Abstract
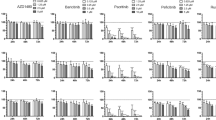
Primary effusion lymphoma (PEL) is an aggressive form of non-Hodgkin’s B-cell lymphoma associated with infection by Kaposi’s sarcoma-associated herpes virus (KSHV). (+)-JQ1 and I-BET151 are two recently described novel small-molecule inhibitors of BET bromodomain chromatin-associated proteins that have shown impressive preclinical activity in cancers in which MYC is overexpressed at the transcriptional level due to chromosomal translocations that bring the MYC gene under the control of a super-enhancer. PEL cells, in contrast, lack structural alterations in the MYC gene, but have deregulated Myc protein due to the activity of KSHV-encoded latent proteins. We report that PEL cell lines are highly sensitive to bromodomain and extra-terminal (BET) bromodomain inhibitors-induced growth inhibition and undergo G0/G1 cell-cycle arrest, apoptosis and cellular senescence, but without the induction of lytic reactivation, upon treatment with these drugs. Treatment of PEL cell lines with BET inhibitors suppressed the expression of MYC and resulted in a genome-wide perturbation of MYC-dependent genes. Silencing of BRD4 and MYC expression blocked cell proliferation and cell-cycle progression, while ectopic expression of MYC from a retroviral promoter rescued cells from (+)-JQ1-induced growth arrest. In a xenograft model of PEL, (+)-JQ1 significantly reduced tumor growth and improved survival. Taken collectively, our results demonstrate that the utility of BET inhibitors may not be limited to cancers in which genomic alterations result in extremely high expression of MYC and they may have equal or perhaps greater activity against cancers in which the MYC genomic locus is structurally intact and c-Myc protein is deregulated at the post-translational level and is only modestly overexpressed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Morris K . Cancer? In Africa? Lancet Oncol 2003; 4: 5.
Wabinga HR, Parkin DM, Wabwire-Mangen F, Mugerwa JW . Cancer in Kampala, Uganda, in 1989-91: changes in incidence in the era of AIDS. Int J Cancer 1993; 54: 26–36.
Nador RG, Cesarman E, Chadburn A, Dawson DB, Ansari MQ, Sald J et al. Primary effusion lymphoma: a distinct clinicopathologic entity associated with the Kaposi's sarcoma-associated herpes virus. Blood 1996; 88: 645–656.
Boulanger E, Daniel MT, Agbalika F, Oksenhendler E . Combined chemotherapy including high-dose methotrexate in KSHV/HHV8-associated primary effusion lymphoma. Am J Hematol 2003; 73: 143–148.
Boulanger E, Hermine O, Fermand JP, Radford-Weiss I, Brousse N, Meignin V et al. Human herpesvirus 8 (HHV-8)-associated peritoneal primary effusion lymphoma (PEL) in two HIV-negative elderly patients. Am J Hematol 2004; 76: 88–91.
Wu SY, Chiang CM . The double bromodomain-containing chromatin adaptor Brd4 and transcriptional regulation. J Biol Chem 2007; 282: 13141–13145.
Chung CW, Witherington J . Progress in the discovery of small-molecule inhibitors of bromodomain--histone interactions. J Biomol Screen 2011; 16: 1170–1185.
Delmore JE, Issa GC, Lemieux ME, Rahl PB, Shi J, Jacobs HM et al. BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 2011; 146: 904–917.
Zuber J, Shi J, Wang E, Rappaport AR, Herrmann H, Sison EA et al. RNAi screen identifies Brd4 as a therapeutic target in acute myeloid leukaemia. Nature 2011; 478: 524–528.
Dawson MA, Prinjha RK, Dittmann A, Giotopoulos G, Bantscheff M, Chan WI et al. Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature 2011; 478: 529–533.
Ott CJ, Kopp N, Bird L, Paranal RM, Qi J, Bowman T et al. BET bromodomain inhibition targets both c-MYC and IL7R in high-risk acute lymphoblastic leukemia. Blood 2012; 120: 2843–2852.
Filippakopoulos P, Qi J, Picaud S, Shen Y, Smith WB, Fedorov O et al. Selective inhibition of BET bromodomains. Nature 2010; 468: 1067–1073.
Nicodeme E, Jeffrey KL, Schaefer U, Beinke S, Dewell S, Chung CW et al. Suppression of inflammation by a synthetic histone mimic. Nature 2010; 468: 1119–1123.
Mertz JA, Conery AR, Bryant BM, Sandy P, Balasubramanian S, Mele DA et al. Targeting MYC dependence in cancer by inhibiting BET bromodomains. Proc Natl Acad Sci USA 2011; 108: 16669–16674.
Loven J, Hoke HA, Lin CY, Lau A, Orlando DA, Vakoc CR et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell 2013; 153: 320–334.
Li X, Chen S, Feng J, Deng H, Sun R . Myc is required for the maintenance of Kaposi's sarcoma-associated herpesvirus latency. J Virol 2010; 84: 8945–8948.
Bubman D, Guasparri I, Cesarman E . Deregulation of c-Myc in primary effusion lymphoma by Kaposi's sarcoma herpesvirus latency-associated nuclear antigen. Oncogene 2007; 26: 4979–4986.
Liu J, Martin HJ, Liao G, Hayward SD . The Kaposi sarcoma associated herpesvirus lana protein stabilizes and activates c-Myc. J Virol 2007; 81: 10451–10459.
Lubyova B, Kellum MJ, Frisancho JA, Pitha PM . Stimulation of c-Myc transcriptional activity by vIRF-3 of Kaposi sarcoma-associated herpesvirus. J Biol Chem 2007; 282: 31944–31953.
Bhatt S, Ashlock B, Natkunam Y, Ramos JC, Mesri EA, Lossos IS Preclinical Activity of Brentuximab Vedotin (SGN-35) in primary effusion lymphoma (PEL); 53rd ASH Annual Meeting and Exposition; San Diego, CA, USA, 2011.
Sarosiek KA, Cavallin LE, Bhatt S, Toomey NL, Natkunam Y, Blasini W et al. Efficacy of bortezomib in a direct xenograft model of primary effusion lymphoma. Proc Natl Acad Sci U S A. 2010; 107: 13069–13074.
Zhao H, Darzynkiewicz Z . Biomarkers of cell senescence assessed by imaging cytometry. Methods Mol Biol 2013; 965: 83–92.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 2005; 102: 15545–15550.
Li Z, Van Calcar S, Qu C, Cavenee WK, Zhang MQ, Ren B . A global transcriptional regulatory role for c-Myc in Burkitt's lymphoma cells. Proc Natl Acad Sci USA 2003; 100: 8164–8169.
Fernandez PC, Frank SR, Wang L, Schroeder M, Liu S, Greene J et al. Genomic targets of the human c-Myc protein. Genes Dev 2003; 17: 1115–1129.
Nakamura H, Lu M, Gwack Y, Souvlis J, Zeichner SL, Jung JU . Global changes in Kaposi's sarcoma-associated virus gene expression patterns following expression of a tetracycline-inducible Rta transactivator. J Virol 2003; 77: 4205–4220.
Zhao J, Punj V, Matta H, Mazzacurati L, Schamus S, Yang Y et al. K13 Blocks KSHV Lytic Replication and Deregulates vIL6 and hIL6 Expression: a Model of Lytic Replication Induced Clonal Selection in Viral Oncogenesis. PLoS One 2007; 2: e1067.
Dang CV, Le A, Gao P . MYC-induced cancer cell energy metabolism and therapeutic opportunities. Clin Cancer Res 2009; 15: 6479–6483.
Pelengaris S, Khan M, Evan G . c-MYC: more than just a matter of life and death. Nat Rev Cancer 2002; 2: 764–776.
Shou Y, Martelli ML, Gabrea A, Qi Y, Brents LA, Roschke A et al. Diverse karyotypic abnormalities of the c-myc locus associated with c-myc dysregulation and tumor progression in multiple myeloma. Proc Natl Acad Sci U S A. 2000; 97: 228–233.
Ballestas ME, Kaye KM . Kaposi's sarcoma-associated herpesvirus latency-associated nuclear antigen 1 mediates episome persistence through cis-acting terminal repeat (TR) sequence and specifically binds TR DNA. J Virol 2001; 75: 3250–3258.
Garber AC, Hu J, Renne R . Latency-associated nuclear antigen (LANA) cooperatively binds to two sites within the terminal repeat, and both sites contribute to the ability of LANA to suppress transcription and to facilitate DNA replication. J Biol Chem 2002; 277: 27401–27411.
You J, Srinivasan V, Denis GV, Harrington WJ Jr., Ballestas ME, Kaye KM et al. Kaposi's sarcoma-associated herpesvirus latency-associated nuclear antigen interacts with bromodomain protein Brd4 on host mitotic chromosomes. J Virol 2006; 80: 8909–8919.
Ottinger M, Christalla T, Nathan K, Brinkmann MM, Viejo-Borbolla A, Schulz TF . Kaposi's sarcoma-associated herpesvirus LANA-1 interacts with the short variant of BRD4 and releases cells from a BRD4- and BRD2/RING3-induced G1 cell cycle arrest. J Virol 2006; 80: 10772–10786.
Nicholas J . Human herpesvirus 8-encoded proteins with potential roles in virus-associated neoplasia. Front Biosci 2007; 12: 265–281.
Bartholomeeusen K, Xiang Y, Fujinaga K, Peterlin BM . BET bromodomain inhibition activates transcription via a transient release of P-TEFb from 7SK snRNP. J Biol Chem 2012; 287: 36609–36616.
Zhu X, Santat LA, Chang MS, Liu J, Zavzavadjian JR, Wall EA et al. A versatile approach to multiple gene RNA interference using microRNA-based short hairpin RNAs. BMC Mol Biol 2007; 8: 98.
Shin KJ, Wall EA, Zavzavadjian JR, Santat LA, Liu J, Hwang JI et al. A single lentiviral vector platform for microRNA-based conditional RNA interference and coordinated transgene expression. Proc Natl Acad Sci USA 2006; 103: 13759–13764.
Acknowledgements
The authors thank Dr James Bradner (Dana-Farber Cancer Institute, Boston, MA, USA) for his generous contribution of (+)- JQ1 and (−)- JQ1, Dr Peter Howley (Harvard University) for BRD4 antibody, Gary Hayward (Johns Hopkins University) for RTA antibody, and Drs Lossos and Ramos from the University of Miami for UM-PEL-1 and UM-PEL-3 cells, respectively. This work was supported by grants from the National Institutes of Health (CA139119, DE019811 and P30CA014089) and STOP Cancer Foundations. Flow cytometry was performed in the USC Flow Cytometry Core Facility that is supported in part by the National Cancer Institute Cancer Center Shared Grant award P30CA014089 and the USC Provost Office Dean’s Development Funds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Tolani, B., Gopalakrishnan, R., Punj, V. et al. Targeting Myc in KSHV-associated primary effusion lymphoma with BET bromodomain inhibitors. Oncogene 33, 2928–2937 (2014). https://doi.org/10.1038/onc.2013.242
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.242
Keywords
This article is cited by
-
High levels of LINE-1 transposable elements expressed in Kaposi’s sarcoma-associated herpesvirus-related primary effusion lymphoma
Oncogene (2021)
-
A novel thermostable beetle luciferase based cytotoxicity assay
Scientific Reports (2021)
-
KSHV transactivator-derived small peptide traps coactivators to attenuate MYC and inhibits leukemia and lymphoma cell growth
Communications Biology (2021)
-
Narciclasine, an isocarbostyril alkaloid, has preferential activity against primary effusion lymphoma
Scientific Reports (2020)
-
BRD4 modulates vulnerability of triple-negative breast cancer to targeting of integrin-dependent signaling pathways
Cellular Oncology (2020)