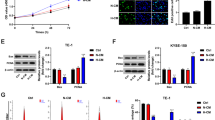
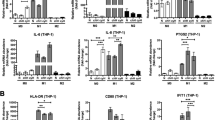
Abstract
5-lipoxygenase (5-LOX), a member of the lipoxygenase gene family, is a key enzyme assisting in the conversion of arachidonic acid to 5-HETE and leukotrienes. Tumor-associated macrophages (TAMs) have a critical role in the progression and metastasis of many tumors, including ovarian tumors. Moreover, TAMs are often found in a high density in the hypoxic areas of tumors. However, the relevant mechanisms have not been studied explicitly until now. In this study, we found that the expression of 5-LOX strongly correlated with the density of TAMs in hypoxic areas of human ovarian tumor tissues. In cultured ovarian cancer cells, 5-LOX metabolites were increased under hypoxic conditons. Increased 5-LOX metabolites from hypoxic ovarian cancer cells promoted migration and invasion of macrophages, which was further demonstrated to be mediated by the upregulation of matrix metalloproteinase (MMP)-7 expression through the p38 pathway. Besides, we also showed that 5-LOX metabolites enhanced the release of tumor necrosis factor (TNF-α) and heparin-binding epidermal growth factor-like growth factor through upregulation of MMP-7. Furthermore, in animal models, Zileuton (a selective and specific 5-LOX inhibitor) reduced the MMP-7 expression and the number of macrophages infiltrating in the xenograft. Our findings suggest for the first time that increased metabolites of 5-LOX from hypoxic ovarian cancer cells promote TAM infiltration. These results of this study have immediate translational implications for the therapeutic exploitation of TAMs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Balkwill F, Mantovani A . Inflammation and cancer: back to Virchow? Lancet 2001; 357: 539–545.
Coussens LM, Werb Z . Inflammation and cancer. Nature 2002; 420: 860–867.
Meng Y, Beckett MA, Liang H, Mauceri HJ, van Rooijen N, Cohen KS et al. Blockade of tumor necrosis factor alpha signaling in tumor-associated macrophages as a radiosensitizing strategy. Cancer Res 2010; 70: 1534–1543.
Pollard JW . Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer 2004; 4: 71–78.
Allavena P, Sica A, Solinas G, Porta C, Mantovani A . The inflammatory micro-environment in tumor progression: the role of tumor-associated macrophages. Crit Rev Oncol Hematol 2008; 66: 1–9.
Murdoch C, Giannoudis A, Lewis CE . Mechanisms regulating the recruitment of macrophages into hypoxic areas of tumors and other ischemic tissues. Blood 2004; 104: 2224–2234.
Rolny C, Mazzone M, Tugues S, Laoui D, Johansson I, Coulon C et al. HRG inhibits tumor growth and metastasis by inducing macrophage polarization and vessel normalization through downregulation of PlGF. Cancer Cell 2011; 19: 31–44.
Ruhrberg C, De Palma M . A double agent in cancer: deciphering macrophage roles in human tumors. Nat Med 2010; 16: 861–862.
Bingham BA, Hatef DA, Chevez-Barrios P, Blackmon SH, Kim MP . Increased FDG activity in a dermatofibroma in esophageal cancer patient. Clin Nucl Med 2013; 38: e140–e142.
Mitchem JB, Brennan DJ, Knolhoff BL, Belt BA, Zhu Y, Sanford DE et al. Targeting tumor-infiltrating macrophages decreases tumor-initiating cells, relieves immunosuppression, and improves chemotherapeutic responses. Cancer Res 2013; 73: 1128–1141.
Reinartz S, Schumann T, Finkernagel F, Wortmann A, Jansen JM, Meissner W et al. Mixed-polarization phenotype of ascites-associated macrophages in human ovarian carcinoma: correlation of CD163 expression, cytokine levels and early relapse. Int J Cancer 2014; 134: 32–42.
Bingle L, Brown NJ, Lewis CE . The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol 2002; 196: 254–265.
Lewis JS, Landers RJ, Underwood JC, Harris AL, Lewis CE . Expression of vascular endothelial growth factor by macrophages is up-regulated in poorly vascularized areas of breast carcinomas. J Pathol 2000; 192: 150–158.
Maniecki MB, Etzerodt A, Ulhoi BP, Steiniche T, Borre M, Dyrskjot L et al. Tumor-promoting macrophages induce the expression of the macrophage-specific receptor CD163 in malignant cells. Int J Cancer 2012; 131: 2320–2331.
Negus RP, Stamp GW, Hadley J, Balkwill FR . Quantitative assessment of the leukocyte infiltrate in ovarian cancer and its relationship to the expression of C-C chemokines. Am J Pathol 1997; 150: 1723–1734.
Lin EY, Nguyen AV, Russell RG, Pollard JW . Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J Exp Med 2001; 193: 727–740.
Negus RP, Stamp GW, Relf MG, Burke F, Malik ST, Bernasconi S et al. The detection and localization of monocyte chemoattractant protein-1 (MCP-1) in human ovarian cancer. J Clin Invest 1995; 95: 2391–2396.
Haeggstrom JZ, Funk CD . Lipoxygenase and leukotriene pathways: biochemistry, biology, and roles in disease. Chem Rev 2011; 111: 5866–5898.
Freedman RS, Wang E, Voiculescu S, Patenia R, Bassett Jr. RL, Deavers M et al. Comparative analysis of peritoneum and tumor eicosanoids and pathways in advanced ovarian cancer. Clin Cancer Res 2007; 13: 5736–5744.
Rocconi RP, Kirby TO, Seitz RS, Beck R, Straughn Jr. JM, Alvarez RD et al. Lipoxygenase pathway receptor expression in ovarian cancer. Reprod Sci 2008; 15: 321–326.
Bonizzi G, Piette J, Schoonbroodt S, Greimers R, Havard L, Merville MP et al. Reactive oxygen intermediate-dependent NF-kappaB activation by interleukin-1beta requires 5-lipoxygenase or NADPH oxidase activity. Mol Cell Biol 1999; 19: 1950–1960.
Dixon RA, Diehl RE, Opas E, Rands E, Vickers PJ, Evans JF et al. Requirement of a 5-lipoxygenase-activating protein for leukotriene synthesis. Nature 1990; 343: 282–284.
Yamaguchi T, Baba K, Doi Y, Yano K . Effect of adrenomedullin on aldosterone secretion by dispersed rat adrenal zona glomerulosa cells. Life Sci 1995; 56: 379–387.
Funk CD . Prostaglandins and leukotrienes: advances in eicosanoid biology. Science 2001; 294: 1871–1875.
Sitia G, Isogawa M, Iannacone M, Campbell IL, Chisari FV, Guidotti LG . MMPs are required for recruitment of antigen-nonspecific mononuclear cells into the liver by CTLs. J Clin Invest 2004; 113: 1158–1167.
Kessenbrock K, Plaks V, Werb Z . Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 2010; 141: 52–67.
Dunsmore SE, Saarialho-Kere UK, Roby JD, Wilson CL, Matrisian LM, Welgus HG et al. Matrilysin expression and function in airway epithelium. J Clin Invest 1998; 102: 1321–1331.
Halpert I, Sires UI, Roby JD, Potter-Perigo S, Wight TN, Shapiro SD et al. Matrilysin is expressed by lipid-laden macrophages at sites of potential rupture in atherosclerotic lesions and localizes to areas of versican deposition, a proteoglycan substrate for the enzyme. Proc Natl Acad Sci USA 1996; 93: 9748–9753.
Gonsalves CS, Kalra VK . Hypoxia-mediated expression of 5-lipoxygenase-activating protein involves HIF-1alpha and NF-kappaB and microRNAs 135a and 199a-5p. J Immunol 2010; 184: 3878–3888.
Steidl C, Lee T, Shah SP, Farinha P, Han G, Nayar T et al. Tumor-associated macrophages and survival in classic Hodgkin's lymphoma. New Engl J Med 2010; 362: 875–885.
Lee SK, Han YM, Yun J, Lee CW, Shin DS, Ha YR et al. Phosphatase of regenerating liver-3 promotes migration and invasion by upregulating matrix metalloproteinases-7 in human colorectal cancer cells. Int J Cancer 2012; 131: E190–E203.
Schroder A, Kirwan TP, Jiang JX, Aitken KJ, Bagli DJ . Rapamycin attenuates bladder hypertrophy during long-term outlet obstruction in vivo: tissue, matrix and mechanistic insights. J Urol 2013; 189: 2377–2384.
Megias J, Busserolles J, Alcaraz MJ . The carbon monoxide-releasing molecule CORM-2 inhibits the inflammatory response induced by cytokines in Caco-2 cells. Br J Pharmacol 2007; 150: 977–986.
Gearing AJ, Beckett P, Christodoulou M, Churchill M, Clements J, Davidson AH et al. Processing of tumour necrosis factor-alpha precursor by metalloproteinases. Nature 1994; 370: 555–557.
Yu WH, Woessner Jr. JF, McNeish JD, Stamenkovic I . CD44 anchors the assembly of matrilysin/MMP-7 with heparin-binding epidermal growth factor precursor and ErbB4 and regulates female reproductive organ remodeling. Genes Dev 2002; 16: 307–323.
Chu J, Pratico D . Pharmacologic blockade of 5-lipoxygenase improves the amyloidotic phenotype of an Alzheimer's disease transgenic mouse model involvement of gamma-secretase. Am J Pathol 2011; 178: 1762–1769.
Keith B, Simon MC . Hypoxia-inducible factors, stem cells, and cancer. Cell 2007; 129: 465–472.
Negus RP, Turner L, Burke F, Balkwill FR . Hypoxia down-regulates MCP-1 expression: implications for macrophage distribution in tumors. J Leukoc Biol 1998; 63: 758–765.
Horiuchi A, Hayashi T, Kikuchi N, Hayashi A, Fuseya C, Shiozawa T et al. Hypoxia upregulates ovarian cancer invasiveness via the binding of HIF-1alpha to a hypoxia-induced, methylation-free hypoxia response element of S100A4 gene. Int J Cancer 2012; 131: 1755–1767.
Imai T, Horiuchi A, Wang C, Oka K, Ohira S, Nikaido T et al. Hypoxia attenuates the expression of E-cadherin via up-regulation of SNAIL in ovarian carcinoma cells. Am J Pathol 2003; 163: 1437–1447.
Kummer NT, Nowicki TS, Azzi JP, Reyes I, Iacob C, Xie S et al. Arachidonate 5 lipoxygenase expression in papillary thyroid carcinoma promotes invasion via MMP-9 induction. J Cell Biochem 2012; 113: 1998–2008.
Chang MC, Chen CA, Chen PJ, Chiang YC, Chen YL, Mao TL et al. Mesothelin enhances invasion of ovarian cancer by inducing MMP-7 through MAPK/ERK and JNK pathways. Biochem J 2012; 442: 293–302.
Ashwell JD . The many paths to p38 mitogen-activated protein kinase activation in the immune system. Nat Rev Immunol 2006; 6: 532–540.
Wagner EF, Nebreda AR . Signal integration by JNK and p38 MAPK pathways in cancer development. Nat Rev Cancer 2009; 9: 537–549.
Murdoch C, Lewis CE . Macrophage migration and gene expression in response to tumor hypoxia. Int J Cancer 2005; 117: 701–708.
Burke B, Giannoudis A, Corke KP, Gill D, Wells M, Ziegler-Heitbrock L et al. Hypoxia-induced gene expression in human macrophages: implications for ischemic tissues and hypoxia-regulated gene therapy. Am J Pathol 2003; 163: 1233–1243.
Kioi M, Yamamoto K, Higashi S, Koshikawa N, Fujita K, Miyazaki K . Matrilysin (MMP-7) induces homotypic adhesion of human colon cancer cells and enhances their metastatic potential in nude mouse model. Oncogene 2003; 22: 8662–8670.
Charles KA, Kulbe H, Soper R, Escorcio-Correia M, Lawrence T, Schultheis A et al. The tumor-promoting actions of TNF-alpha involve TNFR1 and IL-17 in ovarian cancer in mice and humans. J Clin Invest 2009; 119: 3011–3023.
Moore RJ, Owens DM, Stamp G, Arnott C, Burke F, East N et al. Mice deficient in tumor necrosis factor-alpha are resistant to skin carcinogenesis. Nat Med 1999; 5: 828–831.
Wen ZH, Su YC, Lai PL, Zhang Y, Xu YF, Zhao A et al. Critical role of arachidonic acid-activated mTOR signaling in breast carcinogenesis and angiogenesis. Oncogene 2013; 32: 160–170.
Pidgeon GP, Lysaght J, Krishnamoorthy S, Reynolds JV, O'Byrne K, Nie D et al. Lipoxygenase metabolism: roles in tumor progression and survival. Cancer Metastasis Rev 2007; 26: 503–524.
Camp RL, Rimm EB, Rimm DL . Met expression is associated with poor outcome in patients with axillary lymph node negative breast carcinoma. Cancer 1999; 86: 2259–2265.
Brigatte P, Cury Y, de Souza BM, Baptista-Saidemberg NB, Saidemberg DM, Gutierrez VP et al. Hyperalgesic and edematogenic effects of peptides isolated from the venoms of honeybee (Apis mellifera) and neotropical social wasps (Polybia paulista and Protonectarina sylveirae). Amino Acids 2011; 40: 101–111.
Johnson N, Li YC, Walton ZE, Cheng KA, Li D, Rodig SJ et al. Compromised CDK1 activity sensitizes BRCA-proficient cancers to PARP inhibition. Nat Med 2011; 17: 875–882.
Phuangsab A, Lorence RM, Reichard KW, Peeples ME, Walter RJ . Newcastle disease virus therapy of human tumor xenografts: antitumor effects of local or systemic administration. Cancer Lett 2001; 172: 27–36.
Acknowledgements
We thank Colin D Funk, Department of Physiology and Biochemistry, Queen’s University for providing us with essential material and a technical guide. This study was supported by National Natural Science Foundation of China (Nos. 31270971, 81072406).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Wen, Z., Liu, H., Li, M. et al. Increased metabolites of 5-lipoxygenase from hypoxic ovarian cancer cells promote tumor-associated macrophage infiltration. Oncogene 34, 1241–1252 (2015). https://doi.org/10.1038/onc.2014.85
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.85
This article is cited by
-
Lipid metabolism reprogramming in tumor-associated macrophages and implications for therapy
Lipids in Health and Disease (2023)
-
Knock-out of 5-lipoxygenase in overexpressing tumor cells—consequences on gene expression and cellular function
Cancer Gene Therapy (2023)
-
The effects of MYC on tumor immunity and immunotherapy
Cell Death Discovery (2023)
-
Alleviating hypoxia to improve cancer immunotherapy
Oncogene (2023)
-
A study of the correlation between M2 macrophages and lymph node metastasis of colorectal carcinoma
World Journal of Surgical Oncology (2021)