Abstract
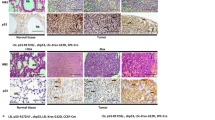
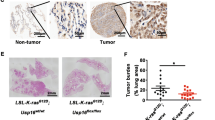
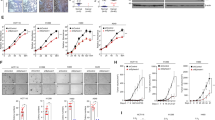
p27Kip1 (p27) can have opposing roles during malignant transformation depending on cellular context: on one hand it functions as a tumor suppressor by inhibiting cyclin–cyclin-dependent kinase (CDK) activity in the nucleus and on the other it may adopt an oncogenic role that is less well understood. To gain further insight into the roles played by p27 during tumorigenesis, we compared the susceptibility with urethane-induced tumorigenesis of two p27 mouse models, p27−/− and p27CK− knockin, in which p27 cannot bind or inhibit cyclin–CDKs. In this K-Ras-driven tumorigenesis model, p27CK− mice had an increase in both tumor number and aggressiveness compared with p27−/−, indicating a cooperation between p27CK− and activated Ras. In the lung, increased tumorigenesis was associated with cytoplasmic localization of p27CK− and bronchiolaveolar stem cell amplification. The ability of p27CK− to cooperate with other oncogenes was not universal. When c-Myc was used as a transforming agent, p27 status became irrelevant and c-Myc was equally potent in transforming p27+/+, p27−/− and p27CK− cells. In fact, c-Myc induced the degradation of wild-type p27 via the Skp-Cullin-F-box (SCF)–Skp2 pathway. In contrast, p27CK− levels were not affected by c-Myc expression, as p27CK− is insensitive to Skp2-mediated degradation because of its inability to bind cyclin E/CDK2. However, in presence of c-Myc, p27CK− remained mostly nuclear, providing an explanation for its inability to cooperate with Myc during transformation. Thus, we propose that the p27CK− protein needs to be localized in the cytoplasm in order to function as an oncogene, otherwise it just behaves similar to a null allele.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Berns K, Martins C, Dannenberg JH, Berns A, Te Riele H, Bernards R . (2000). p27kip1-independent cell cycle regulation by MYC. Oncogene 19: 4822–4827.
Besson A, Assoian RK, Roberts JM . (2004a). Regulation of the cytoskeleton: an oncogenic function for CDK inhibitors? Nat Rev Cancer 4: 948–955.
Besson A, Dowdy SF, Roberts JM . (2008). CDK inhibitors: cell cycle regulators and beyond. Dev Cell 14: 159–169.
Besson A, Gurian-West M, Chen X, Kelly-Spratt KS, Kemp CJ, Roberts JM . (2006). A pathway in quiescent cells that controls p27Kip1 stability, subcellular localization and tumor suppression. Genes Dev 20: 47–64.
Besson A, Gurian-West M, Schmidt A, Hall A, Roberts JM . (2004b). p27Kip1 modulates cell migration through the regulation of RhoA activation. Genes Dev 18: 862–876.
Besson A, Hwang HC, Donovan SL, Cicero S, Gurian-West M, Johnson D et al. (2007). Discovery of an oncogenic activity in p27Kip1 that causes stem cell expansion and a multiple tumor phenotype. Genes Dev 21: 1731–1746.
Boehm M, Yoshimoto T, Crook MF, Nallamshetty S, True A, Nabel GJ et al. (2002). A growth factor-dependent nuclear kinase phosphorylates p27Kip1 and regulates cell cycle progression. EMBO J 21: 3390–3401.
Bouchard C, Thieke K, Maier A, Saffrich R, Hanley-Hyde J, Ansorge W et al. (1999). Direct induction of cyclin D2 by Myc contributes to cell cycle progression and sequestration of p27. EMBO J 18: 5321–5333.
Chu IM, Hengst L, Slingerland JM . (2008). The Cdk inhibitor p27 in human cancer: prognostic potential and relevance to anticancer therapy. Nat Rev Cancer 8: 253–267.
Chu S, McDonald T, Bhatia R . (2010). Role of BCR-ABL-Y177-mediated p27kip1 phosphorylation and cytoplasmic localization in enhanced proliferation of chronic myeloid leukemia progenitors. Leukemia 24: 779–787.
Condeelis J, Pollard JW . (2006). Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124: 263–266.
Connor MK, Kotchetkov R, Cariou S, Resch A, Lupetti R, Beniston RG et al. (2003). CRM1/Ran-mediated nuclear export of p27(Kip1) involves a nuclear export signal and links p27 export and proteolysis. Mol Biol Cell 14: 201–213.
Denicourt C, Saenz CC, Datnow B, Cui XS, Dowdy SF . (2007). Relocalized p27Kip1 tumor suppressor functions as a cytoplasmic metastatic oncogene in melanoma. Cancer Res 67: 9238–9243.
Dovey JS, Zacharek SJ, Kim CF, Lees JA . (2008). Bmi1 is critical for lung tumorigenesis and bronchioalveolar stem cell expansion. Proc Natl Acad Sci USA 105: 11857–11862.
Fero ML, Randel E, Gurley KE, Roberts JM, Kemp CJ . (1998). The murine gene p27Kip1 is haplo-insufficient for tumour suppression. Nature 396: 177–180.
Fero ML, Rivkin M, Tasch M, Porter P, Carow CE, Firpo E et al. (1996). A syndrome of multiorgan hyperplasia with features of gigantism, tumorigenesis, and female sterility in p27(Kip1)-deficient mice. Cell 85: 733–744.
Frescas D, Pagano M . (2008). Deregulated proteolysis by the F-box proteins SKP2 and beta-TrCP: tipping the scales of cancer. Nat Rev Cancer 8: 438–449.
Fujita N, Sato S, Katayama K, Tsuruo T . (2002). Akt-dependent phosphorylation of p27Kip1 promotes binding to 14-3-3 and cytoplasmic localization. J Biol Chem 277: 28706–28713.
Fujita N, Sato S, Tsuruo T . (2003). Phosphorylation of p27Kip1 at threonine 198 by p90 ribosomal protein S6 kinases promotes its binding to 14-3-3 and cytoplasmic localization. J Biol Chem 278: 49254–49260.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Hattori T, Isobe T, Abe K, Kikuchi H, Kitagawa K, Oda T et al. (2007). Pirh2 promotes ubiquitin-dependent degradation of the cyclin-dependent kinase inhibitor p27Kip1. Cancer Res 67: 10789–10795.
Hermeking H, Rago C, Schuhmacher M, Li Q, Barrett JF, Obaya AJ et al. (2000). Identification of CDK4 as a target of c-MYC. Proc Natl Acad Sci USA 97: 2229–2234.
Hong F, Larrea MD, Doughty C, Kwiatkowski DJ, Squillace R, Slingerland JM . (2008). mTOR-raptor binds and activates SGK1 to regulate p27 phosphorylation. Mol Cell 30: 701–711.
Hwang HC, Martins CP, Bronkhorst Y, Randel E, Berns A, Fero M et al. (2002). Identification of oncogenes collaborating with p27Kip1 loss by insertional mutagenesis and high-throughput insertion site analysis. Proc Natl Acad Sci USA 99: 11293–11298.
Ishida N, Hara T, Kamura T, Yoshida M, Nakayama K, Nakayama KI . (2002). Phosphorylation of p27Kip1on serine 10 is required for its binding to CRM1 and nuclear export. J Biol Chem 277: 14355–14358.
Kamura T, Hara T, Matsumoto M, Ishida N, Okumura F, Hatakeyama S et al. (2004). Cytoplasmic ubiquitin ligase KPC regulates proteolysis of p27(Kip1) at G1 phase. Nat Cell Biol 6: 1229–1235.
Kawauchi T, Chihama K, Nabeshima Y, Hoshino M . (2006). Cdk5 phosphorylates and stabilizes p27kip1 contributing to actin organization and cortical neuronal migration. Nat Cell Biol 8: 17–26.
Keller UB, Old JB, Dorsey FC, Nilsson JA, Nilsson L, MacLean KH et al. (2007). Myc targets Cks1 to provoke the suppression of p27Kip1, proliferation and lymphomagenesis. EMBO J 26: 2562–2574.
Kelly-Spratt KS, Philipp-Staheli J, Gurley KE, Hoon-Kim K, Knoblaugh S, Kemp CJ . (2009). Inhibition of PI-3K restores nuclear p27Kip1 expression in a mouse model of Kras-driven lung cancer. Oncogene 28: 3652–3662.
Kfir S, Ehrlich M, Goldshmid A, Liu X, Kloog Y, Henis YI . (2005). Pathway- and expression level -dependent effects of oncogenic N-Ras: p27Kip1 mislocalization by the Ral-GEF pathway and Erk-mediated interference with Smad signaling. Mol Cell Biol 25: 8239–8250.
Kim CF, Jackson EL, Woolfenden AE, Lawrence S, Babar I, Vogel S et al. (2005). Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 121: 823–835.
Larrea MD, Hong F, Wander SA, da Silva TG, Helfman D, Lannigan D et al. (2009). RSK1 drives p27Kip1 phosphorylation at T198 to promote RhoA inhibition and increase cell motility. Proc Natl Acad Sci USA 106: 9268–9273.
Leone G, DeGregori J, Sears R, Jakoi L, Nevins JR . (1997). Myc and Ras collaborate in inducing accumulation of active cyclin E/Cdk2 and E2F. Nature 387: 422–426.
Li Z, Jiao X, Wang C, Ju X, Lu Y, Yuan L et al. (2006). Cyclin D1 Induction of Cellular Migration Requires p27KIP1. Cancer Res 66: 9986–9994.
Liang J, Zubovitch J, Petrocelli T, Kotchetkov R, Connor MK, Han K et al. (2002). PKB/Akt phosphorylates p27, impairs nuclear import of p27 and opposes p27-mediated G1 arrest. Nat Med 8: 1153–1160.
Liu X, Sun Y, Ehrlich M, Lu T, Kloog Y, Weinberg RA et al. (2000). Disruption of TGF-beta growth inhibition by oncogenic ras is linked to p27Kip1 mislocalization. Oncogene 19: 5926–5935.
Malek NP, Sundberg H, McGrew S, Nakayama K, Kyriakides TR, Roberts JM . (2001). A mouse knock-in model exposes sequential proteolytic pathways that regulate p27Kip1 in G1 and S phase. Nature 413: 323–327.
Martins CP, Berns A . (2002). Loss of p27(Kip1) but not p21(Cip1) decreases survival and synergizes with MYC in murine lymphomagenesis. EMBO J 21: 3739–3748.
Meuwissen R, Berns A . (2005). Mouse models for human lung cancer. Genes Dev 19: 643–664.
Montagnoli A, Fiore F, Eytan E, Carrano AC, Draetta GF, Hershko A et al. (1999). Ubiquitination of p27 is regulated by Cdk-dependent phosphorylation and trimeric complex formation. Genes Dev 13: 1181–1189.
Morishita D, Katayama R, Sekimizu K, Tsuruo T, Fujita N . (2008). Pim kinases promote cell cycle progression by phosphorylating and down-regulating p27Kip1 at the transcriptional and posttranscriptional levels. Cancer Res 68: 5076–5085.
Muller D, Bouchard C, Rudolph B, Steiner P, Stuckmann I, Saffrich R et al. (1997). Cdk2-dependent phosphorylation of p27 facilitates its Myc-induced release from cyclin E/cdk2 complexes. Oncogene 15: 2561–2576.
O'Hagan RC, Ohh M, David G, de Alboran IM, Alt FW, Kaelin Jr WG et al. (2000). Myc-enhanced expression of Cul1 promotes ubiquitin-dependent proteolysis and cell cycle progression. Genes Dev 14: 2185–2191.
Old JB, Kratzat S, Hoellein A, Graf S, Nilsson JA, Nilsson L et al. (2010). Skp2 directs Myc-mediated suppression of p27Kip1 yet has modest effects on Myc-driven lymphomagenesis. Mol Cancer Res 8: 353–362.
Papakonstanti EA, Ridley AJ, Vanhaesebroeck B . (2007). The p110delta isoform of PI 3-kinase negatively controls RhoA and PTEN. EMBO J 26: 3050–3061.
Rodier G, Montaglioni A, Di Marcotullio L, Coulombe P, Draetta GF, Pagano M et al. (2001). p27 cytoplasmic localization is regulated by phosphorylation on Ser 10 and is not a prerequisite for its proteolysis. EMBO J 20: 6672–6682.
Sekimoto T, Fukumoto M, Yoneda Y . (2004). 14-3-3 suppresses the nuclear localization of threonine 157-phosphorylated p27(Kip1). Embo J 23: 1934–1942.
Sherr CJ, Roberts JM . (1999). CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 13: 1501–1512.
Shin I, Rotty J, Wu FY, Arteaga CL . (2005). Phosphorylation of p27Kip1 at Thr-157 interferes with its association with importin {alpha} during G1 and prevents nuclear re-entry. J Biol Chem 280: 6055–6063.
Short JD, Houston KD, Dere R, Cai SL, Kim J, Johnson CL et al. (2008). AMP-activated protein kinase signaling results in cytoplasmic sequestration of p27. Cancer Res 68: 6496–6506.
Wu FY, Wang SE, Sanders ME, Shin I, Rojo F, Baselga J et al. (2006). Reduction of cytosolic p27(Kip1) inhibits cancer cell motility, survival, and tumorigenicity. Cancer Res 66: 2162–2172.
Acknowledgements
MPS is supported by a studentship from the Ministère de l’Enseignement Supérieur et de la Recherche. CC is supported by a fellowship from the Fondation pour la Recherche Médicale. This work was supported by NIH grant #1R01CA118043 to JMR. AB is supported by grants from the Association pour la Recherche sur le Cancer and Ligue Nationale Contre le Cancer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website .
Rights and permissions
About this article
Cite this article
Serres, M., Zlotek-Zlotkiewicz, E., Concha, C. et al. Cytoplasmic p27 is oncogenic and cooperates with Ras both in vivo and in vitro. Oncogene 30, 2846–2858 (2011). https://doi.org/10.1038/onc.2011.9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.9
Keywords
This article is cited by
-
Assessment of survivin and p27 expression as potential prognostic markers in urothelial cell carcinoma of urinary bladder in Egyptian patients
African Journal of Urology (2022)
-
Bacterial protein MakA causes suppression of tumour cell proliferation via inhibition of PIP5K1α/Akt signalling
Cell Death & Disease (2022)
-
p27 controls Ragulator and mTOR activity in amino acid-deprived cells to regulate the autophagy–lysosomal pathway and coordinate cell cycle and cell growth
Nature Cell Biology (2020)
-
Quantitative Systems Biology to decipher design principles of a dynamic cell cycle network: the “Maximum Allowable mammalian Trade–Off–Weight” (MAmTOW)
npj Systems Biology and Applications (2017)
-
Cip2a promotes cell cycle progression in triple-negative breast cancer cells by regulating the expression and nuclear export of p27Kip1
Oncogene (2017)