Abstract
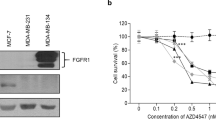
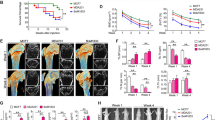
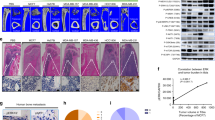
Breast cancer (BCa) bone metastases cause osteolytic bone lesions, which result from the interactions of metastatic BCa cells with osteoclasts and osteoblasts. Osteoclasts differentiate from myeloid lineage cells. To understand the cell-specific role of transforming growth factor beta (TGF-β) in the myeloid lineage, in BCa bone metastases, MDA-MB-231 BCa cells were intra-tibially or intra-cardially injected into LysMCre/Tgfbr2floxE2/floxE2 knockout (LysMCre/Tgfbr2 KO) or Tgfbr2floxE2/floxE2 mice. Metastatic bone lesion development was compared by analysis of both lesion number and area. We found that LysMCre/Tgfbr2 knockout significantly decreased MDA-MB-231 bone lesion development in both the cardiac and tibial injection models. LysMCre/Tgfbr2 knockout inhibited the tumor cell proliferation, angiogenesis and osteoclastogenesis of the metastatic bones. Cytokine array analysis showed that basic fibroblast growth factor (bFGF) was downregulated in MDA-MB-231-injected tibiae from the LysMCre/Tgfbr2 KO group, and intravenous injection of the recombinant bFGF to LysMCre/Tgfbr2 KO mice rescued the inhibited metastatic bone lesion development. The mechanism by which bFGF rescued the bone lesion development was by promotion of tumor cell proliferation through the downstream mitogen-activated protein kinase (MAPK)-extracellular signal–regulated kinase (ERK)-cFos pathway after binding to the FGF receptor 1 (FGFR1). Consistent with animal studies, we found that in human BCa bone metastatic tissues, TGF-β type II receptor (TβRII) and p-Smad2 were expressed in osteoclasts and tumor cells, and were correlated with the expression of FGFR1. Our studies suggest that myeloid-specific TGF-β signaling-mediated bFGF in the bone promotes BCa bone metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Coleman RE . Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res 2006; 12: 6243s–6249s.
Reddi AH, Roodman D, Freeman C, Mohla S . Mechanisms of tumor metastasis to the bone: challenges and opportunities. J Bone Miner Res 2003; 18: 190–194.
Ikushima H, Miyazono K . TGFbeta signalling: a complex web in cancer progression. Nat Rev Cancer 2010; 10: 415–424.
Bhowmick NA, Chytil A, Plieth D, Gorska AE, Dumont N, Shappell S et al. TGF-beta signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science 2004; 303: 848–851.
Cheng N, Bhowmick NA, Chytil A, Gorksa AE, Brown KA, Muraoka R et al. Loss of TGF-beta type II receptor in fibroblasts promotes mammary carcinoma growth and invasion through upregulation of TGF-alpha-, MSP- and HGF-mediated signaling networks. Oncogene 2005; 24: 5053–5068.
Cheng N, Chytil A, Shyr Y, Joly A, Moses HL . Enhanced hepatocyte growth factor signaling by type II transforming growth factor-beta receptor knockout fibroblasts promotes mammary tumorigenesis. Cancer Res 2007; 67: 4869–4877.
Li X, Placencio V, Iturregui JM, Uwamariya C, Sharif-Afshar AR, Koyama T et al. Prostate tumor progression is mediated by a paracrine TGF-beta/Wnt3a signaling axis. Oncogene 2008; 27: 7118–7130.
Li X, Sterling JA, Fan KH, Vessella RL, Shyr Y, Hayward SW et al. Loss of TGF-beta responsiveness in prostate stromal cells alters chemokine levels and facilitates the development of mixed osteoblastic/osteolytic bone lesions. Mol Cancer Res 2012; 10: 494–503.
Kim BG, Li C, Qiao W, Mamura M, Kasprzak B, Anver M et al. Smad4 signalling in T cells is required for suppression of gastrointestinal cancer. Nature 2006; 441: 1015–1019.
Novitskiy SV, Pickup MW, Chytil A, Polosukhina D, Owens P, Moses HL . Deletion of TGF-beta signaling in myeloid cells enhances their anti-tumorigenic properties. J Leukoc Biol 2012; 92: 641–651.
Pang Y, Gara SK, Achyut BR, Li Z, Yan HH, Day CP et al. TGF-beta signaling in myeloid cells is required for tumor metastasis. Cancer Discov 2013; 3: 936–951.
Turner N, Grose R . Fibroblast growth factor signalling: from development to cancer. Nat Rev Cancer 2010; 10: 116–129.
Powers CJ, McLeskey SW, Wellstein A . Fibroblast growth factors, their receptors and signaling. Endocr Relat Cancer 2000; 7: 165–197.
Brunner G, Nguyen H, Gabrilove J, Rifkin DB, Wilson EL . Basic fibroblast growth factor expression in human bone marrow and peripheral blood cells. Blood 1993; 81: 631–638.
Busch S, Acar A, Magnusson Y, Gregersson P, Ryden L, Landberg G . TGF-beta receptor type-2 expression in cancer-associated fibroblasts regulates breast cancer cell growth and survival and is a prognostic marker in pre-menopausal breast cancer. Oncogene 2015; 34: 27–38.
Greenman C, Stephens P, Smith R, Dalgliesh GL, Hunter C, Bignell G et al. Patterns of somatic mutation in human cancer genomes. Nature 2007; 446: 153–158.
Turner N, Grose R . Fibroblast growth factor signalling: from development to cancer. Nat Rev Cancer 2010; 10: 116–129.
Courjal F, Cuny M, Simony-Lafontaine J, Louason G, Speiser P, Zeillinger R et al. Mapping of DNA amplifications at 15 chromosomal localizations in 1875 breast tumors: definition of phenotypic groups. Cancer Res 1997; 57: 4360–4367.
Wan X, Corn PG, Yang J, Palanisamy N, Starbuck MW, Efstathiou E et al. Prostate cancer cell-stromal cell crosstalk via FGFR1 mediates antitumor activity of dovitinib in bone metastases. Sci Transl Med 2014; 6: 252ra122.
Ell B, Mercatali L, Ibrahim T, Campbell N, Schwarzenbach H, Pantel K et al. Tumor-induced osteoclast miRNA changes as regulators and biomarkers of osteolytic bone metastasis. Cancer Cell 2013; 24: 542–556.
Johnson RW, Merkel AR, Page JM, Ruppender NS, Guelcher SA, Sterling JA . Wnt signaling induces gene expression of factors associated with bone destruction in lung and breast cancer. Clin Exp Metastasis 2014; 31: 945–959.
Chytil A, Magnuson MA, Wright CV, Moses HL . Conditional inactivation of the TGF-beta type II receptor using Cre:Lox. Genesis 2002; 32: 73–75.
Zhong Z, Zylstra-Diegel CR, Schumacher CA, Baker JJ, Carpenter AC, Rao S et al. Wntless functions in mature osteoblasts to regulate bone mass. Proc Natl Acad Sci USA 2012; 109: E2197–E2204.
Sethi N, Dai X, Winter CG, Kang Y . Tumor-derived JAGGED1 promotes osteolytic bone metastasis of breast cancer by engaging notch signaling in bone cells. Cancer Cell 2011; 19: 192–205.
Acknowledgements
We thank Dr Julie A Sterling from Vanderbilt University and Dr Yibin Kang from Princeton University for providing cell lines. The Van Andel Research Institute supported this research (VARI start-up to X Li, 53010A). We thank Lisa Turner of the Pathology and Biorepository Core for her pathology expertise, the Vivarium and Transgenics Core for animal technical assistance and Diana Lewis for administrative support. We thank Dr Travis Burgers for the construction of 3D micro-CT pictures. Special thanks to Dr Mary E Winn from the Bioinformatics and Biostatistics Core for her great help in answering statistical questions. Finally, we thank David Nadziejka for his technical editing of this manuscript. The content is solely the responsibility of the authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Meng, X., Vander Ark, A., Lee, P. et al. Myeloid-specific TGF-β signaling in bone promotes basic-FGF and breast cancer bone metastasis. Oncogene 35, 2370–2378 (2016). https://doi.org/10.1038/onc.2015.297
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.297
This article is cited by
-
Systems analysis of avascular necrosis of femoral head using integrative data analysis and literature mining delineates pathways associated with disease
Scientific Reports (2020)
-
Elimination of tumor hypoxia by eribulin demonstrated by 18F-FMISO hypoxia imaging in human tumor xenograft models
EJNMMI Research (2019)
-
Attenuation of TGFBR2 expression and tumour progression in prostate cancer involve diverse hypoxia-regulated pathways
Journal of Experimental & Clinical Cancer Research (2018)
-
miR-130a and miR-145 reprogram Gr-1+CD11b+ myeloid cells and inhibit tumor metastasis through improved host immunity
Nature Communications (2018)
-
Sclerostin induced tumor growth, bone metastasis and osteolysis in breast cancer
Scientific Reports (2017)