Abstract
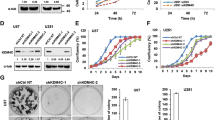
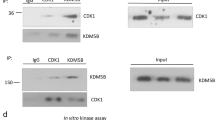
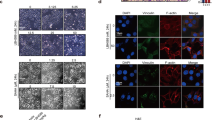
Invasive growth and apoptosis resistance of breast cancer cells are associated with metastasis and disease relapse. Here we identified that the lysine-specific demethylase KDM3A played a dual role in breast cancer cell invasion and apoptosis by demethylating histone and the non-histone protein p53, respectively. While inducing pro-invasive genes by erasing repressive histone H3 lysine 9 methylation, KDM3A promotes chemoresistance by demethylating p53. KDM3A suppressed pro-apoptotic functions of p53 by erasing p53-K372me1, as this methylation is crucial for the stability of chromatin-bound p53. Unexpectedly, depletion of KDM3A was capable of reactivating mutated p53 to induce the expression of pro-apoptotic genes in breast cancer with mutant p53. Moreover, KDM3A knockdown also potently inhibited tumorigenic potentials of breast cancer stem-like cells and rendered them sensitive to apoptosis induced by chemotherapeutic drugs. Taken together, our results suggest that KDM3A might be a potential therapeutic target for human breast cancer treatment and prevention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A . Cancer statistics. CA Cancer J Clin 2014; 64: 9–29.
Acharyya S, Oskarsson T, Vanharanta S, Malladi S, Kim J, Morris PG et al. A CXCL1 paracrine network links chancer chemoresistance and metastasis. Cell 2012; 150: 165–178.
Comoglio PM, Trusolino L . Invasive growth: from development to metastasis. J Clin Invest 2002; 109: 857–862.
Ding X, Pan H, Li J, Zhong Q, Chen X, Dry SM et al. Epigenetic activation of AP1 promotes squamous cell carcinoma metastasis. Sci Signal 2013; 6: ra28 1–13.
Kwon M . Epithelial-to-mesenchymal transition and cancer stem cells: emerging targets for novel cancer therapy. Cancer Gene Ther 2014; 21: 179–180.
Thiery JP, Acloque H, Huang RY, Nieto MA . Epithelial-mesenchymal transitions in development and disease. Cell 2009; 139: 871–890.
Ramadoss S, Chen X, Wang CY . Histone demethylase KDM6B promotes epithelial-mesenchymal transition. J Biol Chem 2012; 287: 44508–44517.
Scully OJ, Bay BH, Yip G, Yu Y . Breast cancer metastasis. Cancer Genomics Proteomics 2012; 9: 311–320.
Alexander S, Friedl P . Cancer invasion and resistance: interconnected processes of disease progression and therapy failure. Trends Mol Med 2012; 18: 13–26.
Muller PA, Vousden KH, Norman JC . p53 and its mutants in tumor cell migration and invasion. J Cell Biol 2011; 192: 209–218.
Shetzer Y, Solomon H, Koifman G, Molchadsky A, Horesh S, Rotter V . The paradigm of mutant p53-expressing cancer stem cells and drug resistance. Carcinogenesis 2014; 35: 1196–1208.
Muller PA, Vousden KH . p53 mutations in cancer. Nat Cell Biol 2013; 15: 2–8.
Mizuno H, Spike BT, Wahl GM, Levine AJ . Inactivation of p53 in breast cancers correlates with stem cell transcriptional signatures. Proc Natl Acad Sci USA 2010; 107: 22745–22750.
Ginestier C, Charafe-Jauffret E, Birnbaum D . p53 and cancer stem cells: the mevalonate connexion. Cell Cycle 2012; 11: 2583–2584.
Freed-Pastor WA, Mizuno H, Zhao X, Langerød A, Moon SH, Rodriguez-Barrueco R et al. Mutant p53 disrupts mammary tissue architecture via the mevalonate pathway. Cell 2012; 148: 244–258.
Bannister AJ, Kouzarides T . Regulation of chromatin by histone modifications. Cell Res 2011; 21: 381–395.
Shi Y, Whetstine JR . Dynamic regulation of histone lysine methylation by demethylases. Mol Cell 2007; 25: 1–14.
Kooistra SM, Helin K . Molecular mechanisms and potential functions of histone demethylases. Nat Rev Mol Cell Biol 2012; 13: 297–311.
Bieging KT, Mello SS, Attardi LD . Unravelling mechanisms of p53 mediated tumor suppression. Nat Rev Cancer 2014; 14: 359–370.
Huang J, Perez-Burgos L, Placek BJ, Sengupta R, Richter M, Dorsey JA et al. Repression of p53 activity by Smyd2-mediated methylation. Nature 2006; 444: 629–632.
Huang J, Dorsey J, Chuikov S, Pérez-Burgos L, Zhang X, Jenuwein T et al. G9a and Glp methylate lysine 373 in the tumor suppressor p53. J Biol Chem 2010; 285: 9636–9641.
Shi X, Kachirskaia I, Yamaguchi H, West LE, Wen H, Wang EW et al. Modulation of p53 function by SET8-mediated methylation at lysine 382. Mol Cell 2007; 27: 636–646.
Chuikov S, Kurash JK, Wilson JR, Xiao B, Justin N, Ivanov GS et al. Regulation of p53 activity through lysine methylation. Nature 2004; 432: 353–360.
Malanchi I, Santamaria-Martínez A, Susanto E, Peng H, Lehr HA, Delaloye JF et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 2011; 481: 85–89.
Baccelli I, Schneeweiss A, Riethdorf S, Stenzinger A, Schillert A, Vogel V et al. Identification of population of blood circulating tumor cells from breast cancer patients that initiates metastasis in a xenograftassay. Nat Biotechnol 2013; 31: 539–544.
Cheng L, Ramesh AV, Flesken-Nikitin A, Choi J, Nikitin AY . Mouse models for cancer stem cell research. Toxicol Pathol 2010; 38: 62–71.
Chang CJ, Chao CH, Xia W, Yang JY, Xiong Y, Li CW et al. p53 regulates epithelial-mesenchymal transition and stem cell properties through modulating miRNAs. Nat Cell Biol 2011; 13: 317–323.
Cordenonsi M, Zanconato F, Azzolin L, Forcato M, Rosato A, Frasson C et al. The Hippo transducer TAZ confers cancer stem cell-related traits on breast cancer cells. Cell 2011; 147: 759–772.
O'Brien CA, Kreso A, Jamieson CH . Cancer stem cells and self-renewal. Clin Cancer Res 2010; 16: 3113–3120.
Okada Y, Scott G, Ray MK, Mishina Y, Zhang Y . Histone demethylase JHDM2A is critical for Tnp1 and Prm1 transcription and spermatogenesis. Nature 2007; 450: 119–123.
Tateishi K, Okada Y, Kallin EM, Zhang Y . Role of Jhdm2a in regulating metabolic gene expression and obesity resistance. Nature 2009; 458: 757–761.
Krieg AJ, Rankin EB, Chan D, Razorenova O, Fernandez S, Giaccia AJ . Regulation of the histone demethylase JMJD1A by hypoxia-inducible factor 1 alpha enhances hypoxic gene expression and tumor growth. Mol Cell Biol 2010; 30: 344–353.
Kuroki S, Matoba S, Akiyoshi M, Matsumura Y, Miyachi H, Mise N et al. Epigenetic regulation of mouse sex determination by the histone demethylase Jmjd1a. Science 2013; 341: 1106–1109.
Boye K, Maelandsmo GM . S100A4 and metastasis: a small actor playing many roles. Am J Pathol 2010; 176: 528–535.
Jiao X, Katiyar S, Willmarth NE, Liu M, Ma X, Flomenberg N et al. c-Jun induces mammary epithelial cellular invasion and breast cancer stem cell expansion. J Biol Chem 2010; 285: 8218–8226.
Vandooren J, Van den Steen PE, Opdenakker G . Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP-9): the next decade. Crit Rev Biochem Mol Biol 2013; 48: 222–272.
Wade MA, Jones D, Wilson L, Stockley J, Coffey K, Robson CN et al. The histone demethylase enzyme KDM3A is a key estrogen receptor regulator in breast cancer. Nucleic Acids Res 2014; 43: 196–207.
Lane DP . Cancer. p53, guardian of the genome. Nature 1992; 358: 15–16.
Lane D, Levine A . p53 research: the past thirty years and the next thirty years. Cold Spring Harb Perspect Biol 2010; 2: a000893.
Kurash JK, Lei H, Shen Q, Marston WL, Granda BW, Fan H et al. Methylation of p53 by Set7/9 mediates p53 acetylation and activity in vivo. Mol Cell 2008; 29: 392–400.
Campaner S, Spreafico F, Burgold T, Doni M, Rosato U, Amati B et al. The methyltransferase Set7/9 (Setd7) is dispensable for the p53-mediated DNA damage response in vivo. Mol Cell 2011; 43: 681–688.
Lehnertz B, Rogalski JC, Schulze FM, Yi L, Lin S, Kast J et al. p53-dependent transcription and tumor suppression are not affected in Set7/9-deficient mice. Mol Cell 2011; 43: 673–680.
Joerger AC, Fersht AR . Structural biology of the tumor suppressor p53 and cancer associated mutants. Adv Cancer Res 2007; 97: 1–23.
Perez RE, Knights CD, Sahu G, Catania J, Kolukula VK, Stoler D et al. Restoration of DNA-binding and growth suppressive activity of mutant forms of p53 via a PCAF-mediated acetylation pathway. J Cell Physiol 2010; 225: 394–405.
Ivanov GS, Ivanova T, Kurash J, Ivanov A, Chuikov S, Gizatullin F et al. Methylation-acetylation interplay activates p53 in response to DNA damage. Mol Cell Biol 2007; 27: 6756–6769.
Ramadoss S, Li J, Ding X, Al Hezaimi K, Wang CY . Transducin β-like protein 1 recruits nuclear factor κB to the target gene promoter for transcriptional activation. Mol Cell Biol 2011; 31: 924–934.
Zeng Q, Li S, Chepeha DB, Giordano TJ, Li J, Zhang H et al. Crosstalk between tumor and endothelial cells promotes tumor angiogenesis by MAPK activation of Notch signaling. Cancer Cell 2005; 8: 13–23.
Acknowledgements
This work was supported by the NIH grants R3713848 and DE15964 and the Shapiro Family Charitable Foundation. We thank Dr Yi Zhang for the generous gift of KDM3A plasmid.
Author contributions
SR and C-YW conceived ideas. All the authors designed experiments, SR and GG performed experiments. SR and C-YW wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Ramadoss, S., Guo, G. & Wang, CY. Lysine demethylase KDM3A regulates breast cancer cell invasion and apoptosis by targeting histone and the non-histone protein p53. Oncogene 36, 47–59 (2017). https://doi.org/10.1038/onc.2016.174
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.174
This article is cited by
-
Knockout of KDM3A in MDA-MB-231 breast cancer cells inhibits tumor malignancy and promotes apoptosis
Journal of Molecular Histology (2024)
-
Targeting epigenetic regulators to overcome drug resistance in cancers
Signal Transduction and Targeted Therapy (2023)
-
Downregulation of miR-335 exhibited an oncogenic effect via promoting KDM3A/YAP1 networks in clear cell renal cell carcinoma
Cancer Gene Therapy (2022)
-
Proteasome Subunit Alpha Type 7 Promotes Proliferation and Metastasis of Gastric Cancer Through MAPK Signaling Pathway
Digestive Diseases and Sciences (2022)
-
microRNA-155-3p attenuates intervertebral disc degeneration via inhibition of KDM3A and HIF1α
Inflammation Research (2021)