Abstract
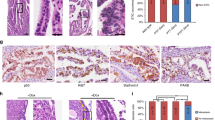
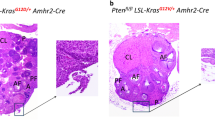
Ovarian cancer is a complex and deadly disease that remains difficult to detect at an early curable stage. Furthermore, although some oncogenic (Kras, Pten/PI3K and Trp53) pathways that are frequently mutated, deleted or amplified in ovarian cancer are known, how these pathways initiate and drive specific morphological phenotypes and tumor outcomes remain unclear. We recently generated Ptenfl/fl; KrasG12D; Amhr2-Cre mice to disrupt the Pten gene and express a stable mutant form of KrasG12D in ovarian surface epithelial (OSE) cells. On the basis of histopathologic criteria, the mutant mice developed low-grade ovarian serous papillary adenocarcinomas at an early age and with 100% penetrance. This highly reproducible phenotype provides the first mouse model in which to study this ovarian cancer subtype. OSE cells isolated from ovaries of mutant mice at 5 and 10 weeks of age exhibit temporal changes in the expression of specific Mullerian epithelial marker genes, grow in soft agar and develop ectopic invasive tumors in recipient mice, indicating that the cells are transformed. Gene profiling identified specific mRNAs and microRNAs differentially expressed in purified OSE cells derived from tumors of the mutant mice compared with wild-type OSE cells. Mapping of transcripts or genes between the mouse OSE mutant data sets, the Kras signature from human cancer cell lines and the human ovarian tumor array data sets, documented significant overlap, indicating that KRAS is a key driver of OSE transformation in this context. Two key hallmarks of the mutant OSE cells in these mice are the elevated expression of the tumor-suppressor Trp53 (p53) and its microRNA target, miR-34a-c. We propose that elevated TRP53 and miR-34a-c may exert negatively regulatory effects that reduce the proliferative potential of OSE cells leading to the low-grade serous adenocarcinoma phenotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Abdullah M, Schultz H, Kähler D, Branscheid D, Dalhoff K, Zabel P et al. (2009). Expression of the acute phase protein haptoglobin in human lung cancer and tumor-free lung tissues. Pathol Res Pract 205: 639–647.
Astanehe A, Arenillas D, Wasserman WW, Leung PC, Dunn SE, Davies BR et al. (2008). Mechanisms underlying p53 regulation of PIK3CA transcription in ovarian surface epithelium and in ovarian cancer. J Cell Sci 121: 664–674.
Auersperg N, Wong AST, Choi K-C, Kang SK, Leung PCK . (2001). Ovarian surface epithelium: biology, endocrinology and pathology. Endocrine Rev 22: 255–288.
Auersperg N, Woo MMM, Gilks CB . (2008). The origin of ovarian carcinomas: a developmental view. Gynecol Oncol 110: 452–454.
Bast RC, Hennessy B, Mills GB . (2009). The biology of ovarian cancer: new opportunities for translation. Nat Rev Cancer 9: 415–428.
Bohlig L, Metzger R, Rother K, Till H, Engeland K . (2008). The CCN3 gene coding an extracellular adhesion-related protein is transcriptionally activated by the p53 tumor suppressor. Cell Cycle 7: 1254–1261.
Bowen NJ, Walker LD, Matyunina L, Logani S, Totten KA, Benigno BB et al. (2009). Gene expression profiling supports the hypothesis that human ovarian surface epithelia are multipotent and capable of serving as ovarian cancer initiating cells. BMC Med Genomics 2: 71.
Cheng W, Liu J, Yoshida H, Rosen D, Naora H . (2005). Lineage infidelity of epithelial ovarian cancer is control by HOX genes that specify regional identity in the reproductive tract. Nat Med 11: 531–537.
Cho KR . (2009). Ovarian cancer update: lessons from morphology, molecules and mice. Arch Pathol Lab Med 133: 1775–1781.
Cho KR, Shih I-M . (2009). Ovarian Cancer. Ann Rev Path 4: 287–313.
Clark-Knowles KV, Senterman MK, Collins O, Vanderhyden BC . (2009). Conditional inactivation of Brca1, p53 and Rb in mouse ovaries results in the development of leiomyosarcomas. PLoS One 4: e8534.
Connolly DC, Bao R, Nikitin Y, Stephens KC, Poole TW, Hua X et al. (2003). Female mice chimeric for expression of the Simian virus 40T Ag under the control of the MISRIIR promoter develop epithelial ovarian cancer. Cancer Res 63: 1389–1397.
Corney DC, Flesken-Nikitin A, Godwin AK, Wang W, Nikitin AY . (2007). MicroRNA-34b and microRNA-34c are targets of p53 and cooperate in control of cell proliferation and adhesion-independent growth. Cancer Res 67: 8433–8438.
Corney DC, Hwang C, Matoso A, Vogt M, Fleskin-Nikitin A, Godwin AK et al. (2010). Frequent downregulation of miR-34 family in human ovarian cancers. Clin Cancer Res 16: 1119–1128.
Costa BM, Smith JS, Chen Y, Chen J, Phillips HS, Aldape KD et al. (2010). Reversing HOXO9 oncogene activation by PI3K inhibition: epigenetic mechanism and prognostic significance in human glioblastoma. Cancer Res 70: 453–462.
Creighton CJ, Fountain MD, Yu Z, Nagaraja AK, Zhu H, Khan M et al. (2010). Molecular-profiling uncovers a p53-associated role for microRNA-31 in inhibiting the proliferation of serous ovarian carcinomas and other cancers. Cancer Res 70: 1906–1915.
Crum CP, Drapkin R, Kindelberger D, Medeiros F, Miran A, Lee Y . (2007). Lessons from BRCA: the tubal fimbria emerges as an origin for pelvic serous cancer. Clin Med Res 5: 35–44.
Dahlya N, Sherman-Baust CA, Wang T-L, Davidson B, Shih I-M, Zhang Y et al. (2008). MicroRNA expression and identification of putative miRNA targets in ovarian cancer. PLoS One 3: 1–11.
Despierre E, Lambrechts D, Neven P, Amant F, Lambrechts S, Vergote L . (2010). The molecular genetic basis of ovarian cancer and its roadmap towards a better treatment. Gynecol Oncol 117: 358–365.
Dinulescu DM, Ince TA, Quade BJ, Shafer SA, Crowley D, Jacks T . (2005). Role of K-ras and Pten in the development of mouse models of endometriosis and endometrioid ovarian cancer. Nat Med 11: 63–70.
Dubeau L . (2008). The cell origin of ovarian epithelial tumours. Lancet Oncol 9: 1191–1197.
Fan HY, Liu Z, Paquet M, Wang J, Lydon JP, DeMayo FJ et al. (2009a). Cell type specific targeted mutation of Kras and Pten document proliferation arrest in granulosa cells versus oncogenic insult in ovarian surface epithelial cells. Cancer Res 69: 6463–6472.
Fan HY, Liu Z, Shimada M, Sterneck E, Johnson PF, Hedrick SM et al. (2009b). MAPK3/1 (ERK1/2) in ovarian granulosa cells are essential for female fertility. Science 324: 938–941.
Fan HY, Shimada M, Liu Z, Cahill N, Noma N, Wu Y et al. (2008). Selective expression of KrasG12D in granulosa cells of the mouse ovary causes defects in follicular development and ovulation. Development 135: 2127–2137.
Flesken-Nikitin A, Choi K-C, Eng JP, Shmidt N, Nikitin AY . (2003). Induction of carcinogenesis by concurrent inactivation of p53 and Rb1 in the mouse ovarian surface epithelium. Cancer Res 63: 3459–3463.
Gava N, Clarke CL, Bye C, Byth K, deFazio A . (2008). Global gene expression profiles of ovarian surface epithelial cells in vivo. J Mol Endocrinol 40: 281–296.
GlaxoSmithKline GSK (2008). Cancer cell line genomic profiling data. https://cabig.nci.nih.gov/tools/caArray.
Gorringe KL, Campbell IG . (2009). Large-scale genomic analysis of ovarian carcinomas. Mol Oncol 3: 157–164.
Hermeking H . (2007). p53 Enters the microRNA world. Cancer Cell 12: 414–418.
Iorio MV, Visone R, Di Leva G, Donati V, Petrocca F, Casalini P et al. (2007). MicroRNA signatures in human ovarian cancer. Cancer Res 67: 8699–8707.
Jamin SP, Arango NA, Mishina Y, Hanks MC, Behringer RR . (2002). Requirement of Bmpr1a for Mullerian duct regression during male sexual development. Nat Genet 32: 408–410.
Ji Q, Hao X, Zhang M, Tang W, Meng Y, Li L et al. (2009). MicroRNA miR-34 inhibits human pancreatic cancer tumor-initiating cells. PLos One 4: 1–13.
Kaplinas K, Kessler CB, Delany AM . (2009). miR-29 suppression of osteonectin in osteoblasts: regulation during differentiation by canonical Wnt signaling. J Cell Biochem 108: 216–224.
Karst AM, Drapkin R . (2010). Ovarian cancer pathogenesis: a model in evolution. J Oncol 2010: 932371.
Kindelberger DW, Lee Y, Miron A, Hirsch MS, Feltmate C, Medeiros F et al. (2007). Intraepithelial carcinoma of the fimbria and pelvic serous adenocarcinoma: evidence for a causal relationship. Am J Surg Pathol 31: 161–169.
Ko SY, Lengyel E, Naora H . (2010). The Mullerian HOXA10 gene promotes growth of ovarian surface epithelial cells by stimulating epithelial-stroma interactions. Mol Cell Endocrinol 317: 112–119.
Kobel M, Kalloger SE, Boyd N, McKinney S, Mehl E, Palmer C et al. (2008). Ovarian carcinoma subtypes are different diseases: implications for biomarker studies. PLoS Med 5: 1749–1760.
Konstantinopoulos PA, Sppentzos D, Cannistra SA . (2008). Gene-expression profiling in epithelial ovarian cancer. Nat Clin Pract 5: 577–587.
Krichevsky AM, Gabriety G . (2009). miRNA-21: a small multi-faceted RNA. J Cell Mol Med 13: 39–53.
Kurman RJ, Shih I-M . (2010). The origin and pathogeneis of epithelial ovarian cancer: a proposed unifying theory. Am J Surg Pathol 34: 433–443.
Lague MN, Paquet M, Fan HY, Kaartinene MJ, Chu S, Jamin SP et al. (2008). Synergistic effects of Pten loss and WNT/CTNNB1 signaling pathway activation in granulosa cell tumor development and progression. Carcinogenesis 29: 2062 .
Lee JM, Dedhar S, Kalluri R, Thompson EW . (2006). The epithelial-mesenchymal transition: new insights in signaling, development and disease. J Cell Biol 172: 973–981.
Lee Y, Miron A, Drapkin R, Nucci MR, Medeiros F, Saleemuddin A et al. (2007). A candidate precursor to serous carcinoma that originates in the distal fallopian tube. J Pathol 211: 26–35.
Liang S, Yang N, Pan Y, Deng S, Lin X, Yang X et al. (2009). Expression of activated PIK3CA in ovarian surface epithelium results in hyperplasia but not tumor formation. PLoS One 4: e4295.
Malpica A, Deavers MT, Lu K, Bodurka DC, Atkinson EN, Gershenson DM et al. (2004). Grading ovarian serous carcinoma using a two-tier system. Am J Surg Pathol 28: 496–504.
Miller KA, Yeager N, Baker K, Liao X-H, Referoff S, De Cristofano A . (2009). Oncogenic Kras requires simultaneous PI3K signaling to induce ERK activation and transform thyroid epithelial cells in vivo. Cancer Res 69: 3689–3694.
Nam EJ, Yoon H, Kim SW, Kim H, Kim YT, Kim JH et al. (2008). MicroRNA expression profiles in serous ovarian carcinoma. Clin Cancer Res 14: 2690–2695.
Orsulic S, Li Y, Soslow RA, Vitale-Cross LA, Gutkind JS, Varmus HE . (2002). Induction of ovarian cancer by defined multiple genetic changes in a mouse model system. Cancer Cell 1: 53–62.
Page CL, Ouellet V, Madore J, Ren F, Hudson TJ, Tonin PN et al. (2006). Gene expression profiling of primary cultures of ovarian epithelial cell identifies novel molecular classifiers of ovarian cancer. Br J Cancer 94: 436–445.
Park SY, Lee JH, Nam JW, Kim VN . (2009). miR-29 miRNAs activate p53 by targeting p85 alpha and CDC42. Nat Struct Mol Biol 16: 23–29.
Raimondi AR, Molinolo A, Gutkind JS . (2009). Rapamycin prevents early onset of tumorigenesis inan oral-specific K-ras and p53 two-hit carcinogenesis model. Cancer Res 69: 4159–4166.
Roh MH, Yassin Y, Miron A, Mehra KK, Mehrad M, Monte NM et al. (2010). High-grade fimbrial-ovarian carcinomas are unified by altered p53, PTEN and PAX2 expression. Mod Pathol 23: 1316–1324.
Roush S, Slack FJ . (2008). The let-7 family of microRNAs. Trends Cell Biol 18: 505–516.
Rybak A, Fuchs H, Smirnova L, Brandt C, Pohl EE, Pohl EE et al. (2008). A feedback loop comprising lin-28 and let-7 controls pre-let-7 maturation during neural stem cell commitment. Nat Cell Biol 10: 987–993.
Saldanha AJ . (2004). Janva Treeview --extensible visualization of microarray data. Bioinformatics 20: 3246–3248.
Santin AD, Zhan F, Bellone S, Palmieri M, Cane S, Bignotti E et al. (2004). Gene expression profiles in primary ovarian serous papillary tumors and normal ovarian epithelium: identification of candidate molecular markers for ovarian cancer diagnosis and therapy. Int J Cancer 112: 14–25.
Schwartz DR, Kardia SL, Shedden KA, Kuick R, Michailidis G, Taylor JM et al. (2002). Gene expression in ovarian cancer reflects both morphology and biological behavior, distinguishing clear cell from other poor-prognosis ovarian carcinomas. Cancer Res 62: 4722–4729.
Stafani G, Slack FJ . (2008). Small non-coding RNAs in animal development. Nat Rev Mol Cell Biol 9: 219–230.
Wang D, DuBois RN . (2010). Eicosanoids and cancer. Nat Rev Cancer 10: 181–193.
Wang Y, Krivtsov AV, Sinha AU, North TE, Goessling W, Feng Z et al. (2010). The Wnt/b-catenin pathway is required for the development of leukemia stem cells in AML. Science 327: 1650–1653.
Wee S, Jagani Z, Xiang XX, Loo A, Dorsch M, Yao Y-M et al. (2009). PI3K pathway activation mediates resistance to MEK inhibitors in KRAS mutant cancers. Cancer Res 69: 4286–4293.
Wong KK, Lu KH, Malpica A, Bodurka DC, Shvartsman HS, Schmandt RE et al. (2007). Significantly greater expression of ER, PR, and ECAD in advanced-stage low-grade ovarian serous carcinoma as revealed by immunohistochemical analysis. Int J Gynecol Pathol 26: 404–409.
Wu R, Hendrix-Lucas N, Kiuck R, zhai Y, Schwartz DR, Akyol A et al. (2007). Mouse model of human ovarian endometrioid adenocarcinoma based on somatic defects in the Wnt/b-catenin and PI3K/Pten pathways. Cancer Cell 11: 321–333.
Wyman SK, Parkin RK, Mitchell PS, Fritz BR, O'Briant K, Godwin AK et al. (2009). Repertoire of microRNAs in epithelial ovarian cancer as determined by next generation sequencing of small RNA cDNA libraries. PLoS One 4: e5311.
Xing D, Scangas G, Nitta M, He L, Xu X, Ioffe YJM et al. (2009). A role for BRCA1 in uterine leiomyosarcoma. Cancer Res 69: 8231–8235.
Yang H, Kong W, He L, Zhao J-J, O'Donnell JD, Wang J et al. (2008). MicroRNA expression profiling in human ovarian cancer: miR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res 68: 425–433.
Zhang CC, Kaba M, Ge G, Tong W, Hug C, Lodish HF . (2006). Angiopoietin-like proteins stimulate ex vivo expansion of hemapoitetic stem cells. Nat Med 12: 240–245.
Zhao Z, Zuber J, Diaz-Flores E, Lintault L, Kogan SC, Shannon K et al. (2010). p53 loss promotes acute myeloid leukemia by enabling aberrant self-renewal. Genes Dev 24: 1389–1402.
Acknowledgements
We Alan J Herron DVM, Professor and Head of the Comparative Pathology Laboratory for his advice and insights into the mouse ovarian tumor phenotype, Yuet Lo and Azam Zariff for technical assistance and the Microscopy Core at Baylor College of Medicine for their expertise. We also thank the Immunohistochemistry Laboratory at The University of Texas MD Anderson Cancer Center for performing the calretinin and ESR1 immunostaining. Supported in part by NIH-HD-16229 (JSR), NRSA (LM), a Program Project Development Grant from the Ovarian Cancer Research Fund (CJC, PG, MA) and The University of Texas MD Anderson Cancer Center Specialized Program of Research Excellence in Ovarian Cancer (P50 CA08369) (KKW).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Mullany, L., Fan, HY., Liu, Z. et al. Molecular and functional characteristics of ovarian surface epithelial cells transformed by KrasG12D and loss of Pten in a mouse model in vivo. Oncogene 30, 3522–3536 (2011). https://doi.org/10.1038/onc.2011.70
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.70
Keywords
This article is cited by
-
MiR-34b/c play a role in early sex differentiation of Amur sturgeon, Acipenser schrenckii
Frontiers in Zoology (2022)
-
Identification of ovarian high-grade serous carcinoma cell lines that show estrogen-sensitive growth as xenografts in immunocompromised mice
Scientific Reports (2020)
-
Dysregulation of mitotic machinery genes precedes genome instability during spontaneous pre-malignant transformation of mouse ovarian surface epithelial cells
BMC Genomics (2016)
-
Ovary and fimbrial stem cells: biology, niche and cancer origins
Nature Reviews Molecular Cell Biology (2015)
-
Immunobiology of human mucin 1 in a preclinical ovarian tumor model
Oncogene (2013)