Abstract
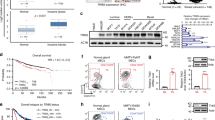
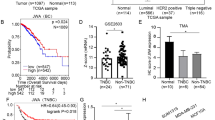
c-Jun N-terminal kinase (JNK) plays a vital role in malignant transformation of different cancers, and JNK is highly activated in basal-like triple-negative breast cancer (TNBC). However, the roles of JNK in regulating cancer stem-like cell (CSC) phenotype and tumorigenesis in TNBC are not well defined. JNK is known to mediate many cellular events via activating c-Jun. Here, we found that JNK regulated c-Jun activation in TNBC cells and that JNK activation correlated with c-Jun activation in TNBC tumors. Furthermore, the expression level of c-Jun was significantly higher in TNBC tumors than in non-TNBC tumors, and high c-Jun mRNA level was associated with shorter disease-free survival of patients with TNBC. Thus, we hypothesized that the JNK/c-Jun signaling pathway contributes to TNBC tumorigenesis. We found that knockdown of JNK1 or JNK2 or treatment with JNK-IN-8, an adenosine triphosphate-competitive irreversible pan-JNK inhibitor, significantly reduced cell proliferation, the ALDH1+ and CD44+/CD24− CSC subpopulations, and mammosphere formation, indicating that JNK promotes CSC self-renewal and maintenance in TNBC. We further demonstrated that both JNK1 and JNK2 regulated Notch1 transcription via activation of c-Jun and that the JNK/c-Jun signaling pathway promoted CSC phenotype through Notch1 signaling in TNBC. In a TNBC xenograft mouse model, JNK-IN-8 significantly suppressed tumor growth in a dose-dependent manner by inhibiting acquisition of the CSC phenotype. Taken together, our data demonstrate that JNK regulates TNBC tumorigenesis by promoting CSC phenotype through Notch1 signaling via activation of c-Jun and indicate that JNK/c-Jun/Notch1 signaling is a potential therapeutic target for TNBC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, Shyr Y et al. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J clin invest 2011; 121: 2750–2767.
D'Amato NC, Ostrander JH, Bowie ML, Sistrunk C, Borowsky A, Cardiff RD et al. Evidence for phenotypic plasticity in aggressive triple-negative breast cancer: human biology is recapitulated by a novel model system. PloS one 2012; 7: e45684.
Kong D, Li Y, Wang Z, Sarkar FH . Cancer stem cells and epithelial-to-mesenchymal transition (EMT)-phenotypic cells: are they cousins or twins? Cancers 2011; 3: 716–729.
Li F, Tiede B, Massague J, Kang Y . Beyond tumorigenesis: cancer stem cells in metastasis. Cell res 2007; 17: 3–14.
Economopoulou P, Kaklamani VG, Siziopikou K . The role of cancer stem cells in breast cancer initiation and progression: potential cancer stem cell-directed therapies. oncologist 2012; 17: 1394–1401.
Trosko JE . Review paper: cancer stem cells and cancer nonstem cells: from adult stem cells or from reprogramming of differentiated somatic cells. Vet pathol 2009; 46: 176–193.
McDermott SP, Wicha MS . Targeting breast cancer stem cells. Mol oncol 2010; 4: 404–419.
Park CY, Tseng D, Weissman IL . Cancer stem cell-directed therapies: recent data from the laboratory and clinic. Mol ther 2009; 17: 219–230.
Chen F . JNK-induced apoptosis, compensatory growth, and cancer stem cells. Cancer res 2012; 72: 379–386.
Cubero FJ, Zhao G, Trautwein C . JNK: a double-edged sword in tumorigenesis. Hepatology 2011; 54: 1470–1472.
Bogoyevitch MA, Kobe B . Uses for JNK: the many and varied substrates of the c-Jun N-terminal kinases. Microbiol mol biol rev 2006; 70: 1061–1095.
Li JY, Wang H, May S, Song X, Fueyo J, Fuller GN et al. Constitutive activation of c-Jun N-terminal kinase correlates with histologic grade and EGFR expression in diffuse gliomas. J neuro-oncol 2008; 88: 11–17.
Chang Q, Chen J, Beezhold KJ, Castranova V, Shi X, Chen F . JNK1 activation predicts the prognostic outcome of the human hepatocellular carcinoma. Mol cancer 2009; 8: 64.
Wang X, Chao L, Li X, Ma G, Chen L, Zang Y et al. Elevated expression of phosphorylated c-Jun NH2-terminal kinase in basal-like and 'triple-negative' breast cancers. Hum pathol 2010; 41: 401–406.
Yeh YT, Hou MF, Chung YF, Chen YJ, Yang SF, Chen DC et al. Decreased expression of phosphorylated JNK in breast infiltrating ductal carcinoma is associated with a better overall survival. Int j cancer 2006; 118: 2678–2684.
Zhang T, Inesta-Vaquera F, Niepel M, Zhang J, Ficarro SB, Machleidt T et al. Discovery of potent and selective covalent inhibitors of JNK. Chem biol 2012; 19: 140–154.
Kappelmann M, Bosserhoff A, Kuphal S . AP-1/c-Jun transcription factors: regulation and function in malignant melanoma. Eur j cell biol 2014; 93: 76–81.
Gee JM, Barroso AF, Ellis IO, Robertson JF, Nicholson RI . Biological and clinical associations of c-jun activation in human breast cancer. Int j cancer 2000; 89: 177–186.
Xia Y, Yang W, Bu W, Ji H, Zhao X, Zheng Y et al. Differential regulation of c-Jun protein plays an instrumental role in chemoresistance of cancer cells. J biol chem 2013; 288: 19321–19329.
Bartholomeusz C, Gonzalez-Angulo AM, Liu P, Hayashi N, Lluch A, Ferrer-Lozano J et al. High ERK protein expression levels correlate with shorter survival in triple-negative breast cancer patients. oncologist 2012; 17: 766–774.
Wang Y, Klijn JG, Zhang Y, Sieuwerts AM, Look MP, Yang F et al. Gene-expression profiles to predict distant metastasis of lymph-node-negative primary breast cancer. Lancet 2005; 365: 671–679.
Schmidt M, Bohm D, von Torne C, Steiner E, Puhl A, Pilch H et al. The humoral immune system has a key prognostic impact in node-negative breast cancer. Cancer res 2008; 68: 5405–5413.
Yoon CH, Kim MJ, Kim RK, Lim EJ, Choi KS, An S et al. c-Jun N-terminal kinase has a pivotal role in the maintenance of self-renewal and tumorigenicity in glioma stem-like cells. Oncogene 2012; 31: 4655–4666.
Chaterjee M, van Golen KL . Breast cancer stem cells survive periods of farnesyl-transferase inhibitor-induced dormancy by undergoing autophagy. Bone marrow res 2011; 2011: 362938.
Wang YJ, Bailey JM, Rovira M, Leach SD . Sphere-forming assays for assessment of benign and malignant pancreatic stem cells. Meth mol biol 2013; 980: 281–290.
Wojtaszek PA, Heasley LE, Siriwardana G, Berl T . Dominant-negative c-Jun NH2-terminal kinase 2 sensitizes renal inner medullary collecting duct cells to hypertonicity-induced lethality independent of organic osmolyte transport. J biol chem 1998; 273: 800–804.
Liu J, Lin A . Role of JNK activation in apoptosis: a double-edged sword. Cell res 2005; 15: 36–42.
Gururajan M, Chui R, Karuppannan AK, Ke J, Jennings CD, Bondada S . c-Jun N-terminal kinase (JNK) is required for survival and proliferation of B-lymphoma cells. Blood 2005; 106: 1382–1391.
Fujishita T, Aoki M, Taketo MM . JNK signaling promotes intestinal tumorigenesis through activation of mTOR complex 1 in Apc(Delta716) mice. Gastroenterology 2011; 140: 1556–1563 e1556.
Song W, Ma Y, Wang J, Brantley-Sieders D, Chen J . JNK signaling mediates EPHA2-dependent tumor cell proliferation, motility, and cancer stem cell-like properties in non-small cell lung cancer. Cancer res 2014; 74: 2444–2454.
Wang J, Kuiatse I, Lee AV, Pan J, Giuliano A, Cui X . Sustained c-Jun-NH2-kinase activity promotes epithelial-mesenchymal transition, invasion, and survival of breast cancer cells by regulating extracellular signal-regulated kinase activation. Mol cancer res 2010; 8: 266–277.
Gozdecka M, Lyons S, Kondo S, Taylor J, Li Y, Walczynski J et al. JNK suppresses tumor formation via a gene-expression program mediated by ATF2. Cell rep 2014; 9: 1361–1374.
Liu H, Deng X, Shyu YJ, Li JJ, Taparowsky EJ, Hu CD . Mutual regulation of c-Jun and ATF2 by transcriptional activation and subcellular localization. EMBO j 2006; 25: 1058–1069.
Sabapathy K, Jochum W, Hochedlinger K, Chang L, Karin M, Wagner EF . Defective neural tube morphogenesis and altered apoptosis in the absence of both JNK1 and JNK2. Mech dev 1999; 89: 115–124.
Liu J, Minemoto Y, Lin A . c-Jun N-terminal protein kinase 1 (JNK1), but not JNK2, is essential for tumor necrosis factor alpha-induced c-Jun kinase activation and apoptosis. Mol cell biol 2004; 24: 10844–10856.
She QB, Chen N, Bode AM, Flavell RA, Dong Z . Deficiency of c-Jun-NH(2)-terminal kinase-1 in mice enhances skin tumor development by 12-O-tetradecanoylphorbol-13-acetate. Cancer res 2002; 62: 1343–1348.
Capaccione KM, Pine SR . The Notch signaling pathway as a mediator of tumor survival. Carcinogenesis 2013; 34: 1420–1430.
Leong KG, Karsan A . Recent insights into the role of Notch signaling in tumorigenesis. Blood 2006; 107: 2223–2233.
Wang J, Sullenger BA, Rich JN . Notch signaling in cancer stem cells. Adv exp med biol 2012; 727: 174–185.
Dontu G, Jackson KW, McNicholas E, Kawamura MJ, Abdallah WM, Wicha MS . Role of Notch signaling in cell-fate determination of human mammary stem/progenitor cells. Breast cancer res 2004; 6: R605–R615.
Malhotra GK, Zhao X, Band H, Band V . Shared signaling pathways in normal and breast cancer stem cells. J carcinogen 2011; 10: 38.
Harrison H, Farnie G, Howell SJ, Rock RE, Stylianou S, Brennan KR et al. Regulation of breast cancer stem cell activity by signaling through the Notch4 receptor. Cancer res 2010; 70: 709–718.
Cantrell MA, Ebelt ND, Pfefferle AD, Perou CM, Van Den Berg CL . c-Jun N-terminal kinase 2 prevents luminal cell commitment in normal mammary glands and tumors by inhibiting p53/Notch1 and breast cancer gene 1 expression. Oncotarget 2015; 6: 11863–11881.
Tsuiki H, Tnani M, Okamoto I, Kenyon LC, Emlet DR, Holgado-Madruga M et al. Constitutively active forms of c-Jun NH2-terminal kinase are expressed in primary glial tumors. Cancer res 2003; 63: 250–255.
Ito M, Hiramatsu H, Kobayashi K, Suzue K, Kawahata M, Hioki K et al. NOD/SCID/gamma(c)(null) mouse: an excellent recipient mouse model for engraftment of human cells. Blood 2002; 100: 3175–3182.
Cheng L, Ramesh AV, Flesken-Nikitin A, Choi J, Nikitin AY . Mouse models for cancer stem cell research. Toxicol pathol 2010; 38: 62–71.
Acknowledgements
The MIGR1 vectors were generous gifts from Dr Khaled A. Hassan (University of Michigan, Ann Arbor, MI). We thank Naoko Matsuda of the Department of Breast Medical Oncology at MD Anderson for assistance with the RPPA data analysis. We acknowledge Stephanie P Deming of the Department of Scientific Publications at MD Anderson for editorial assistance and Wendy Schober, Nalini Patel and Duncan Mak of the Flow Cytometry and Cellular Imaging Facility at MD Anderson for assistance with cell cycle distribution and stem cell subpopulation analyses. This work was supported by MD Anderson Cancer Center Inflammatory Breast Cancer funds (105655) and Nylene Eckles funds (101478) to Naoto T Ueno; startup funds from MD Anderson Cancer Center (111411) to Chandra Bartholomeusz; Cancer Prevention and Research Institute of Texas (RP140648 and RP140649) to Kevin N Dalby; a Susan G Komen for the Cure Postdoctoral Fellowship to Xuemei Xie (KG111170); and National Institutes of Health Cancer Center Support Grant to MD Anderson Cancer Center (CA016672).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Xie, X., Kaoud, T., Edupuganti, R. et al. c-Jun N-terminal kinase promotes stem cell phenotype in triple-negative breast cancer through upregulation of Notch1 via activation of c-Jun. Oncogene 36, 2599–2608 (2017). https://doi.org/10.1038/onc.2016.417
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.417
This article is cited by
-
Aggresome formation promotes ASK1/JNK signaling activation and stemness maintenance in ovarian cancer
Nature Communications (2024)
-
MUC1-C intersects chronic inflammation with epigenetic reprogramming by regulating the set1a compass complex in cancer progression
Communications Biology (2023)
-
Targeting c-Jun inhibits fatty acid oxidation to overcome tamoxifen resistance in estrogen receptor-positive breast cancer
Cell Death & Disease (2023)
-
Lipocalin-2 inhibits pancreatic cancer stemness via the AKT/c-Jun pathway
Human Cell (2022)
-
Triptonide effectively inhibits triple-negative breast cancer metastasis through concurrent degradation of Twist1 and Notch1 oncoproteins
Breast Cancer Research (2021)