Abstract
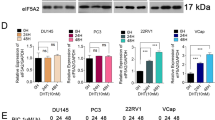
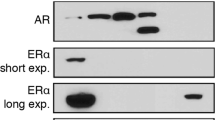
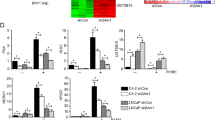
Estrogen receptor β (ERβ) is emerging as a critical factor in understanding prostate cancer biology. Although reduced in prostate cancer above Gleason grade 3, ERβ is a potential drug target at the initial stage of the disease. In human prostate cancer cells, we found that ERβ causes apoptosis by increasing the expression of pro-apoptotic factor p53-upregulated modulator of apoptosis (PUMA), independent of p53, but dependent on the forkhead transcription factor class-O family member, FOXO3a. FOXO3a has previously been shown to induce PUMA after growth factor withdrawal and inhibition of the Akt pathway. Surprisingly, the phosphorylation of FOXO3a remained unchanged, while the mRNA and total protein levels of FOXO3a were increased in response to ERβ expression or treatment of PC3, 22Rv1 and LNCaP cells with the ERβ-specific ligands 3β-Adiol (5α-androstane-3β,17β-diol), DPN (diarylpropionitrile) or 8β-VE2 (8-vinylestra-1,3,5 (10)-triene-3,17β-diol). Knockdown of FOXO3a or ERβ expression abolished the increase of PUMA in response to 3β-Adiol in LNCaP and PC3 cells, suggesting that FOXO3a mediates the apoptotic effect of 3β-Adiol-activated ERβ. Moreover, the ventral prostate of ERβ−/− mice had decreased expression of FOXO3a and PUMA compared with the ERβ+/+ mice, indicating a relationship between ERβ and FOXO3a expression. The regulation of FOXO3a by ERβ in normal basal epithelial cells indicates a function of ERβ in cell differentiation and maintenance of cells in a quiescent state. In addition, the expression of ERβ, FOXO3a and PUMA is comparable and higher in benign prostatic hyperplasia than in prostate cancer Gleason grade 4 or higher, where there is substantial loss of ERβ, FOXO3a and PUMA. We conclude that ERβ induces apoptosis of prostate cancer cells by increasing transcription of FOXO3a, leading to an increase of PUMA and subsequent triggering of apoptosis via the intrinsic pathway involving caspase-9. Furthermore, we conclude that ligands specifically activating ERβ could be useful pharmaceuticals in the treatment of prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout











Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A . Cancer statistics 2012 CA Cancer J Clin 2012; 62: 10–29.
Cornforth AN, Davis JS, Khanifar E, Nastiuk KL, Krolewski JJ . FOXO3a mediates the androgen-dependent regulation of FLIP and contributes to TRAIL-induced apoptosis of LNCaP cells. Oncogene 2008; 27: 4422–4433.
Yu J, Zhang L, Hwang PM, Kinzler KW, Vogelstein B . PUMA induces the rapid apoptosis of colorectal cancer cells. Mol Cell 2001; 7: 673–682.
You H, Pellegrini M, Tsuchihara K, Yamamoto K, Hacker G, Erlacher M et al. FOXO3a-dependent regulation of Puma in response to cytokine/growth factor withdrawal. J Exper Med 2006; 203: 1657–1663.
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999; 96: 857–868.
Tran H, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS et al. DNA repair pathway stimulated by the forkhead transcription factor FOXO3a through the Gadd45 protein. Science 2002; 296: 530–534.
Yamamoto M, Kondo E, Takeuchi M, Harashima A, Otani T, Tsuji-Takayama K et al. miR-155, a modulator of FOXO3a protein expression, is underexpressed and cannot be upregulated by stimulation of HOZOT, a line of multifunctional Treg. PLoS One 2011; 6: e16841.
Zou Y, Tsai WB, Cheng CJ, Hsu C, Chung YM, Li PC et al. Forkhead box transcription factor FOXO3a suppresses estrogen-dependent breast cancer cell proliferation and tumorigenesis. Breast Cancer Res 2008; 10: R21.
Giroux V, Bernatchez G, Carrier JC . Chemopreventive effect of ERbeta-Selective agonist on intestinal tumorigenesis in Apc(Min/+) mice. Mol Carcinogen 2011; 50: 359–369.
Giroux V, Lemay F, Bernatchez G, Robitaille Y, Carrier JC . Estrogen receptor beta deficiency enhances small intestinal tumorigenesis in ApcMin/+ mice. International journal of cancer. J Int Cancer 2008; 123: 303–311.
Saleiro D, Murillo G, Lubahn DB, Kopelovich L, Korach KS, Mehta RG et al. Enhanced induction of mucin-depleted foci in estrogen receptor {beta} knockout mice. Cancer Prev Res (Phila) 2010; 3: 1198–1204.
Slusarz A, Jackson GA, Day JK, Shenouda NS, Bogener JL, Browning JD et al. Aggressive prostate cancer is prevented in ERalphaKO mice and stimulated in ERbetaKO TRAMP mice. Endocrinology 2012; 153: 4160–4170.
Bossard C, Busson M, Vindrieux D, Gaudin F, Machelon V, Brigitte M et al. Potential role of estrogen receptor Beta as a tumor suppressor of epithelial ovarian cancer. PLoS One 2012; 7: e44787.
Dey P, Jonsson P, Hartman J, Williams C, Ström A, Gustafsson JÅ et al. Estrogen receptors beta1 and beta2 have opposing roles in regulating proliferation and bone metastasis genes in the prostate cancer cell line PC3. Mol Endocrinol 2012; 26: 1991–2003.
Paruthiyil S, Parmar H, Kerekatte V, Cunha GR, Firestone GL, Leitman DC et al. Estrogen receptor beta inhibits human breast cancer cell proliferation and tumor formation by causing a G2 cell cycle arrest. Cancer Res 2004; 64: 423–428.
Strom A, Hartman J, Foster JS, Kietz S, Wimalasena J, Gustafsson JA et al. Estrogen receptor beta inhibits 17beta-estradiol-stimulated proliferation of the breast cancer cell line T47D. Proc Natl Acad Sci USA 2004; 101: 1566–1571.
McPherson SJ, Hussain S, Balanathan P, Hedwards SL, Niranjan B, Grant M et al. Estrogen receptor-beta activated apoptosis in benign hyperplasia and cancer of the prostate is androgen independent and TNFalpha mediated. Proc Natl Acad Sci USA 2010; 107: 3123–3128.
Hussain S, Lawrence MG, Taylor RA, Lo CY, Frydenberg M, Risbridger GP et al. Estrogen receptor beta activation impairs prostatic regeneration by inducing apoptosis in murine and human stem/progenitor enriched cell populations. PLoS One 2012; 7: e40732.
Hodges-Gallagher L, Valentine CD, El Bader S, Kushner PJ . Estrogen receptor beta increases the efficacy of antiestrogens by effects on apoptosis and cell cycling in breast cancer cells. Breast Cancer Res Treat 2008; 109: 241–250.
Ji Q, Liu PI, Elshimali Y, Stolz A . Frequent loss of estrogen and progesterone receptors in human prostatic tumors determined by quantitative real-time PCR. Mol Cell Endocrinol 2005; 229: 103–110.
Leung YK, Lam HM, Wu S, Song D, Levin L, Cheng L et al. Estrogen receptor beta2 and beta5 are associated with poor prognosis in prostate cancer, and promote cancer cell migration and invasion. Endocrine Relat Cancer 2010; 17: 675–689.
Oliveira AG, Coelho PH, Guedes FD, Mahecha GA, Hess RA, Oliveira CA et al. 5alpha-Androstane-3beta,17beta-diol (3beta-diol), an estrogenic metabolite of 5alpha-dihydrotestosterone, is a potent modulator of estrogen receptor ERbeta expression in the ventral prostrate of adult rats. Steroids 2007; 72: 914–922.
Weihua Z, Lathe R, Warner M, Gustafsson JA . An endocrine pathway in the prostate, ERbeta, AR, 5alpha-androstane-3beta,17beta-diol, and CYP7B1, regulates prostate growth. Proc Natl Acad Sci USA 2002; 99: 13589–13594.
Kandel ES, Hay N . The regulation and activities of the multifunctional serine/threonine kinase Akt/PKB. Exp Cell Res 1999; 253: 210–229.
Greer EL, Brunet A . FOXO transcription factors at the interface between longevity and tumor suppression. Oncogene 2005; 24: 7410–7425.
Kuiper GG, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JA . Cloning of a novel receptor expressed in rat prostate and ovary. Proc Natl Acad Sci USA 1996; 93: 5925–5930.
Cvoro A, Paruthiyil S, Jones JO, Tzagarakis-Foster C, Clegg NJ, Tatomer D et al. Selective activation of estrogen receptor-beta transcriptional pathways by an herbal extract. Endocrinology 2007; 148: 538–547.
Cvoro A, Tzagarakis-Foster C, Tatomer D, Paruthiyil S, Fox MS, Leitman DC et al. Distinct roles of unliganded and liganded estrogen receptors in transcriptional repression. Mol Cell 2006; 21: 555–564.
Ellem SJ, Risbridger GP . The dual, opposing roles of estrogen in the prostate. Ann NY Acad Sci 2009; 1155: 174–186.
Mak P, Leav I, Pursell B, Bae D, Yang X, Taglienti CA et al. ERbeta impedes prostate cancer EMT by destabilizing HIF-1alpha and inhibiting VEGF-mediated snail nuclear localization: implications for Gleason grading. Cancer Cell 2010; 17: 319–332.
Dondi D, Piccolella M, Biserni A, Della Torre S, Ramachandran B, Locatelli A et al. Estrogen receptor beta and the progression of prostate cancer: role of 5alpha-androstane-3beta,17beta-diol. Endocrine Relat Cancer 2010; 17: 731–742.
Saijo K, Collier JG, Li AC, Katzenellenbogen JA, Glass CK . An ADIOL-ERbeta-CtBP transrepression pathway negatively regulates microglia-mediated inflammation. Cell 2011; 145: 584–595.
Muthusamy S, Andersson S, Kim HJ, Butler R, Waage L, Gustafsson JÅ et al. Estrogen receptor beta and 17beta-hydroxysteroid dehydrogenase type 6, a growth regulatory pathway that is lost in prostate cancer. Proc Natl Acad Sci USA 2011; 108: 20090–20094.
Olsson M, Gustafsson O, Skogastierna C, Tolf A, Rietz BD, Morfin R et al. Regulation and expression of human CYP7B1 in prostate: overexpression of CYP7B1 during progression of prostatic adenocarcinoma. Prostate 2007; 67: 1439–1446.
Nakano K, Vousden KH . PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell 2001; 7: 683–694.
Roberts CG, Millar EK, O'Toole SA, McNeil CM, Lehrbach GM, Pinese M et al. Identification of PUMA as an estrogen target gene that mediates the apoptotic response to tamoxifen in human breast cancer cells and predicts patient outcome and tamoxifen responsiveness in breast cancer. Oncogene 2011; 30: 3186–3197.
Hartman J, Lindberg K, Morani A, Inzunza J, Ström A, Gustafsson JA et al. Estrogen receptor beta inhibits angiogenesis and growth of T47D breast cancer xenografts. Cancer Res 2006; 66: 11207–11213.
Hartman J, Edvardsson K, Lindberg K, Zhao C, Williams C, Ström A et al. Tumor repressive functions of estrogen receptor beta in SW480 colon cancer cells. Cancer Res 2009; 69: 6100–6106.
Maggiolini M, Recchia AG, Carpino A, Vivacqua A, Fasanella G, Rago V et al. Oestrogen receptor beta is required for androgen-stimulated proliferation of LNCaP prostate cancer cells. J Mol Endocrinol 2004; 32: 777–791.
Nanni S, Benvenuti V, Grasselli A, Priolo C, Aiello A, Mattiussi S et al. Endothelial NOS, estrogen receptor beta, and HIFs cooperate in the activation of a prognostic transcriptional pattern in aggressive human prostate cancer. J Clin Invest 2009; 119: 1093–1108.
Dignam JD, Lebovitz RM, Roeder RG . Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res 1983; 11: 1475–1489.
Hartman J, Müller P, Foster JS, Wimalasena J, Gustafsson JA, Ström A et al. HES-1 inhibits 17beta-estradiol and heregulin-beta1-mediated upregulation of E2F-1. Oncogene 2004; 23: 8826–8833.
Helguero LA, Hedengran Faulds M, Forster C, Gustafsson JA, Haldosen LA . DAX-1 expression is regulated during mammary epithelial cell differentiation. Endocrinology 2006; 147: 3249–3259.
Acknowledgements
This work was supported by The Cancer Prevention and Research Institute of Texas (CPRIT) grants HIRP100680 and RP110444, the Texas Emerging Technology Fund under Agreement number 300-9-1958, the Robert A. Welch Foundation (E-0004) and the Swedish Cancer Fund. We are grateful to Linda Waage, Department of Urology at Danderyd Hospital, Stockholm, Sweden, for providing us the human prostate cancer samples, and Ulf Bergerheim for Gleason grading the tissue samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Dey, P., Ström, A. & Gustafsson, JÅ. Estrogen receptor β upregulates FOXO3a and causes induction of apoptosis through PUMA in prostate cancer. Oncogene 33, 4213–4225 (2014). https://doi.org/10.1038/onc.2013.384
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.384
Keywords
This article is cited by
-
Contribution of microRNA-30d to the prevention of the thyroid cancer occurrence and progression: mechanism and implications
Apoptosis (2023)
-
Deciphering sex-specific miRNAs as heat-recorders in zebrafish
Scientific Reports (2022)
-
Association of estrogen receptor β polymorphisms with posterior tibial tendon dysfunction
Molecular and Cellular Biochemistry (2020)
-
Circulating IGF-1 promotes prostate adenocarcinoma via FOXO3A/BIM signaling in a double-transgenic mouse model
Oncogene (2019)
-
ZFHX3 is indispensable for ERβ to inhibit cell proliferation via MYC downregulation in prostate cancer cells
Oncogenesis (2019)