cirrhotic livers were transfected with lentiviruses carrying vector-siRNAs or HGF-siRNAs. Four weeks later, EGFP− and EGFP+ cells were sorted from the above treated autophagic cirrhotic livers and non-autophagic cirrhotic livers. The autophagy status, degradation of DNMT1 and HGF expression of the sorted cells were confirmed by western blotting. Met and β-catenin were significantly expressed in EGFP+ cells from all types of cirrhotic livers. However, Met, JNK and STAT3 were only significantly phosphorylated, and CD90 was exclusively expressed in EGFP+ cells with aberrant autophagy and HGF expression. Transfection of HGF-shRNAs significantly suppressed the expression of HGF and the phosphorylation of Met, JNK and STAT3 in EGFP+ cells with aberrant autophagy, further inhibiting CD90 expression. (a) Representative western blots (left panel) and densitometric analyses (right panel) of LC3-II, P62, DNMT1, HGF, β-catenin, total-Met and CD90 normalized to β-actin and phosphorylated signaling molecules normalized to total signaling molecules in EGFP− and EGFP+ cells from transgenic rat cirrhotic livers with or without transfection of HGF-shRNAs (n=4). (b) Immunofluorescence staining for CD90 in the above hepatic EGFP− and EGFP+ cells from transgenic rats. Scale bar: 50 μm. Data are expressed as the means±s.em. or representative images of each group from three separate experiments. *P<0.05 vs EGFP− cells from cirrhotic livers without aberrant autophagy.
Blockade of Met/JNK or Met/STAT3 signaling attenuates the transition of Axin2+ cells into Axin2+CD90+ cells in liver cirrhosis
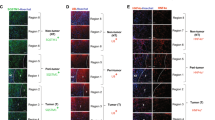
To further evaluate the role of Met/JNK and Met/STAT3 signaling in the autophagy-dependent transition of Axin2+ cells into Axin2+CD90+ cells, rats with aberrant autophagic liver cirrhosis were randomized and administered with a specific inhibitor of JNK, SP600125, or a specific inhibitor of STAT3, stattic, for 4 weeks. EGFP+ cells with aberrant autophagy and HGF expression were then sorted (Figure 7a). Western blot analysis was performed to demonstrate that Met phosphorylation and expression and β-catenin expression were not influenced by treatment with either SP600125 or stattic; however, JNK and STAT3 phosphorylation was inhibited by SP600125 and stattic, respectively. As a result, CD90 was not expressed in these JNK- and STAT3-inactivated EGFP+ cells but was expressed in EGFP+ cells with phosphorylated JNK and STAT3, which were from cirrhotic livers treated with vehicle (Figures 7a and b).
Blockade of HGF/Met/JNK or HGF/Met/STAT3 signaling reduces the generation of Axin2+CD90+ cells in cirrhotic livers. Transgenic rats with autophagic cirrhotic livers received vehicle, SP600125 or stattic. After 4 weeks, EGFP+ cells were sorted from the above cirrhotic livers, and their aberrant autophagy and HGF expression levels were confirmed by western blotting. Treatment with SP600125 and stattic in cirrhotic livers abrogated JNK and STAT3 phosphorylation in sorted EGFP+ cells; however, HGF and β-catenin expression and Met phosphorylation were not influenced. Either JNK or STAT3 inhibition was capable of preventing CD90 expression in EGFP+ hepatic cells. (a) Representative western blots (left panel) and densitometric analysis (right panel) for LC3-II, P62, HGF, β-catenin, phosphorylated signaling molecules and CD90 normalized to β-actin and total signaling molecules in sorted EGFP+ hepatic cells. Data are expressed as the means±s.e.m. of each group (n=4) from three separate experiments. *P<0.05 vs EGFP+ cells from cirrhotic livers with vehicle. (b) Immunofluorescence staining for CD90 in the above hepatic EGFP+ cells from transgenic rats. Scale bar: 50 μm. (c) Double immunofluorescence staining (upper panel—dashed squares, scale bar: 50 μm; lower panel—quantitative percentages) and (d) flow cytometry analysis for EGFP(Axin2) and CD90 in the above-described cirrhotic livers showed that the generation of EGFP+CD90+ hepatic cells was reduced by systemic treatment with SP600125 or stattic in rat liver cirrhosis with aberrant autophagy. The percentages of double-positive cells are shown in the upper right quadrants of the representative flow cytometry graphs. Data are representative images or are expressed as the means±s.e.m. of each group (n=4) from three separate experiments. *P<0.05 vs liver cirrhosis with vehicle.
In addition, immunofluorescence staining and flow cytometry also revealed that treatment with either SP600125 or stattic significantly reduced EGFP+(Axin2+)CD90+ cells in rat cirrhotic livers with aberrant autophagy (Figures 7c and d), suggesting that Met/JNK and Met/STAT3 signaling was required for autophagy-dependent generation of Axin2+CD90+ cells in liver cirrhosis.
Blockade of autophagy-dependent HGF/Met/JNK or HGF/Met/STAT3 signaling prevents hepatocarcinogenesis from liver cirrhosis by decreasing Axin2+CD90+ cells
Since previous data have shown that Axin2+CD90+ cells possess cancer stem-like cell properties and that liver cirrhosis with rare Axin2+CD90+ cells fail to progress into hepatocarcinoma (Figure 1 and Supplementary Table S1), we found that rat liver cirrhosis without aberrant autophagy and EGFP(Axin2)+CD90+ cells did not develop hepatocarcinoma. However, induction of autophagy by rapamycin in those rats liver cirrhosis caused that 4/5 of the rat cirrhotic livers generated EGFP(Axin2)+CD90+ cells and developed hepatocarcinoma. We also found that while 3/4 of the rat liver cirrhosis with aberrant autophagy and EGFP+CD90+ cells developed hepatocarcinoma, none of the rats with liver cirrhosis and a reduced number of EGFP+CD90+ cells developed hepatocarcinoma after treatment with chloroquine, Atg3 and Atg7-shRNA lentiviruses or HGF-shRNA lentiviruses, or with SP600125 and static (Supplementary Table S2). Figure 8 shows the representative histological findings of these liver samples, which were revealed by hematoxylin and eosin and Sirius staining at 8 weeks after the last treatment. Collectively, either inhibition of autophagy or autophagy-dependent HGF/Met/JNK or HGF/Met/STAT3 signaling was capable of preventing hepatocarcinogenesis from liver cirrhosis by decreasing the generation of Axin2+CD90+ cells.
Histology of rat cirrhotic livers at 4 weeks after different treatments. Rat cirrhotic livers with different autophagy status were determined by biopsy at 4 weeks after diethylinitrosamine induction. The rats were then administered with rapamycin, chloroquine, Atg3 and Atg7-shRNA lentiviruses or HGF-shRNA lentiviruses, or SP600125 and stattic for 4 weeks. After additional 4 weeks of observation, liver samples were collected to receive hematoxylin and eosin and Sirius staining. Scale bar: 200 μm. The non-autophagic cirrhotic livers with rapamycin and aberrant autophagic cirrhotic livers without treatment or with vehicle/vector treatment developed hepatocarcinoma. The hepatocarcinoma area was indicated by arrow heads. No single rat cirrhotic livers without aberrant autophagy and treatment or with chloroquine, Atg3 and Atg7-shRNA lentiviruses or HGF-shRNA lentiviruses, or SP600125 and stattic treatment developed hepatocarcinoma. Representative histological images of each group (n=4) from three separate experiments.
Discussion
Liver cirrhosis poses a high risk for hepatocarcinogenesis. In this study, we present novel insights into the progression from liver cirrhosis to hepatocarcinoma by identifying the origin of liver CSC-like cells in cirrhotic livers. We showed that hepatic Axin2/EGFP+CD90+ cells originated from Axin2/EGFP+ cells, aquired CSC properties in human and rat cirrhotic livers with aberrant autophagy, and contributed to hepatocarcinogenesis. The induction or inhibition of autophagy in cirrhotic livers promoted or prevented hepatocarcinogenesis by inducing or reducing the generation of hepatic Axin2/EGFP+CD90+ cells from Axin2/EGFP+ cells due to an increase or decrease in HGF expression. Further cytological experiments revealed that autophagy-dependent HGF expression in liver cirrhosis activated Met/JNK and Met/STAT3 signaling in hepatic Axin2/EGFP+ cells, resulting in the transition to Axin2/EGFP+CD90+ CSC-like cells. Either HGF knockdown or inhibiting the activation of Met/JNK or Met/STAT3 signaling in liver cirrhosis prevented the formation of Axin2/EGFP+CD90+ cells and further hepatocarcinogenesis. Our results indicate that in the mechanism of CSCs generation in liver cirrhosis, autophagy-dependent activation of HGF/Met/JNK and HGF/Met/STAT3 signaling plays a crucial role in hepatocarcinogenesis. Disruption of this process may provide a promising therapeutic approach to prevent the progression of liver cirrhosis into hepatocarcinoma.
Liver CSCs are a small subset of cancer cells that express the stemness-related transcription factors Sox2 and Oct4 and have potent capacity for self-renewal, differentiation and tumorigenesis.1, 2, 3 Recently, liver CSCs have been isolated from hepatocarcinoma cell lines and some liver cancer tissues.4, 5 However, the mechanism of the origin of liver CSCs and their role in hepatocarcinogenesis is unclear. Liver CSCs are theoretically presumed to originate from normal stem cells,28 and a recent study has shown that hepatic Axin2+ cells can self-renew in response to Wnt/β-catenin stimulation.11 In the present study, we sorted cells from liver cirrhosis using the stemness-responsive marker, Axin2, together with a well-documented CSC marker, CD90, which shows stable expression in liver CSCs that does not vary in different cell lines.22 We found that these hepatic Axin2+CD90+ cells expressed high levels of Sox2 and Oct4 and were capable of forming spheres in vitro as well as generating tumor xenografts in vivo. In addition, compared with cirrhotic livers with few or no Axin2+CD90+ cells, cirrhotic livers with high percentages of hepatic Axin2+CD90+ cells progressed to hepatocarcinoma. Furthermore, these Axin2+CD90+ cells were also present in the tumor xenografts and hepatocarcinoma tissues, which exhibited the asymmetric self-renewal division trait of CSCs.29, 30 In addition to the similar results obtained for hepatic EGFP+CD90+ cells from Axin2Cre;Rosa26EGFP cirrhotic rat liver with EGFP-tagged Axin2 cells, treatment that reduced the numbers of EGFP+CD90+ cells in rat liver cirrhosis significantly prevented hepatocarcinogenesis. These findings demonstrated that hepatic Axin2+CD90+ cells assume the responsibility of CSCs during the progression of liver cirrhosis to hepatocarcinoma.
The current study further demonstrated that the generation of hepatic Axin2+CD90+ cells from Axin2+ cells in liver cirrhosis was dependent on the activation of Met/JNK and Met/STAT3 signaling. Previous studies have shown that JNK and STAT3 signaling is crucial for the tumorigenesis of leiomyosarcoma,27 self-renewal of glioma stem cells25 and the CSC properties of non-small-cell lung cancer.26 However, we showed that the activation of Met, as the upstream receptor tyrosine kinase, was critical to activating downstream JNK and STAT3 signaling and inducing CD90 expression in Axin2/EGFP+ cells. Either the inhibition of Met/JNK or Met/STAT3 signaling in liver cirrhosis by pharmacological inhibitors was capable of reducing the transition of sorted hepatic Axin2/EGFP+ cells into Axin2/EGFP+CD90+ cells and preventing hepatocarcinogenesis. Although Wnt/β-catenin signaling comprehensively participates in inducing and maintaining the CSC phenotype in several cancers,31, 32, 33 we found that β-catenin was significantly expressed in both hepatic Axin2/EGFP+CD90− cells and Axin2/EGFP+CD90+ cells from liver cirrhosis and that β-catenin expression was not regulated by phosphorylated Met, indicating that Wnt/β-catenin signaling was not required for the transition of hepatic Axin2/EGFP+ cells into Axin2/EGFP+CD90+ cells. Conversely, Wnt/β-catenin signaling is required for hepatic Axin2+ cell self-renewal in normal livers,11 and β-catenin was not expressed in Axin2/EGFP− cells but was expressed in Axin2/EGFP+ cells, which still exhibited a certain degree of Sox2 and Oct4 expression and clonality, suggesting that Wnt/β-catenin signaling was responsible for maintaining normal stemness characteristics in Axin2+ cells and that Met/JNK and Met/STAT3 signaling was responsible for the tumorigenicity of Axin2/EGFP+CD90+ cells in liver cirrhosis. Although epigenetic dysregulation of Axin2 would affect CSC-like traits by dysregulating Wnt/β-catenin signaling,12, 13, 15 our data also indicated that under the context without dysregulation, Axin2/β-catenin signaling provided the stem cell context for other signaling to generate liver CSCs. Furthermore, in addition to the absence of Met phosphorylation in hepatic Axin2/EGFP− cells, Met expression was also significantly lower in hepatic Axin2/EGFP− cells than that in Axin2/EGFP+ cells. This finding indicated that adequate expression of Met was likely a prerequisite for its phosphorylation and the activation of downstream JNK and STAT3 signaling to induce the transition of Axin2/EGFP+ cells into Axin2/EGFP+CD90+ cells.
HGF is the ligand for Met phosphorylation.34 Our results showed that both Met and downstream JNK and STAT3 were phosphorylated in Axin2/EGFP+ cells from liver cirrhosis with significant HGF expression. Knockdown of HGF expression in cirrhotic livers by shRNA interference effectively inhibited Met, JNK and STAT3 phosphorylation in EGFP+ cells, abrogated their transition into EGFP+CD90+ cells and reduced hepatocarcinogenesis, demonstrating that the generation of EGFP+CD90+ CSCs and hepatocarcinogenesis in cirrhotic liver occurred in an HGF/Met/JNK- and HGF/Met/STAT3-dependent manner. β-Catenin expression in both hepatic Axin2/EGFP− and Axin2/EGFP+ cells was not regulated by HGF expression in liver cirrhosis, further implying that HGF/Met/JNK and HGF/Met/STAT3 signaling was primarily responsible for tumorigenicity during the generation of Axin2+CD90+ CSCs in liver cirrhosis. However, the activation of HGF/Met/JNK and HGF/Met/STAT3 signaling in hepatic Axin2+/EGFP+ cells and the subsequent transition into Axin2+/EGFP+CD90+ cells relied on autophagy in liver cirrhosis. Because of pathological changes, cirrhotic livers suffer from chronic dystrophy injury,20 resulting in autophagy which supports cell survival against denutrition through nutrients and growth factors production.16, 17 We found that induction or inhibition of aberrant autophagy respectively reduced or increased expression of DNMT1 in cirrhotic livers, indicating the autophagy-mediated degradation of DNMT1.35 DNMT1 blocks HGF expression in cells through methylating HGF gene promoter.24 Autophagy presence or induction degraded DNMT1 and further induced expression of HGF in cirrhotic livers. Autophagy deficiency or interference in liver cirrhosis preserved DNMT1 expression and inhibited HGF expression and then failed to activate Met/JNK and Met/STAT3 signaling in Axin2+ cells. The most important, autophagy deficiency or interference effectively prevented hepatocarcinogenesis in liver cirrhosis by reducing the generation of Axin2+CD90+ CSCs through the inhibition of HGF/Met/JNK and HGF/Met/STAT3 signaling, supporting the notion that autophagy induction promotes neoplastic liver nodules.36
In summary, our findings indicate that hepatic Axin2+CD90+ cells play a CSC-like role in the progression from liver cirrhosis to hepatocarcinoma. The origin of these liver cancer stem-like cells in liver cirrhosis is dependent on the autophagy-induced activation of HGF/Met/JNK and HGF/Met/STAT3 signaling in Axin2+ hepatic cells. Therefore, the manipulation of autophagy, HGF/Met/JNK or HGF/Met/STAT3 signaling in liver cirrhosis may effectively prevent hepatocarcinogenesis. If our findings are validated in a larger population of human patients, aberrant autophagy and elevated HGF expression may be valuable biomarkers to predict hepatocarcinogenesis in liver cirrhosis. Therapeutic targeting of aberrant autophagy and HGF may be new clinical strategies to prevent the progression of liver cirrhosis to hepatocarcinoma.
Materials and methods
More details of the experiment methods are available in Supplementary Information.
Human tissue samples
A total of eight non-liver cirrhosis surgical samples and blood samples were collected from patients with hepatic hemangioma; 18 alcohol-related, 30 chronic hepatitis B-related and 18 chronic hepatitis C-related liver cirrhosis biopsied samples and blood samples were collected at Union Hospital, Tongji Medical College, Huazhong University of Science and Technology from September 2007. Those patients were followed-up to December 2015. Three surgical hepatocarcinoma samples from patients with alcohol-related liver cirrhosis, eight hepatocarcinoma samples from those with hepatitis B-related liver cirrhosis and four hepatocarcinoma samples from those with hepatitis C-related liver cirrhosis were collected by surgery. These tissue samples were immediately used for subsequent experiments or were fixed in paraformaldehyde and paraffin-embedded after biopsy or surgical removal. Histopathological diagnosis of hepatocarcinoma was performed according to World Health Organization criteria. Fibrolamellarhepatocarcinoma, cholangiocarcinoma and combined hepatocholangiocarcinoma were excluded. This study was approved by the Ethics Committee of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology. Written informed consent was obtained from each patient.
Generation of transgenic rats
To trace the origin of Axin2+CD90+ cells, an Axin2Cre rat whose Cre expression was controlled by Axin2 promoter was crossed with a ROSA26R-EGFP reporter rat to generate an Axin2Cre;Rosa26EGFP rat (Biocytogen, Beijing, China). In Axin2Cre;Rosa26EGFP rat, Cre cut the termination sequence between loxP sites in ROSA26R-EGFP and was labeled by EGFP. Axin2+ cells were labeled by EGFP through Cre expression.
Generation of lentiviral shRNA vectors
Specific shRNAs of Atg3, Atg7 and HGF were constructed and carried by lentiviral vectors with a specific albumin promoter.
Animal models
To establish a model of the progression of liver cirrhosis to hepatocarcinoma, 8-week-old male Axin2Cre;Rosa26EGFP SD rats were injected intraperitoneally with 50 mg/kg diethylinitrosamine (N0258-1G; Sigma-Aldrich, Shanghai, China) twice per week for 8 weeks. The rats were fed a regular diet for an additional 4 weeks. The rats were biopsied to evaluate the presence of liver cirrhosis and autophagy status. Rats with non-autophagic liver cirrhosis were intraperitoneally injected with rapamycin (2 mg/kg, S1039; Selleck, Shanghai, China) twice per week for 4 weeks. Rats with aberrant autophagic liver cirrhosis were then number randomized and injected intravenously with 1 × 109 TU/ml shRNA-vector, shRNA-Atg3, shRNA-Atg7 and shRNA-HGF lentiviruses in transduction enhancement reagent (Envirus; Engreen Biosystem, Beijing, China) twice per week for 4 weeks. In addition, the rats also received chloroquine (50 mg/kg, an inhibitor of the late stage of autophagy; Sigma-Aldrich) weekly, with SP600125 (a JNK inhibitor, 15 mg/kg, S1460; Selleck) or stattic (a STAT3 inhibitor, 2 mg/kg, S7024; Selleck) administered twice per week by intraperitoneal injection for 4 weeks. A group of normal rats or rats with vehicle injection served as the control where appropriate. The rats were killed after an additional 4-week observation after the final treatment. The entire experimental period was 20 weeks.
Cytokines detection
HGF, IL-2 R, IL-6, IL-16, IL-18 and CXCL12 levels in serum from individual patients and rats were analyzed by enzyme-linked immunosorbent assay using specific kits according to the manufacturers’ instructions (Boster, Wuhan, China).
Flow cytometry
The percentages of human Axin2+CD90+, Axin2+CD133+ and Axin2+Epcam+ cells or rat EGFP+CD90+, EGFP+CD133+ and EGFP+Epcam+ cells were determined by flow cytometry using BD Accuri C6 (BD Biosciences, San Jose, CA, USA). The sorting of Axin2+CD90+, Axin2+CD90− and Axin2−CD90− cells and EGFP+CD90+, EGFP+CD90− and EGFP−CD90− cells was performed using a commercial sorter (FACSAria; BD Biosciences), which was also used to evaluate sphere and tumor xenograft formation capabilities. Axin2+ and Axin2− cells from each type of human cirrhotic liver, along with EGFP+ and EGFP− cells from rat cirrhotic livers with or without treatment, were sorted and further analyzed in vitro.
Tumor formation assay
Individual nude mice were injected subcutaneously with 5 × 105 human Axin2+CD90+, Axin2+CD90− and Axin2−CD90− cells or rat EGFP+CD90+, EGFP+CD90− and EGFP−CD90− cells, respectively. The formation and growth of implanted tumors were monitored for 8 weeks. The mice were then killed, and the tumor xenografts were dissected and compared in volumes.
Sphere formation assay
The sorted human Axin2+CD90+, Axin2+CD90− and Axin2−CD90− cells and the rat EGFP+CD90+, EGFP+CD90− and EGFP−CD90− cells were cultured at a density of 1000 cells/well in Dulbecco's modified Eagle's medium/F12 medium (HyClone, Logan, UT, USA) supplemented with 2% B27 (Invitrogen, Waltham, MA, USA), 100 U/ml of penicillin and 100 μg/ml of streptomycin, 20 ng/ml epidermal growth factor and 20 ng/ml basic fibroblast growth factor (PeproTech; Rocky Hill, NJ, USA) using ultra-low attachment six-well plates (Corning, New York, NY, USA) for 2 weeks. The cells were exposed to fresh medium every 3 days. The formed spheres with diameters of >75 μm were counted.
Western blotting
LC3-I/LC3-II conversion, P62, DNMT1 and HGF expression were detected by western blotting in above sorted cells and in different human and rat liver samples with or without treatment. Western blotting was also performed to detect the expression of Sox2 and Oct4, β-catenin and CD90 and the phosphorylation of Met, JNK and STAT3 in the sorted cells described above. The interference efficiencies of Atg3 and Atg7 were confirmed by western blot analysis in rat cirrhotic livers transfected with shRNA lentiviruses.
Immunocytofluorescence
Immunocytofluorescence was performed using the sorted Axin2− and Axin2+ cells from human cirrhotic livers and sorted EGFP− and EGFP+ cells from rat cirrhotic livers described above.
Immunohistofluorescence
The expression levels of HGF, Axin2 and CD90 in different human and rat liver tissue samples were determined by immunofluorescence.
Immunohistochemistry
The expression levels of α-fetoprotein and cytokeratin 19 in individual tumor xenograft samples were detected by immunohistochemistry.
Histology
At 4 weeks after the last treatment (12 weeks after the last diethylinitrosamine injection), 4-μm-thick cross-sections of rat cirrhotic livers with different autophagy status and different treatments were cut and subjected to hematoxylin–eosin and Sirius staining.
Statistical analysis
Sample size was computed by using formulas for two proportions comparison available on website: http://powerandsamplesize.com/Calculators/. All data are presented as the means±s.e.m. After demonstration of homogeneity of variance with the Bartlett test, one-way ANOVA, followed by the Student–Newman–Keuls test where appropriate, was used to evaluate statistical significance. Values of P<0.05 were considered statistically significant. All experiments were performed in triplicate.
References
Ma S, Chan KW, Hu L, Lee TK, Wo JY, Ng IO et al. Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology 2007; 132: 2542–2556.
Terris B, Cavard C, Perret C . EpCAM, a new marker for cancer stem cells in hepatocellular carcinoma. J Hepatol 2010; 52: 280–281.
Yang ZF, Ho DW, Ng MN, Lau CK, Yu WC, Ngai P et al. Significance of CD90+ cancer stem cells in human liver cancer. Cancer Cell 2008; 13: 153–166.
Yang ZF, Ngai P, Ho DW, Yu WC, Ng MN, Lau CK et al. Identification of local and circulating cancer stem cells in human liver cancer. Hepatology 2008; 47: 919–928.
Yamashita T, Ji J, Budhu A, Forgues M, Yang W, Wang HY et al. EpCAM-positive hepatocellular carcinoma cells are tumor-initiating cells with stem/progenitor cell features. Gastroenterology 2009; 136: 1012–1024.
Jho EH, Zhang T, Domon C, Joo CK, Freund JN, Costantini F . Wnt/beta-catenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol Cell Biol 2002; 22: 1172–1183.
Bowman AN, van Amerongen R, Palmer TD, Nusse R . Lineage tracing with Axin2 reveals distinct developmental and adult populations of Wnt/beta-catenin-responsive neural stem cells. Proc Natl Acad Sci USA 2013; 110: 7324–7329.
Katoh M, Katoh M . WNT signaling pathway and stem cell signaling network. Clin Cancer Res 2007; 13: 4042–4045.
Lim X, Tan SH, Yu KL, Lim SB, Nusse R . Axin2 marks quiescent hair follicle bulge stem cells that are maintained by autocrine Wnt/beta-catenin signaling. Proc Natl Acad Sci USA 2016; 113: E1498–E1505.
van Amerongen R, Bowman AN, Nusse R . Developmental stage and time dictate the fate of Wnt/beta-catenin-responsive stem cells in the mammary gland. Cell Stem Cell 2012; 11: 387–400.
Wang B, Zhao L, Fish M, Logan CY, Nusse R . Self-renewing diploid Axin2(+) cells fuel homeostatic renewal of the liver. Nature 2015; 524: 180–185.
Fang L, Cai J, Chen B, Wu S, Li R, Xu X et al. Aberrantly expressed miR-582-3p maintains lung cancer stem cell-like traits by activating Wnt/beta-catenin signalling. Nat Commun 2015; 6: 8640.
Wu G, Liu A, Zhu J, Lei F, Wu S, Zhang X et al. MiR-1207 overexpression promotes cancer stem cell-like traits in ovarian cancer by activating the Wnt/beta-catenin signaling pathway. Oncotarget 2015; 6: 28882–28894.
Martins-Neves SR, Corver WE, Paiva-Oliveira DI, van den Akker BE, Briaire-de-Bruijn IH, Bovee JV et al. Osteosarcoma stem cells have active Wnt/beta-catenin and overexpress SOX2 and KLF4. J Cell Physiol 2016; 231: 876–886.
Chai S, Ng KY, Tong M, Lau EY, Lee TK, Chan KW et al. Octamer 4/microRNA-1246 signaling axis drives Wnt/beta-catenin activation in liver cancer stem cells. Hepatology 2016; 64: 2062–2076.
Levine B, Kroemer G . Autophagy in the pathogenesis of disease. Cell 2008; 132: 27–42.
Cecconi F, Levine B . The role of autophagy in mammalian development: cell makeover rather than cell death. Dev Cell 2008; 15: 344–357.
Wang X, Sun W, Shen W, Xia M, Chen C, Xiang D et al. Long non-coding RNA DILC regulates liver cancer stem cells via IL-6/STAT3 axis. J Hepatol 2016; 64: 1283–1294.
Cao L, Fan X, Jing W, Liang Y, Chen R, Liu Y et al. Osteopontin promotes a cancer stem cell-like phenotype in hepatocellular carcinoma cells via an integrin-NF-kappaB-HIF-1alpha pathway. Oncotarget 2015; 6: 6627–6640.
Wiegand J, Berg T . The etiology, diagnosis and prevention of liver cirrhosis: part 1 of a series on liver cirrhosis. Dtsch Arztebl Int 2013; 110: 85–91.
Song YJ, Zhang SS, Guo XL, Sun K, Han ZP, Li R et al. Autophagy contributes to the survival of CD133+ liver cancer stem cells in the hypoxic and nutrient-deprived tumor microenvironment. Cancer Lett 2013; 339: 70–81.
Zhang X, Jiang P, Shuai L, Chen K, Li Z, Zhang Y et al. miR-589-5p inhibits MAP3K8 and suppresses CD90+ cancer stem cells in hepatocellular carcinoma. J Exp Clin Cancer Res 2016; 35: 176.
Capone F, Guerriero E, Colonna G, Maio P, Mangia A, Castello G et al. Cytokinome profile evaluation in patients with hepatitis C virus infection. World J Gastroenterol 2014; 20: 9261–9269.
Sakai K, Takeda M, Okamoto I, Nakagawa K, Nishio K . Multiple regulatory mechanisms of hepatocyte growth factor expression in malignant cells with a short poly(dA) sequence in the HGF gene promoter. Oncol Lett 2015; 9: 405–410.
Kitanaka C, Sato A, Okada M . JNK signaling in the control of the tumor-initiating capacity associated with cancer stem cells. Genes Cancer 2013; 4: 388–396.
Song W, Ma Y, Wang J, Brantley-Sieders D, Chen J . JNK signaling mediates EPHA2-dependent tumor cell proliferation, motility, and cancer stem cell-like properties in non-small cell lung cancer. Cancer Res 2014; 74: 2444–2454.
Zhang YW, Wang LM, Jove R, Vande Woude GF . Requirement of Stat3 signaling for HGF/SF-Met mediated tumorigenesis. Oncogene 2002; 21: 217–226.
Oikawa T . Cancer stem cells and their cellular origins in primary liver and biliary tract cancers. Hepatology 2016; 64: 645–651.
Wicha MS, Liu S, Dontu G . Cancer stem cells: an old idea—a paradigm shift. Cancer Res 2006; 66: 1883–1890; discussion 1895-1886.
Pardal R, Clarke MF, Morrison SJ . Applying the principles of stem-cell biology to cancer. Nat Rev Cancer 2003; 3: 895–902.
Liu D, Du L, Chen D, Ye Z, Duan H, Tu T et al. Reduced CD146 expression promotes tumorigenesis and cancer stemness in colorectal cancer through activating Wnt/beta-catenin signaling. Oncotarget 2016; 7: 40704–40718.
Wang Y, He L, Du Y, Zhu P, Huang G, Luo J et al. The long noncoding RNA lncTCF7 promotes self-renewal of human liver cancer stem cells through activation of Wnt signaling. Cell Stem Cell 2015; 16: 413–425.
Gong L, Song J, Lin X, Wei F, Zhang C, Wang Z et al. Serine-arginine protein kinase 1 promotes a cancer stem cell-like phenotype through activation of Wnt/beta-catenin signalling in NSCLC. J Pathol 2016; 240: 184–196.
Cecchi F, Rabe DC, Bottaro DP . Targeting the HGF/Met signalling pathway in cancer. Eur J Cancer 2010; 46: 1260–1270.
Bourdeau V, Ferbeyre G . CDK4-CDK6 inhibitors induce autophagy-mediated degradation of DNMT1 and facilitate the senescence antitumor response. Autophagy 2016; 12: 1965–1966.
Kowalik MA, Perra A, Ledda-Columbano GM, Ippolito G, Piacentini M, Columbano A et al. Induction of autophagy promotes the growth of early preneoplastic rat liver nodules. Oncotarget 2016; 7: 5788–5799.
Acknowledgements
This work was supported by the National Natural Science Foundation of China under Grant (No. 81400674) and China Scholarship Council under Grant (No. 201508500057) to JL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Li, J., Hu, S., Wang, L. et al. Autophagy-dependent generation of Axin2+ cancer stem-like cells promotes hepatocarcinogenesis in liver cirrhosis. Oncogene 36, 6725–6737 (2017). https://doi.org/10.1038/onc.2017.272
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2017.272
This article is cited by
-
Crosstalk between autophagy and CSCs: molecular mechanisms and translational implications
Cell Death & Disease (2023)
-
Effects of SP600125 and hypothermic machine perfusion on livers donated after cardiac death in a pig allograft transplantation model
European Journal of Medical Research (2021)
-
Interplay of autophagy and cancer stem cells in hepatocellular carcinoma
Molecular Biology Reports (2021)
-
CCND1 silencing suppresses liver cancer stem cell differentiation through inhibiting autophagy
Human Cell (2020)