Abstract
R-spondin1 is a secreted regulator of WNT signaling, involved in both embryonic development and homeostasis of adult organs. It can have a dual role, acting either as a mitogen or as a tumor suppressor. During ovarian development, Rspo1 is a key factor required for sex determination and differentiation of the follicular cell progenitors, but is downregulated after birth. In human, increased RSPO1 expression is associated with ovarian carcinomas, but it is not clear whether it is a cause or a consequence of the tumorigenic process. To address the role of Rspo1 expression in adult ovaries, we generated an Rspo1 gain-of-function mouse model. Females were hypofertile and exhibited various ovarian defects, ranging from cysts to ovarian tumors. Detailed phenotypical characterization showed anomalies in the ovulation process. Although follicles responded to initial follicle-stimulating hormone stimulation and developed normally until the pre-ovulatory stage, they did not progress any further. Although non-ovulated oocytes degenerated, the surrounding follicular cells did not begin atresia. RSPO1-induced expression not only promotes canonical WNT signaling but also alters granulosa cell fate decisions by maintaining epithelial-like traits in these cells. This prevents follicle cells from undergoing apoptosis, leading to the accumulation of granulosa cell tumors that reactivates the epithelial program from their progenitors. Taken together, our data demonstrate that activation of RSPO1 is sufficient in promoting ovarian tumors and thus supports a direct involvement of this gene in the commencement of ovarian cancers.
Similar content being viewed by others

Introduction
WNT/β-catenin (CTNNB1) signaling is involved in numerous biological processes from embryogenesis to stem cell activation. Deregulation of this pathway is responsible for many diseases, most notably colorectal cancer. Indeed, 90% of colorectal cancers result from APC mutations leading to the activation of this pathway.1 WNT signaling is tightly controlled by negative and positive modulators like APC and R-spondins (RSPO), a family of four members of secreted proteins. Fusions of RSPO2 and RSPO3 activate WNT signaling in colorectal cancers,2 and amplifications of RSPO1 have been associated with 8% of ovarian epithelial cancers.3, 4 However, functional evidence for a direct involvement of RSPO1 in ovarian tumor development is still missing.
Depending on the cell type, Rspo1 may act either as a mitogen or a tumor suppressor:5, 6 variants of the RSPO1 locus have been reported in ovarian cancers highlighting its role as an oncogene.3, 4 By contrast, RSPO1 loss-of-function mutations predispose to squamous cell carcinoma.6 On the molecular level, RSPO1 binds to the recruitment receptors LGR4, LGR5 or LGR6 leading to stabilization of CTNNB1, and in turn CTNNB1 interacts with transcription factors such as LEF/TCF in the nucleus and trans-activates target genes such as Axin2 (for a model, see ref. 7).
CTNNB1 is not only a key factor of the WNT signal-transduction pathway, but is also a component of adherens junctions linking cadherins via α-catenin to the actin cytoskeleton at the plasma membrane of epithelial cells.8 The membrane function of CTNNB1 can be partially substituted by the related protein JUP (Plakoglobin/γ-catenin), an important component of desmosomes that anchors epithelial cells to intermediate filaments and can also act as a signaling molecule at least in vitro.9, 10 Several studies support a role for JUP in cancer.11 Although a direct link between RSPO1 and JUP has not yet been established, skin cells from patients affected by RSPO1 loss-of-function mutations exhibit defects reminiscent of desmosome abnormalities.6
In addition to the skin diseases, such as palmoplantar hyperkeratosis, patients carrying mutations in RSPO1 exhibit female-to-male sex reversal, and predisposition to squamous cell carcinoma and seminoma.6, 12 Using functional analysis in mice, the role of RSPO1 and canonical WNT signaling has been unraveled in female ovarian development.13, 14, 15 Prior to sex determination, Rspo1 is involved in the proliferation of progenitor cells that are located within the surface epithelium of the undifferentiated gonads.16, 17 Following sex determination, cells of the ovarian surface epithelium (OSE) continue to proliferate and enter the gonads to give rise to precursors of the granulosa cells.18, 19 At this stage, Rspo1 is required to maintain granulosa precursors in the undifferentiated state.14, 20 Accordingly, LGR4- and LGR5-expressing precursor cells contribute to the formation of the follicle pool from which follicles are recruited during reproductive life.21 In mice, Rspo1 expression becomes significantly downregulated in the first week post partum,22 suggesting that RSPO1 is not required for the recruitment and maturation of follicles, and consequently proper ovarian functioning.
From the onset of puberty, the ovary undergoes successive cycles of follicular recruitment, maturation, OSE exfoliation, subsequent ovulation and corpus luteum (CL) formation.23 Progesterone synthetized by the CL is necessary to prepare the uterine wall for implantation. LGR5-positive OSE cells are presumed to contribute to both post-ovulatory repair of the OSE24 and, together with LGR4-positive lutein cells, progesterone production.25, 26 Activation of canonical WNT signaling must therefore be tightly controlled in the adult ovary. Consequently premature activation of canonical WNT signaling in differentiating granulosa cells leads to the appearance of precancerous follicle-like lesions around 6 weeks post partum, and 57% of these become granulosa cell tumors (GCTs) before the age of 7.5 months.27
In women, ovarian cancers derive from three major histotypes: surface epithelia, sex-chord stromal cells and germ cells.28 Approximately 3% of ovarian cancers are derived from germ cells, 7% come from sex-chord stromal cells and 90% arise from OSE or fallopian tube epithelium.28 GCTs are categorized into two distinct subtypes, the juvenile and the adult form.29 Although less common than OSE-derived cancers in women, GCT frequently occurs in primates and domestic animals.28 Moreover, in mouse models adult granulosa cells originate from two distinct waves of migration of progenitor cells of the OSE.18 Given that (i) ectopic activation of canonical WNT signaling within the ovary has oncogenic effects,27 (ii) RSPO1 is an activator of canonical WNT signaling during ovarian development14 and (iii) RSPO1 is amplified in ovarian cancers,3 we hypothesized that RSPO1 misexpression could be a cause of ovarian defects and eventually cancer development. Using a gain-of-function mouse model (Sf1:CreTg/+; R26Rspo1/+), we show that post partum maintenance of Rspo1 expression in somatic cells of the mouse ovary impairs homeostasis of the healthy ovary.
Results
Rspo1 expression is downregulated in adult follicles
RSPO1/Rspo1 is highly expressed in somatic cells of the ovary during embryogenesis.6, 30 In the 2 weeks following birth, however, Rspo1 expression is downregulated as evidenced by qRT–PCR analysis.22 To obtain a more detailed expression profile and identify the cell types that express Rspo1 late in development and during the postnatal period, we performed in situ hybridization experiments. Strong expression of Rspo1 was found within the fetal ovary at 16.5 days post coitum (dpc), which significantly decreased after birth and remained at low levels in granulosa cells of cortical follicles in adult ovaries (Figures 1a–c). In contrast, Rspo1 was robustly expressed in the OSE at all stages (Figures 1a–c). To understand whether RSPO1 was able to activate canonical WNT signaling, we next analyzed whether the Axin2+/LacZ reporter of this pathway31 was expressed in the adult ovary. β-Galactosidase staining was detected in the OSE and in the ovarian stroma as well as in oocytes (Figures 1e–g). By contrast, granulosa cells were weakly stained after birth, indicating that RSPO1/WNT signaling is downregulated in this compartment of the follicle.
Pattern of Rspo1 expression and activation of WNT signaling in ovaries. Expression of Rspo1 revealed by in situ hybridization in R26Rspo1/+mouse ovaries at 16.5 dpc (a), 4 dpp (b) and 12 months (c). Rspo1 is expressed at low level in the R26Rspo1/+mouse adult ovary, mainly in the epithelial surface and in granulosa cells of some follicles in the ovarian cortex, whereas adult Sf1-CreTg/+; R26Rspo1/+ovaries display Rspo1 expression throughout the whole organ (d). Activation of the canonical WNT signaling by RSPO1 visualized by β-galactosidase staining of ovaries expressing the Axin2-LacZ reporter. Axin2-LacZ is expressed at 2.5 and 12 months in pre-antral follicles and the stroma (e and g) and strongly expressed in oocytes and the epithelial surface surrounding the ovary (e and f, arrow) in R26Rspo1/+mice. Induced Rspo1 expression leads to Axin2-LacZ expression in the whole Sf1-CreTg/+; R26Rspo1/+ovary (h) with strong staining in granulosa cells (arrow) and weaker staining in aberrant follicular structures lesions (*). Scale bars: 300 μm.
Increased RSPO1 expression activates canonical WNT signaling in adult ovaries
To assess the importance of Rspo1 repression in the postnatal ovary, we next investigated the effect of induced Rspo1 expression on ovarian homeostasis. To this end, we generated Sf1-CreTg/+; R26Rspo1/+ mice (Supplementary Figure S1). We first established the ability of female Sf1-CreTg/+; R26Rspo1/+ mice to ectopically express Rspo1 throughout the ovary (Figures 1c and d). Next, we assessed that induced expression of Rspo1 led to a strong, although variable, follicular activation of canonical WNT signaling by detecting β-galactosidase in ovaries from Sf1-CreTg/+; R26Rspo1/+; Axin2+/LacZ mice (Figures 1g and h). This confirmed that sustained expression of Rspo1 activates canonical WNT signaling in ovaries.
Rspo1 activation induces ovarian defects, subfertility and cancer
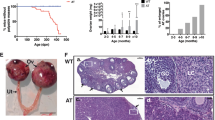
To evaluate the effect of RSPO1 expression on ovarian function we first assessed the fertility of Sf1-CreTg/+; R26Rspo1/+ female mice. Litter sizes were significantly reduced with only 1.25 pup/female (±1.58) compared with 7.25 pups/female (±1.16) for R26Rspo1/+ littermates (n=8 plugged female per genotype). By the age of 6 months double-transgenic mice were all sterile (n=8). Macroscopic observation of dissected ovaries revealed a marked increase in the size of mutant ovaries when compared with those of the control littermates (Figures 2a–c). The size of the transgenic ovaries increased over time (data not shown).
Ovarian defects and tumors in Sf1-CreTg/+; R26Rspo1/+ovaries. Macroscopic view of 18-month-old R26Rspo1/+ (a) and Sf1-CreTg/+; R26Rspo1/+ovaries (b and c). The size of the Sf1-CreTg/+; R26Rspo1/+ovaries is increased in comparison with the control ovary owing to the presence of prominent tumor tissue (c) and/or blood-filled cysts (b). Magnification 10 × . Histological sections (haematoxylin and eosin staining) analysis of 12-month-old ovaries. Sf1-CreTg/+; R26Rspo1/+ovaries (e) contain numerous multifocal aggregates of neoplastic cells that arrange either in discrete solid aggregates (*) or in microfollicular pattern (arrows) in comparison with R26Rspo1/+ovaries (d). Scale bars, 500 μm. Magnifications of structural abnormalities in Sf1-CreTg/+; R26Rspo1/+ovaries (f–n): neoplastic cells arranged in microfollicular or rosette-like pattern (f; *) with central pale eosinophilic globule (Call–Exner body; f and g arrow); neoplastic cells arranged in solid sheets composed of bland, ovoid, scant amount of cytoplasm and ovoid nuclei granulosa cells. (h, arrow): hematocysts (i and j), with columnar cells (j, arrow), CL or regressing CL with erythrocytes infiltration (l and m, arrow), Ovarian epithelial cyst composed of cystic and papillary structures lined with tall columnar ciliated epithelium. (k, arrow) and ovarian epithelial inclusion cyst composed of keratinized squamous epithelium (n, arrow). Scale bars: 100 μm. a, antral follicle; cl, corpus luteum.
To gain insides into the ovarian phenotype, we examined 170 haematoxylin and eosin-stained slides containing serial sections from 14 murine ovaries from Sf1-CreTg/+; R26Rspo1/+ mice. Histological analysis revealed a variety of abnormalities, ranging from 100% penetrance of dense follicle-like lesions to tumors in about 10% of ovaries (Figure 2). The ovarian tissue presented multifocal aggregates of neoplastic cells that arrange either in discrete solid aggregates, solid sheets or in microfollicular pattern (Figures 2f–h) reminiscent to granulosa cells. They are bland, ovoid, have distinct borders, scant amount of cytoplasm and ovoid nuclei. On the basis of these observations, we conclude that GCTs accumulate in Sf1-CreTg/+; R26Rspo1/+ mouse ovaries. None of these abnormalities were detected in control ovaries (Figure 2d). A proportion of these follicle-like structures developed into blood-filled cysts (Figures 2i and j) surrounded by cuboidal cells (Figure 2j) that were never observed in ovaries from control animals. In addition, fewer corpora lutea (CLs) were observed which is in agreement with the reduced fertility of the Sf1-CreTg/+; R26Rspo1/+ females (Figure 2e). A proportion of regressing CLs contained erythrocytes infiltration (Figures 2l and m). One-year-old animals carried bigger cysts (Figures 2k and n), which, in some cases, contained layered keratinocytes in the cavity (Figure 2n). In addition to the accumulation of GCT, solid tumors were observed in 12–18-month-old females with 1 tumor/13 Sf1-CreTg/+; R26Rspo1/+ and 3/19 Sf1-CreTg/+; R26Rspo1/Rspo1 females, whereas 0/22 control littermates were scored (Figure 2c). The severity of the different defects varied between animals and was not linked to whether they were heterozygous or homozygous for the Rosa26-Rspo1 transgene.
We next analyzed whether GCTs were proliferative or undergoing apoptosis in 40 days post partum (dpp) Sf1-CreTg/+; R26Rspo1/+ ovaries using immunostaining for MKI67 and active CASP3, respectively, two markers of these processes. Only occasionally, we observed a cell expressing one of these markers in the GCT, implying that cell proliferation and apoptosis were negligible in these structures (Supplementary Figures S2A–D). Interestingly, increased proliferation was detected in the stromal compartment of Sf1-CreTg/+; R26Rspo1/+ ovaries (Supplementary Figures S2A and B). Consistent with this proliferative activity and clear staining for the theca cell marker SF1 (Supplementary Figures S2E and F), the theca cell layer seems to be expanded in size as indicated by SF1 staining (Supplementary Figure S2F).
We conclude that Rspo1 acts as an oncogene in the ovary because its upregulation leads to ovarian tumor development.
Ovarian defects induced by RSPO1 expression occur from the onset of puberty
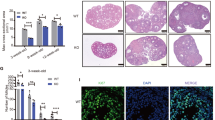
We next examined the development of the ovarian phenotype in Sf1-CreTg/+; R26Rspo1/+ mice before and soon after pubertal age. Immunostaining revealed that follicle-like structures are composed of FOXL2-positive granulosa cells (Figures 3e and f) indicating they are indeed aberrant follicles. To investigate when these abnormal follicular structures first appear, we analyzed histological sections at different stages. Pre-pubertal ovaries (21 dpp) appeared normal and no follicular lesions were detected (Figures 5a and b). The first phenotypic changes became apparent in 28 dpp ovaries (n=6; Figures 3a and b). At this age most of the follicles contained an oocyte. By 40 dpp, serial histological sections analysis revealed an increased abundance of aberrant follicular structures with the majority of follicles being devoid of oocytes (Figures 3c and d). To further investigate the status of the aberrant follicles, we stained sections of ovaries from 40 dpp mice for AMH (anti-Müllerian hormone), a specific marker of early granulosa cells in primary to early antral follicles.32 AMH-positive follicles were observed at this stage in Sf1-CreTg/+; R26Rspo1/+ mice, however, the abnormal follicular structures were negative for this marker (Figures 3g and h). Vimentin (VIM), a marker for mural granulosa cells of antral follicles, was expressed in both R26Rspo1/+and Sf1-CreTg/+; R26Rspo1/+ ovaries (Figures 3i and j) and was also readily detectable in aberrant follicles (Figure 3j). Thus, GCT in Sf1-CreTg/+; R26Rspo1/+ ovaries originate from maturing antral follicles and appear around the onset of puberty, suggesting that their formation is sensitive to gonadotrophins, two specifications of adult GCT.29
RSPO1 promotes GCT. Histological analysis (haematoxylin and eosin staining) of R26Rspo1/+ and Sf1-CreTg/+; R26Rspo1/+ ovaries at 28 dpp (a and b) and 40 dpp (c and d). Follicle-like structures (*) consisting of nests of disorganized, pleiomorphic granulosa cells become evident at 28 dpp (b) in Sf1-CreTg/+; R26Rspo1/+ ovaries, whereas they are not observed in R26Rspo1/+ovaries (a). They enclose oocytes at 28 dpp (b), whereas they lack an antrum and contain no visible or a degenerating oocyte at 40 dpp (d). Scale bar: 300 μm. Immunolabelling of FOXL2 (e and f), AMH (g and h) and Vimentin (VIM) (i and j) in R26Rspo1/+ (left panel) and Sf1-CreTg/+; R26Rspo1/+ ovaries (right panel) at 40 dpp. FOXL2 staining in the GCT (f; *) confirms the presence of granulosa cells in these follicle-like structures in R26Rspo1/+ovaries (e). The absence of AMH staining (h) and the presence of VIM staining (j) in GCT (*) suggest that follicular maturation can occur up to the antral stages. Nuclei are labeled with DAPI (blue). Scale bars: 100 μm. a, antral follicle; pa, pre-antral follicle, cl, corpus luteum.
Sf1-CreTg/+; R26Rspo1/+ ovaries respond to hormonal stimulation. Histological analysis (haematoxylin and eosin staining) of R26Rspo1/+ (a, c and e) and Sf1-CreTg/+; R26Rspo1/+ (b, d, f and g) ovaries of 21 dpp (a and b) and at 21–24 dpp stimulated by PMSG injection and collected 48 h later (c–g). When stimulated by PMSG, ovaries from both genotypes contain large antral (a) follicles (c and d) but in Sf1-CreTg/+; R26Rspo1/+ mice, most of these follicles exhibit dissymmetric mural granulosa (f and g, arrowheads) when compared with follicles of R26Rspo1/+ ovaries (e). Aberrant follicles are also observed containing naked (*) or degenerating (**) oocytes (g). Scale bar: 300 μm (a–d) and 100 μm (e–g).
RSPO1 expression impairs ovulation leading to the formation of GCT
As the Sf1-Cre transgene is also expressed in the pituitary that produces multiple hormones and especially growth hormone, we examined Sf1-CreTg/+; R26Rspo1/+ pups. Female and male mice were healthy and their body size was normal (n=6; data not shown), indicating that activating Rspo1 mutations do not hamper the development and functions of this organ. We next investigated serum levels of gonadotropins, estradiol and progesterone in adult Sf1-CreTg/+; R26Rspo1/+ females (Figure 4a). Luteinizing Hormone (LH) and progesterone levels were considerably lower in Sf1-CreTg/+; R26Rspo1/+ mice, compared with R26Rspo1/+females. This observation was consistent with the low level of LH receptors observed in the ovaries (Figures 4c and e) and the lack of corpora lutea (Figure 2e). In contrast, follicle-stimulating hormone (FSH) levels were three times higher in Sf1-CreTg/+; R26Rspo1/+ mice (Figure 4a).
Induced Rspo1 expression impairs LH-mediated luteinization. (a) Serum levels of FSH, LH, estradiol and progesterone in R26Rspo1/+ (black bars) and Sf1-CreTg/+; R26Rspo1/+ ovaries (white bars) from adult females (n=5 for each genotype). Bars represent mean±s.e.m. with significance set at P<0.05 (*). Expression of LH receptor revealed by in situ hybridization in adult (P40) R26Rspo1/+and Sf1-CreTg/+; R26Rspo1/+ovaries (b–e). Lhr is strongly expressed in mural granulosa cells of mature antral (a) follicles (b and d) and in the theca cells of R26Rspo1/+ovaries, whereas Lhr expression is clearly downregulated in antral follicles (c and e) of Sf1-CreTg/+; R26Rspo1/+ ovaries and is absent from GCT (*). Scale bars: 100 μm. ns, not significant.
In laboratory mouse strains, puberty occurs between 30–40 dpp.33 Although GCT accumulation arises at the onset of puberty, we hypothesized that gonadotropins are involved in GCT formation in Sf1-CreTg/+; R26Rspo1/+ ovaries. Thus, we stimulated follicular maturation by injecting PMSG to pre-pubertal females (21–24 dpp). Forty-eight hours after PMSG administration, numerous large antral follicles in both Sf1-CreTg/+; R26Rspo1/+ and R26Rspo1/+ ovaries were observed (Figures 5c and d), whereas they were absent in the untreated females (Figures 5a and b). This indicates that RSPO1 does not impair follicular growth upon FSH stimulation. However, most of these large antral follicles in Sf1-CreTg/+; R26Rspo1/+mice exhibited structural abnormalities ranging from asymmetric repartition of granulosa cells to naked or abnormal oocytes (Figures 5c–g) as described in ref. 34.
In order to test whether the antral follicles present in PMSG-treated pre-pubertal Sf1-CreTg/+; R26Rspo1/+ females are competent for ovulation, superovulations were induced by injecting human chorionic gonadotropin (hCG) 48 h after PMSG administration. Ovulation takes place about 12 h post-hCG injection. We first examined the expression of Fshr and Foxo1, two factors involved in follicular development, 4 h after LH/hCG injection and found no significant differences between the two genotypes (Figure 6a). Twenty hours after hCG injection, control ovaries were mainly constituted by post-ovulatory CLs, but none or only a few were identified in Sf1-CreTg/+; R26Rspo1/+ females (Figures 6b and c). Strikingly, in Sf1-CreTg/+; R26Rspo1/+ ovaries, follicular lesions began to emerge and many large antral follicles were still apparent, some of which still contained oocytes (Figure 6c), suggesting that ovulation did not process efficiently. To confirm this observation, we quantified ovulated oocytes in superovulated females. Indeed, the number of ovulated oocytes was significantly reduced in Sf1-CreTg/+; R26Rspo1/+ females (Figure 6d) and when ovulated, the oocytes were devoid of a cumulus oophorus (data not shown). Moreover, we observed a dramatic reduction of the level of expression of Amphiregulin (Areg) and Epiregulin (Ereg), two EGF-like factors involved in cumulus cell-oocyte complex expansion and thus required for ovulation (Figure 6a), which could at least partially explain the improper ovulation phenotype in Sf1-CreTg/+; R26Rspo1/+ females. The lack of CLs in Sf1-CreTg/+; R26Rspo1/+ ovaries (Figure 6c) suggests that RSPO1 expression impairs the terminal differentiation of antral follicles. Indeed, the expression of Lhr, Cyp11a1 and Star, key genes of luteinization process of the granulosa cells which occurs upon LH/hCG surge, was significantly downregulated in Sf1-CreTg/+; R26Rspo1/+ (Figure 6a). As a result, although R26Rspo1/+ ovaries were filled with CLs expressing CYP11A1, only few CYP11A1-positive CL were observed in Sf1-CreTg/+; R26Rspo1/+ ovaries (Figures 6f and g). Interestingly, the newly formed follicular lesions were devoid of CYP11A1 (Figure 6g). In addition, 21–24 dpp superovulated Sf1-CreTg/+; R26Rspo1/+ ovaries contained aberrant follicles including degenerating oocytes as observed in 40 dpp non-superovulated Sf1-CreTg/+; R26Rspo1/+ ovaries (Figures 3d and 6e), suggesting that follicular lesions become apparent in the later stages of follicular development.
Induced Rspo1 expression prevents ovulation. Quantitative RT–PCR analysis of Fshr, Foxo1, Areg, Ereg, Lhr, Cyp11a1 and Star expression in R26Rspo1/+ (black bars) and Sf1-CreTg/+; R26Rspo1/+ ovaries (white bars) from 21–24 dpp superovulated mice, using Sdha1 as the normalization control (a). Bars represent mean±s.e.m. (n=6 for each genotype) with significance set at P<0.05 (*), P<0.01 (**) and P<0.001 (***). Histological analysis (haematoxylin and eosin staining) of R26Rspo1/+ and Sf1-CreTg/+; R26Rspo1/+ ovaries at 21–24 dpp stimulated by hCG (b, c and e). R26Rspo1/+ ovaries (b) contain many corpora lutea (cl) which are rarely observed in Sf1-CreTg/+; R26Rspo1/+ ovaries (c). Instead, these ovaries display several GCT (*) and naked oocytes (arrowhead) in antral (indicated as a) follicles (c) and degenerating follicles (arrowhead) (e). Superovulation experiments show a reduced number of ovulated oocytes (d) with an average of 8 and 0.6 oocyte/ovary (n=8–10) in R26Rspo1/+ and Sf1-CreTg/+; R26Rspo1/+ females, respectively. CYP11A1 revealed by immunostaining is highly expressed in CL of R26Rspo1/+ ovaries (f) but absent in the abnormal follicles (*) of Sf1-CreTg/+; R26Rspo1/+ ovaries (g). Nuclei are labeled with DAPI (blue). Scale bars: 100 μm.
Altogether our results demonstrate that follicular abnormalities observed in Sf1-CreTg/+; R26Rspo1/+ ovaries are driven by puberty, that Sf1-CreTg/+; R26Rspo1/+ ovaries are unable to properly respond to LH and that GCT formation is linked to FSH stimulation. These results show that superovulation experiments induce and accelerate the formation of GCT and that Rspo1 downregulation within adult ovary is required for the proper occurrence of the ovulation process and the subsequent formation of CLs.
Induced expression of RSPO1 promotes cell junction adherence and epithelial traits in GCT
Next, we searched for an explanation for the persistence of these abnormal follicular structures. RSPO1 is typically associated with canonical WNT signaling as corroborated by the expression of the Axin2+/LacZreporter31 (Figures 1g and h). However, CTNNB1 is also a component of adherens junctions, where it associates with cadherins (CDH).8, 35 Maintaining adherence junctions has been shown previously to inhibit apoptosis of granulosa cells thus preventing follicular degeneration.36 Therefore, the presence of adherens junctions was investigated in ovaries of 6-month-old Sf1-CreTg/+; R26Rspo1/+ and R26Rspo1/+ mice by immunostainings using the markers CTNNB1 and cell-adhesion molecule N-cadherin (CDH2). CTNNB1 was mostly detected at the membrane of granulosa cells of pre-antral follicles and in granulosa cells surrounding the antrum in larger antral follicles in control ovaries (Figures 7a and c). CTNNB1 was enriched at the membrane of the cells in the GCT in Sf1-CreTg/+; R26Rspo1/+ ovaries (Figures 7b and d). The presence of CDH2 was evident at all stages during maturation in both genotypes (Supplementary Figures S3A, B). GCT also strongly expressed membrane-bound plakoglobin (JUP), a component of desmosomes and intermediate junctions (Figures 7e and f), and the epithelial markers KRT18 and pan-cytokeratin (Figures 7g and h and Supplementary Figures S3C–F) that are absent or weakly expressed in follicles in control ovaries. These data reveal the presence of strong intercellular junctions in GCT in Sf1-CreTg/+; R26Rspo1/+ovaries, indicating that induced expression of Rspo1 increases cellular adhesion, which in turn may promote epithelialisation of granulosa cells and the persistence of the follicular remnants.
RSPO1 expression maintains intercellular junctions and epithelial identity in GCT. Immunostaining for CTNNB1 (a–d), JUP (e and f), and KRT18 (g and h) in 6-month-old R26Rspo1/+ (left panel) and Sf1-CreTg/+; R26Rspo1/+ (right panel) ovaries. Membrane staining for CTNNB1 and JUP underline cellular junctions in these follicle-like structures (h). Nuclei are labeled with DAPI (blue). Scale bars: 100 μm.
Discussion
Although RSPO1 is crucial for ovarian development,14 here we show that its postnatal downregulation is critical for the maintenance of a healthy adult ovary. When RSPO1 expression is maintained, GCT appear at the onset of puberty in 100% of the cases. Prolonged RSPO1 expression allows follicular maturation until the late antral stage. Most follicles are capable of responding to FSH stimulation as previously reported for induced CTNNB1 expression,37 and fail to respond to LH. It has been shown that FSH increases the occurrence and onset of GCT formation.38 These GCT are classified as adult GCT.29 Accordingly, we observed high FSH levels that could contribute to the adult GCT formation in Sf1-CreTg/+; R26Rspo1/+ mice.
Endocrine functionality of CL involves LGR receptors.25, 26 However, neither Rspo1 nor the CTNNB1 target Axin2 is expressed in these structures, which suggests that LGRs may act through an RSPO1/CTNNB1 independent pathway in this cell type. In contrast, RSPO1, stabilized CTNNB1 and LGR5 are expressed in the OSE stem cells that contribute to post-ovulatory repair,24 putting forward that these factors act together in the ovulation process. However, the ovulatory defects observed in our model prevented us from analyzing whether induced Rspo1 affects CL physiology and post-ovulatory OSE repair.
In addition to the granulosa cell phenotype, we observed an enriched population of SF1-positive theca cells suggesting a role of CTNNB1 signaling in differentiation of steroidogenic lineages. The steroidogenic precursors of adrenal and gonadal cells have a common origin39 and both arise from a pool of WT1-positive cells.40, 41 In adrenals, CTNNB1 is required for the formation and maintenance of the steroidogenic cells of the adrenal cortex,42, 43 but the role of this signaling pathway in the fate of the theca cells has not been investigated to date.
Ablation of Rspo1 induces a precocious differentiation of granulosa cells during fetal development.20 This demonstrates that Rspo1 is required to maintain an undifferentiated state of the granulosa cell progenitors during early development, before Rspo1 expression decreases within the ovary around birth. Here, we show that when Rspo1 expression is maintained, it alters the differentiation of granulosa cells via their epithelialization. First, CTNNB1 and JUP are maintained at the plasma membrane of aberrant follicular structures and contribute to cell junction formation. Although follicular atresia is promoted by oocytes disruption,34 the maintenance of intercellular junctions can prevent degeneration of the granulosa cells.35 This suggests that stabilization of cell–cell contacts promote survival of the GCT in Sf1-CreTg/+; R26Rspo1/+mice. Interestingly, in a granulosa cell dominant-stable CTNNB1 mutant mouse model, GCTs develop via a mechanism that mainly implies canonical WNT signaling.44 Here, we show that RSPO1 activation in adult ovaries induces formation of GCTs by regulating not only canonical WNT signaling activation but also intercellular junction homeostasis in granulosa cells.
Second, GCT exhibit not only granulosa cell identity as highlighted by FOXL2 and Vimentin expression, but also epithelial cell traits are reactivated as shown by the concomitant expression of epithelial markers like pan-cytokeratin and KRT18. Granulosa cells originate from surface epithelial progenitors.45 Our results suggest that adult granulosa cells maintain the potential to reactivate their epithelial program if properly stimulated, and that RSPO1 is sufficient to induce this program. Moreover, FOXO1/3 and PTEN factors mutations in granulosa cells also promoted epithelial traits reactivation in GCTs.38
The mixed identity of GCT as granulosa cells and epithelial progenitors is likely a susceptibility factor for ovarian pathologies including ovarian cancers. Indeed, our results evidence that after-birth maintenance of RSPO1 expression induces GCT but also severe ovarian pathologies and eventually cancers in mice. In human RSPO1 has been reported as a susceptibility factor of ovarian epithelial cancers and is amplified in 8% of them,3, 4 but so far its exact role in ovarian cancers was not clear. Here, we show for the first time that the overexpression of RSPO1 in mouse ovarian tissues is sufficient to lead to GCT formation with 100% penetrance. Our results highlight RSPO1 as a main player in the ovarian oncogenic process and provide new insights into the molecular roles of RSPO1 in the pathology.
Materials and methods
Mouse strains and genotyping
The experiments were carried out in compliance with the relevant institutional and French animal welfare laws, guidelines and policies and have been approved by the French ethics committee (CIEPAL: NCE/2011-12).
Rspo1 gain-of-function model (Supplementary Figure S1) carries an inducible Rspo1 gene knocked into the Rosa26 locus. The construct is similar to the one reported in ref. 46 with the exception of the promoter. Here, the Rosa26 promoter controls Rspo1 expression. R26Rspo1/+ mice were mated with the Sf1-Crehigh (Sf1-CreTg/+) strain, which drives expression of the Cre recombinase in somatic cells of the gonad from 11.5 dpc onward.47 Axin2-LacZ mice were described previously,31 and crossed with Sf1-CreTg/+; R26Rspo1/+ males to generate Sf1-CreTg/+; R26Rspo1/+; Axin2+/LacZ animals. Fertility tests were performed using 3–4-month-old Sf1-CreTg/+; R26Rspo1/+ females mated with R26Rspo1/+ males (Supplementary Figure S1). Genotyping was performed using DNA extracted from tail tips or ear biopsies of mice. The presence of the Y chromosome and the Axin2-LacZ transgene was determined as described previously.14
Histological analysis
Ovaries were dissected, fixed in Bouin’s solution overnight, and embedded in paraffin. Five-micrometer-thick sections were stained with hematoxylin and eosin. To determine the ovarian phenotype in Rspo1 expressing ovaries, consecutive sections were analyzed. Pictures were taken with an Axioscope 2 (Zeiss, Oberkochen, Germany) or MZ9.5 (Leica, Mannheim, Germany) microscope coupled with an Axiocam MRc5 (Zeiss) or DHC490 (Leica) camera and Axiovision 4.8 (Zeiss) or application suite V3.3.0 (Leica) software, and processed with Adobe Photoshop (San Jose, CA, USA).
Serum collection and hormone assays
Blood was collected from adult mice in the same stage of estrous cycle and allowed to clot for 90 min at room temperature. Samples were then centrifuged and the serum was collected and stored at −20 °C. Hormone analyses were performed by the Ligand Assay and Analysis Core Laboratory at The Center for Research and Reproduction, University of Virginia (Charlottesville, VA, USA). LH, FSH, estradiol and progesterone measurements were run in duplicates using specific assays (n=5 for each genotype).
Superovulations
Immature 3–4-week-old mice were injected intraperitoneally (i.p.) with 5 IU/mouse pregnant mare serum gonadotropin (PMSG) (Sigma-Aldrich, St Louis, MO, USA) to promote follicle maturation, followed by 5 IU/mouse hCG (Sigma-Aldrich) 48 h later to induce ovulation. Mice were humanely killed 48 h after the first hormone injection for follicle maturation studies or 4 h and 20 h after the second injection for ovulation and luteinization studies. Ovaries, including fat pad, oviduct and partial uterine tube, were fixed in 4% paraformaldehyde or Bouin’s solution overnight. For qPCR analyses, ovaries were carefully isolated from the genital tract and quickly frozen in liquid nitrogen. For oocyte counting, immature females (n=8 (R26Rspo1/+) and 10 (Sf1-CreTg/+; R26Rspo1/+) ovaries) were similarly superovulated and killed 20 h after the hCG injection. Oviducts were isolated from ovary and uterus and opened to collect the oocytes.
X-gal staining and immunological analyses
Tissue samples were fixed in 4% paraformaldehyde overnight and then processed for paraffin embedding or equilibrated in sucrose and embedded in Cryomount (Histolab, Göteburg, Sweden) for cryosectioning. Cryostat or microtome sections of 10 μm and 5 μm thickness respectively were processed for X-Gal and immunostaining. Samples for X-Gal staining were processed as described previously.14
Immunohistochemical experiments were performed as described in.14 The following dilutions of primary antibodies were used: AMH (C-20, cat sc6886, Santa Cruz Biotechnology, Dallas, TX, USA) 1:200, CTNNB1 (cat 610153, BD Biosciences, San Jose, CA, USA) 1:250, active CASP3 (cat AF835, R&D Systems) 1:200, CDH2 (cat 33-3900, Invitrogen) 1:200, CYP11A148 1:300, FOXL221 1:400, JUP (cat 610254, BD Biosciences) 1:250, KRT18 (cat ab52948, Abcam, Cambridge, UK) 1:300, LHR (LHR-29, ATCC-CRL-2685, ATCC) 1:150, MKi67 (clone SP6, cat 9106, Thermo Fisher Scientific, Waltham, MA, USA) 1:200, SF1 (kindly provided by Professor Morohashi) 1:1000 and Vimentin (cat ab92547, Abcam) 1:200. Slides were counterstained with DAPI diluted in the mounting medium at 10 μg/ml (Vectashield, Vector laboratories, Burlingame, CA, USA) to detect nuclei. Imaging was performed with a motorized Axio ImagerZ1 microscope (Zeiss) coupled with an Axiocam Mrm camera (Zeiss) and processed with Axiovision LE (Zeiss) and Adobe Photoshop.
In situ hybridization
Tissue samples were fixed in 4% paraformaldehyde in 1 × PBS overnight at 4 °C. Seven-micrometer sections were cut for in situ hybridization experiments using Rspo1 and Lhr digoxigenin-labeled riboprobes as described previously.14 Each experiment was repeated on at least two gonads. Post-hybridization washes were done in 100 mM maleic acid Ph7.5, 150 mM NaCl, 0.1% (v/v) tween-20 (MABT). Imaging was performed on a MZ9.5 microscope (Leica) as described above and processed with Adobe Photoshop.
Quantitative PCR analysis
Individual ovaries from either adult or 21–24 dpp superovulated mice were dissected in 1 × PBS and quickly frozen in liquid nitrogen. RNA was extracted using the RNeasy Qiagen kit and reverse transcribed using the RNA RT–PCR kit (Stratagene). Primers and probes were designed by Roche Assay Design Center (http://qpcr.probefinder.com/organism.jsp). Primer sequences are available upon request to the authors. All real-time PCR assays were carried out using LightCycler Taqman Master kit (Roche, Basel, Switzerland). QPCR was performed on cDNA from one gonad and compared with a standard curve. Relative expression levels of each gene was determined in the same run and normalized on the levels of endogenous Sdha1 cDNA. QPCR were repeated at least twice. For adult mice, n=4 (R26Rspo1/+) and 8 (Sf1-CreTg/+; R26Rspo1/+) gonads. For 21–24 dpp superovulated mice, n=6 gonads for each genotype.
Statistical analysis
Data are shown as mean±s.e.m.
All the data were analyzed by unpaired one-sided Student’s t-test using Microsoft Excel (Redmond, WA, USA). Asterisks highlight the pertinent comparisons and indicate levels of significance: *P<0.05, **P<0.01 and ***P<0.001.
References
Clevers H, Nusse R . Wnt/beta-catenin signaling and disease. Cell 2012; 149: 1192–1205.
Seshagiri S, Stawiski EW, Durinck S, Modrusan Z, Storm EE, Conboy CB et al. Recurrent R-spondin fusions in colon cancer. Nature 2012; 488: 660–664.
Kuchenbaecker KB, Ramus SJ, Tyrer J, Lee A, Shen HC, Beesley J et al. Identification of six new susceptibility loci for invasive epithelial ovarian cancer. Nat Genet 2015; 47: 164–171.
Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 2013; 6: pl1.
Kim KA, Kakitani M, Zhao J, Oshima T, Tang T, Binnerts M et al. Mitogenic influence of human R-spondin1 on the intestinal epithelium. Science 2005; 309: 1256–1259.
Parma P, Radi O, Vidal V, Chaboissier MC, Dellambra E, Valentini S et al. R-spondin1 is essential in sex determination, skin differentiation and malignancy. Nat Genet 2006; 38: 1304–1309.
de Lau W, Peng WC, Gros P, Clevers H . The R-spondin/Lgr5/Rnf43 module: regulator of Wnt signal strength. Genes Dev 2014; 28: 305–316.
Kemler R . From cadherins to catenins: cytoplasmic protein interactions and regulation of cell adhesion. Trends Genet 1993; 9: 317–321.
Lyashenko N, Winter M, Migliorini D, Biechele T, Moon RT, Hartmann C et al. Differential requirement for the dual functions of beta-catenin in embryonic stem cell self-renewal and germ layer formation. Nat Cell Biol 2011; 13: 753–761.
Maeda O, Usami N, Kondo M, Takahashi M, Goto H, Shimokata K et al. Plakoglobin (gamma-catenin) has TCF/LEF family-dependent transcriptional activity in beta-catenin-deficient cell line. Oncogene 2004; 23: 964–972.
Chidgey M, Dawson C . Desmosomes: a role in cancer? Br J Cancer 2007; 96: 1783–1787.
Tomaselli S, Megiorni F, De Bernardo C, Felici A, Marrocco G, Maggiulli G et al. Syndromic true hermaphroditism due to an R-spondin1 (RSPO1) homozygous mutation. Hum Mutat 2008; 29: 220–226.
Maatouk DM, DiNapoli L, Alvers A, Parker KL, Taketo MM, Capel B et al. Stabilization of beta-catenin in XY gonads causes male-to-female sex-reversal. Hum Mol Genet 2008; 17: 2949–2955.
Chassot AA, Ranc F, Gregoire EP, Roepers-Gajadien HL, Taketo MM, Camerino G et al. Activation of beta-catenin signaling by Rspo1 controls differentiation of the mammalian ovary. Hum Mol Genet 2008; 17: 1264–1277.
Tomizuka K, Horikoshi K, Kitada R, Sugawara Y, Iba Y, Kojima A et al. R-spondin1 plays an essential role in ovarian development through positively regulating Wnt-4 signaling. Hum Mol Genet 2008; 17: 1278–1291.
Karl J, Capel B . Sertoli cells of the mouse testis originate from the coelomic epithelium. Dev Biol 1998; 203: 323–333.
Chassot AA, Bradford ST, Auguste A, Gregoire EP, Pailhoux E, de Rooij DG et al. WNT4 and RSPO1 together are required for cell proliferation in the early mouse gonad. Development 2012; 139: 4461–4472.
Mork L, Maatouk DM, McMahon JA, Guo JJ, Zhang P, McMahon AP et al. Temporal differences in granulosa cell specification in the ovary reflect distinct follicle fates in mice. Biol Reprod 2012; 86: 37.
Zheng W, Zhang H, Gorre N, Risal S, Shen Y, Liu K et al. Two classes of ovarian primordial follicles exhibit distinct developmental dynamics and physiological functions. Hum Mol Genet 2014; 23: 920–928.
Maatouk DM, Mork L, Chassot AA, Chaboissier MC, Capel B . Disruption of mitotic arrest precedes precocious differentiation and transdifferentiation of pregranulosa cells in the perinatal Wnt4 mutant ovary. Dev Biol 2013; 383: 295–306.
Rastetter RH, Bernard P, Palmer JS, Chassot AA, Chen H, Western PS et al. Marker genes identify three somatic cell types in the fetal mouse ovary. Dev Biol 2014; 394: 242–252.
Auguste A, Chassot AA, Grégoire EP, Renault L, Pannetier M, Treier M et al. Loss of R-spondin1 and Foxl2 amplifies female-to-male sex reversal in XX mice. Sex Dev 2011; 5: 304–317.
Hirshfield AN . Overview of ovarian follicular development: considerations for the toxicologist. Environ Mol Mutagen 1997; 29: 10–15.
Ng A, Tan S, Singh G, Rizk P, Swathi Y, Tan TZ et al. Lgr5 marks stem/progenitor cells in ovary and tubal epithelia. Nat Cell Biol 2014; 16: 745–757.
Pan H, Cui H, Liu S, Qian Y, Wu H, Li L et al. Lgr4 gene regulates corpus luteum maturation through modulation of the WNT-mediated EGFR-ERK signaling pathway. Endocrinology 2014; 155: 3624–3637.
Sun X, Terakawa J, Clevers H, Barker N, Daikoku T, Dey SK et al. Ovarian LGR5 is critical for successful pregnancy. Faseb J 2014; 28: 2380–2389.
Boerboom D, Paquet M, Hsieh M, Liu J, Jamin SP, Behringer RR et al. Misregulated Wnt/beta-catenin signaling leads to ovarian granulosa cell tumor development. Cancer Res 2005; 65: 9206–9215.
Mullany LK, Richards JS . Minireview: Animal Models and Mechanisms of Ovarian Cancer Development. Endocrinology 2012; 153: 1585–1592.
Jamieson S, Fuller PJ . Molecular pathogenesis of granulosa cell tumors of the ovary. Endocr Rev 2012; 33: 109–144.
Tomaselli S, Megiorni F, Lin L, Mazzilli MC, Gerrelli D, Majore S et al. Human RSPO1/R-spondin1 is expressed during early ovary development and augments beta-catenin signaling. PLoS ONE 2011; 6: e16366.
Lustig B, Jerchow B, Sachs M, Weiler S, Pietsch T, Karsten U et al. Negative feedback loop of Wnt signaling through upregulation of conductin/axin2 in colorectal and liver tumors. Mol Cell Biol 2002; 22: 1184–1193.
Durlinger AL, Visser JA, Themmen AP . Regulation of ovarian function: the role of anti-Mullerian hormone. Reproduction 2002; 124: 601–609.
Ahima RS, Dushay J, Flier SN, Prabakaran D, Flier JS . Leptin accelerates the onset of puberty in normal female mice. J Clin Invest 1997; 99: 391–395.
Osman P . Rate and course of atresia during follicular development in the adult cyclic rat. J Reprod Fertil 1985; 73: 261–270.
Rowlands TM, Symonds JM, Farookhi R, Blaschuk OW . Cadherins: crucial regulators of structure and function in reproductive tissues. Rev Reprod 2000; 5: 53–61.
Makrigiannakis A, Coukos G, Christofidou-Solomidou M, Gour BJ, Radice GL, Blaschuk O et al. N-cadherin-mediated human granulosa cell adhesion prevents apoptosis: a role in follicular atresia and luteolysis? Am J Pathol 1999; 154: 1391–1406.
Fan HY, O'Connor A, Shitanaka M, Shimada M, Liu Z, Richards JS et al. Beta-catenin (CTNNB1) promotes preovulatory follicular development but represses LH-mediated ovulation and luteinization. Mol Endocrinol 2010; 24: 1529–1542.
Liu Z, Ren YA, Pangas SA, Adams J, Zhou W, Castrillon DH et al. FOXO1/3 and PTEN Depletion in Granulosa Cells Promotes Ovarian Granulosa Cell Tumor Development. Mol Endocrinol 2015; 29: 1006–1024.
Hatano O, Takakusu A, Nomura M, Morohashi K . Identical origin of adrenal cortex and gonad revealed by expression profiles of Ad4BP/SF-1. Genes Cells 1996; 1: 663–671.
Bandiera R, Vidal VP, Motamedi FJ, Clarkson M, Sahut-Barnola I, von Gise A et al. WT1 maintains adrenal-gonadal primordium identity and marks a population of AGP-like progenitors within the adrenal gland. Dev Cell 2013; 27: 5–18.
Liu C, Peng J, Matzuk MM, Yao HH . Lineage specification of ovarian theca cells requires multicellular interactions via oocyte and granulosa cells. Nat Commun 2015; 6: 6934.
Kim AC, Reuter AL, Zubair M, Else T, Serecky K, Bingham NC et al. Targeted disruption of beta-catenin in Sf1-expressing cells impairs development and maintenance of the adrenal cortex. Development 2008; 135: 2593–2602.
Huang CC, Miyagawa S, Matsumaru D, Parker KL, Yao HH . Progenitor cell expansion and organ size of mouse adrenal is regulated by sonic hedgehog. Endocrinology 2010; 151: 1119–1128.
Boerboom D, White LD, Dalle S, Courty J, Richards JS . Dominant-stable beta-catenin expression causes cell fate alterations and Wnt signaling antagonist expression in a murine granulosa cell tumor model. Cancer Res 2006; 66: 1964–1973.
Albrecht KH, Eicher EM . Evidence that Sry is expressed in pre-Sertoli cells and Sertoli and granulosa cells have a common precursor. Dev Biol 2001; 240: 92–107.
Rocha AS, Vidal V, Mertz M, Kendall TJ, Charlet A, Okamoto H et al. The Angiocrine Factor Rspondin3 Is a Key Determinant of Liver Zonation. Cell Rep 2015; 13: 1757–1764.
Bingham NC, Verma-Kurvari S, Parada LF, Parker KL . Development of a steroidogenic factor 1/Cre transgenic mouse line. Genesis 2006; 44: 419–424.
Svingen T, François M, Wilhelm D, Koopman P . Three-dimensional imaging of Prox1-EGFP transgenic mouse gonads reveals divergent modes of lymphangiogenesis in the testis and ovary. PLoS ONE 2012; 7: e52620.
Acknowledgements
We thank Mireille Cutajar-Bossert for her work and management of funding and also Fariba Jianmotamedi, Aurélie Charlet and Béatrice Polo for their punctual help in this work. We are thankful to Walter Birchmeier (Berlin, Germany) for the Axin2+/LacZ mice and Ken Morohashi (Fukuoka, Japan) for the SF1 antibody. We thank Aitana Perea-Gomez for critical reading of the manuscript. This work was supported by grants from the Fondation ARC pour la Recherche sur le Cancer (PJA 20131200236) and Agence Nationale de la Recherche (ANR-09-GENM-009-03 and ANR-11-LABX-0028-01). EP was supported by a fellowship from INRA and Ligue Nationale contre le Cancer.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
De Cian, MC., Pauper, E., Bandiera, R. et al. Amplification of R-spondin1 signaling induces granulosa cell fate defects and cancers in mouse adult ovary. Oncogene 36, 208–218 (2017). https://doi.org/10.1038/onc.2016.191
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.191