Abstract
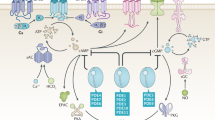
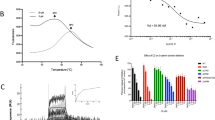
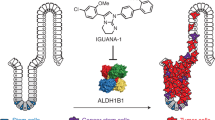
The cyclic nucleotide phosphodiesterase 10A (PDE10) has been mostly studied as a therapeutic target for certain psychiatric and neurological conditions, although a potential role in tumorigenesis has not been reported. Here we show that PDE10 is elevated in human colon tumor cell lines compared with normal colonocytes, as well as in colon tumors from human clinical specimens and intestinal tumors from ApcMin/+ mice compared with normal intestinal mucosa, respectively. An isozyme and tumor-selective role of PDE10 were evident by the ability of small-molecule inhibitors and small interfering RNA knockdown to suppress colon tumor cell growth with reduced sensitivity of normal colonocytes. Stable knockdown of PDE10 by short hairpin RNA also inhibits colony formation and increases doubling time of colon tumor cells. PDE10 inhibition selectively activates cGMP/cGMP-dependent protein kinase signaling to suppress β-catenin levels and T-cell factor (TCF) transcriptional activity in colon tumor cells. Conversely, ectopic expression of PDE10 in normal and precancerous colonocytes increases proliferation and activates TCF transcriptional activity. These observations suggest a novel role of PDE10 in colon tumorigenesis and that inhibitors may be useful for the treatment or prevention of colorectal cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Francis SH, Blount MA, Corbin JD . Mammalian cyclic nucleotide phosphodiesterases: molecular mechanisms and physiological functions. Physiol Rev 2011; 91: 651–690.
Bender AT, Beavo JA . Cyclic nucleotide phosphodiesterases: molecular regulation to clinical use. Pharmacol Rev 2006; 58: 488–520.
Marko D, Pahlke G, Merz KH, Eisenbrand G . Cyclic 3',5'-nucleotide phosphodiesterases: potential targets for anticancer therapy. Chem Res Toxicol 2000; 13: 944–948.
Piazza GA, Thompson WJ, Pamukcu R, Alila HW, Whitehead CM, Liu L et al. Exisulind, a novel proapoptotic drug, inhibits rat urinary bladder tumorigenesis. Cancer Res 2001; 61: 3961–3968.
Saravani R, Karami-Tehrani F, Hashemi M, Aghaei M, Edalat R . Inhibition of phosphodiesterase 9 induces cGMP accumulation and apoptosis in human breast cancer cell lines, MCF-7 and MDA-MB-468. Cell Prolif 2012; 45: 199–206.
Shimizu K, Murata T, Watanabe Y, Sato C, Morita H, Tagawa T . Characterization of phosphodiesterase 1 in human malignant melanoma cell lines. Anticancer Res 2009; 29: 1119–1122.
Zhang L, Murray F, Zahno A, Kanter JR, Chou D, Suda R et al. Cyclic nucleotide phosphodiesterase profiling reveals increased expression of phosphodiesterase 7B in chronic lymphocytic leukemia. Proc Natl Acad Sci USA 2008; 105: 19532–19537.
D'Andrea MR, Qiu Y, Haynes-Johnson D, Bhattacharjee S, Kraft P, Lundeen S . Expression of PDE11A in normal and malignant human tissues. J Histochem Cytochem 2005; 53: 895–903.
Tinsley HN, Gary BD, Thaiparambil J, Li N, Lu W, Li Y et al. Colon tumor cell growth-inhibitory activity of sulindac sulfide and other nonsteroidal anti-inflammatory drugs is associated with phosphodiesterase 5 inhibition. Cancer Prev Res 2010; 3: 1303–1313.
Savai R, Pullamsetti SS, Banat GA, Weissmann N, Ghofrani HA, Grimminger F et al. Targeting cancer with phosphodiesterase inhibitors. Expert Opin Investig Drugs 2010; 19: 117–131.
Lucas KA, Pitari GM, Kazerounian S, Ruiz-Stewart I, Park J, Schulz S et al. Guanylyl cyclases and signaling by cyclic GMP. Pharmacol Rev 2000; 52: 375–414.
Li N, Xi Y, Tinsley HN, Gurpinar E, Gary BD, Zhu B et al. Sulindac Selectively Inhibits Colon Tumor Cell Growth by Activating the cGMP/PKG Pathway to Suppress Wnt/beta-Catenin Signaling. Mol Cancer Ther 2013; 12: 1848–1859.
Pitari GM, Di Guglielmo MD, Park J, Schulz S, Waldman SA . Guanylyl cyclase C agonists regulate progression through the cell cycle of human colon carcinoma cells. Proc Natl Acad Sci USA 2001; 98: 7846–7851.
Steinbrecher KA, Tuohy TM, Heppner Goss K, Scott MC, Witte DP, Groden J et al. Expression of guanylin is downregulated in mouse and human intestinal adenomas. Biochem Biophys Res Commun 2000; 273: 225–230.
Shailubhai K, Yu HH, Karunanandaa K, Wang JY, Eber SL, Wang Y et al. Uroguanylin treatment suppresses polyp formation in the Apc(Min/+) mouse and induces apoptosis in human colon adenocarcinoma cells via cyclic GMP. Cancer Res 2000; 60: 5151–5157.
Thompson WJ, Piazza GA, Li H, Liu L, Fetter J, Zhu B et al. Exisulind induction of apoptosis involves guanosine 3',5'-cyclic monophosphate phosphodiesterase inhibition, protein kinase G activation, and attenuated beta-catenin. Cancer Res 2000; 60: 3338–3342.
Hou Y, Gupta N, Schoenlein P, Wong E, Martindale R, Ganapathy V et al. An anti-tumor role for cGMP-dependent protein kinase. Cancer Lett 2006; 240: 60–68.
Deguchi A, Thompson WJ, Weinstein IB . Activation of protein kinase G is sufficient to induce apoptosis and inhibit cell migration in colon cancer cells. Cancer Res 2004; 64: 3966–3973.
Kwon IK, Schoenlein PV, Delk J, Liu K, Thangaraju M, Dulin NO et al. Expression of cyclic guanosine monophosphate-dependent protein kinase in metastatic colon carcinoma cells blocks tumor angiogenesis. Cancer 2008; 112: 1462–1470.
Loughney K, Snyder PB, Uher L, Rosman GJ, Ferguson K, Florio VA . Isolation and characterization of PDE10A, a novel human 3′, 5′-cyclic nucleotide phosphodiesterase. Gene 1999; 234: 109–117.
Fujishige K, Kotera J, Michibata H, Yuasa K, Takebayashi S, Okumura K et al. Cloning and characterization of a novel human phosphodiesterase that hydrolyzes both cAMP and cGMP (PDE10A). J Biol Chem 1999; 274: 18438–18445.
Soderling SH, Bayuga SJ, Beavo JA . Isolation and characterization of a dual-substrate phosphodiesterase gene family: PDE10A. Proc Natl Acad Sci USA 1999; 96: 7071–7076.
Coskran TM, Morton D, Menniti FS, Adamowicz WO, Kleiman RJ, Ryan AM et al. Immunohistochemical localization of phosphodiesterase 10A in multiple mammalian species. J Histochem Cytochem 2006; 54: 1205–1213.
Xie Z, Adamowicz WO, Eldred WD, Jakowski AB, Kleiman RJ, Morton DG et al. Cellular and subcellular localization of PDE10A, a striatum-enriched phosphodiesterase. Neuroscience 2006; 139: 597–607.
Siuciak JA, McCarthy SA, Chapin DS, Fujiwara RA, James LC, Williams RD et al. Genetic deletion of the striatum-enriched phosphodiesterase PDE10A: evidence for altered striatal function. Neuropharmacology 2006; 51: 374–385.
Siuciak JA, Chapin DS, Harms JF, Lebel LA, McCarthy SA, Chambers L et al. Inhibition of the striatum-enriched phosphodiesterase PDE10A: a novel approach to the treatment of psychosis. Neuropharmacology 2006; 51: 386–396.
Richter M, Jurek D, Wrba F, Kaserer K, Wurzer G, Karner-Hanusch J et al. Cells obtained from colorectal microadenomas mirror early premalignant growth patterns in vitro. Eur J Cancer 2002; 38: 1937–1945.
Tinsley HN, Gary BD, Keeton AB, Zhang W, Abadi AH, Reynolds RC et al. Sulindac sulfide selectively inhibits growth and induces apoptosis of human breast tumor cells by phosphodiesterase 5 inhibition, elevation of cyclic GMP, and activation of protein kinase G. Mol Cancer Ther 2009; 8: 3331–3340.
Whitt JD, Li N, Tinsley HN, Chen X, Zhang W, Li Y et al. A Novel Sulindac Derivative that Potently Suppresses Colon Tumor Cell Growth by Inhibiting cGMP Phosphodiesterase and beta-Catenin Transcriptional Activity. Cancer Prev Res 2012; 5: 822–833.
Pusztai L, Zhen JH, Arun B, Rivera E, Whitehead C, Thompson WJ et al. Phase I and II study of exisulind in combination with capecitabine in patients with metastatic breast cancer. J Clin Oncol 2003; 21: 3454–3461.
Whitehead CM, Earle KA, Fetter J, Xu S, Hartman T, Chan DC et al. Exisulind-induced apoptosis in a non-small cell lung cancer orthotopic lung tumor model augments docetaxel treatment and contributes to increased survival. Mol Cancer Ther 2003; 2: 479–488.
Deguchi A, Soh JW, Li H, Pamukcu R, Thompson WJ, Weinstein IB . Vasodilator-stimulated phosphoprotein (VASP) phosphorylation provides a biomarker for the action of exisulind and related agents that activate protein kinase G. Mol Cancer Ther 2002; 1: 803–809.
Smolenski A, Bachmann C, Reinhard K, Honig-Liedl P, Jarchau T, Hoschuetzky H et al. Analysis and regulation of vasodilator-stimulated phosphoprotein serine 239 phosphorylation in vitro and in intact cells using a phosphospecific monoclonal antibody. J Biol Chem 1998; 273: 20029–20035.
Kwon IK, Wang R, Thangaraju M, Shuang H, Liu K, Dashwood R et al. PKG inhibits TCF signaling in colon cancer cells by blocking beta-catenin expression and activating FOXO4. Oncogene 2010; 29: 3423–3434.
Kim PJ, Plescia J, Clevers H, Fearon ER, Altieri DC . Survivin and molecular pathogenesis of colorectal cancer. Lancet 2003; 362: 205–209.
Anderson CB, Neufeld KL, White RL . Subcellular distribution of Wnt pathway proteins in normal and neoplastic colon. Proc Natl Acad Sci USA 2002; 99: 8683–8688.
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B et al. Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science 1997; 275: 1787–1790.
Liu L, Li H, Underwood T, Lloyd M, David M, Sperl G et al. Cyclic GMP-dependent protein kinase activation and induction by exisulind and CP461 in colon tumor cells. J Pharmacol Exp Ther 2001; 299: 583–592.
Li Q, Dashwood WM, Zhong X, Al-Fageeh M, Dashwood RH . Cloning of the rat beta-catenin gene (Ctnnb1) promoter and its functional analysis compared with the Catnb and CTNNB1 promoters. Genomics 2004; 83: 231–242.
Gudi T, Lohmann SM, Pilz RB . Regulation of gene expression by cyclic GMP-dependent protein kinase requires nuclear translocation of the kinase: identification of a nuclear localization signal. Mol Cell Biol 1997; 17: 5244–5254.
Moyer MP, Manzano LA, Merriman RL, Stauffer JS, Tanzer LR . NCM460, a normal human colon mucosal epithelial cell line. In Vitro Cell Dev Biol Animal 1996; 32: 315–317.
Li X, Gao L, Cui Q, Gary BD, Dyess DL, Taylor W et al. Sulindac inhibits tumor cell invasion by suppressing NF-kappaB-mediated transcription of microRNAs. Oncogene 2012; 31: 4979–4986.
Tian X, Vroom C, Ghofrani HA, Weissmann N, Bieniek E, Grimminger F et al. Phosphodiesterase 10A upregulation contributes to pulmonary vascular remodeling. PloS One 2011; 6: e18136.
Chappie TA, Humphrey JM, Allen MP, Estep KG, Fox CB, Lebel LA et al. Discovery of a series of 6,7-dimethoxy-4-pyrrolidylquinazoline PDE10A inhibitors. J Med Chem 2007; 50: 182–185.
Acknowledgements
We thank Dr Graeme Bolger for critical reading of the manuscript. We also thank Dr Marian from Medical University Vienna, Austria for the generous gift of LT97 adenoma cell line and Dr Dan Dixon from University of Kansas Cancer Center for providing specimens from the ApcMin/+ mouse model. We are also grateful to Dr Darren Browning from Georgia Regents University Cancer Center for providing the CTNNB1 luciferase reporter construct. This work was supported by National Institutes of Health Grants, 1R01CA131378, 1R01CA148817 and 1R01CA155638 to GAP.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
MPM has ownership interest (including patents) in INCELL Corporation. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Li, N., Lee, K., Xi, Y. et al. Phosphodiesterase 10A: a novel target for selective inhibition of colon tumor cell growth and β-catenin-dependent TCF transcriptional activity. Oncogene 34, 1499–1509 (2015). https://doi.org/10.1038/onc.2014.94
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.94
This article is cited by
-
Phosphodiesterase 10A (PDE10A) as a novel target to suppress β-catenin and RAS signaling in epithelial ovarian cancer
Journal of Ovarian Research (2022)
-
Therapeutic targeting of 3′,5′-cyclic nucleotide phosphodiesterases: inhibition and beyond
Nature Reviews Drug Discovery (2019)
-
Positively selected enhancer elements endow osteosarcoma cells with metastatic competence
Nature Medicine (2018)
-
Phosphodiesterases in non-neoplastic appearing colonic mucosa from patients with colorectal neoplasia
BMC Cancer (2016)