Abstract
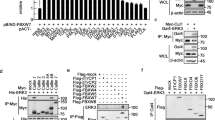
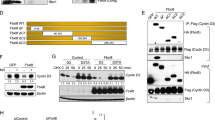
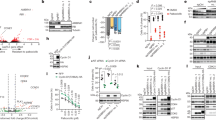
Dysregulated behavior of cell cycle proteins and their control by ubiquitin E3 ligases is an emerging theme in human lung cancer. Here, we identified and characterized the activity of a novel F-box protein, termed FBXL2, belonging to the SCF (Skip-Cullin1-F-box protein) E3 ligase family. Ectopically expressed FBXL2 triggered G2/M-phase arrest, induced chromosomal anomalies and increased apoptosis of transformed lung epithelia by mediating polyubiquitination and degradation of the mitotic regulator, cyclin D3. Unlike other F-box proteins that target phosphodegrons within substrates, FBXL2 uniquely recognizes a canonical calmodulin (CaM)-binding motif within cyclin D3 to facilitate its polyubiquitination. CaM bound and protected cyclin D3 from FBXL2 by direct intermolecular competition with the F-box protein for access within this motif. The chemotherapeutic agent vinorelbine increased apoptosis of human lung carcinoma cells by inducing FBXL2 expression and cyclin D3 degradation, an effect accentuated by CaM knockdown. Depletion of endogenous FBXL2 stabilized cyclin D3 levels, accelerated cancer cell growth and increased cell viability after vinorelbine treatment. Last, ectopic expression of FBXL2 significantly inhibited the growth and migration of tumorogenic cells and tumor formation in athymic nude mice. These observations implicate SCFFBXL2 as an indispensible regulator of mitosis that serves as a tumor suppressor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Agassandian M, Chen BB, Schuster CC, Houtman JC, Mallampalli RK . (2010). 14-3-3zeta escorts CCTalpha for calcium-activated nuclear import in lung epithelia. FASEB J 24: 1271–1283.
Brown EM, Shoback DM . (1984). The relationship between PTH secretion and cytosolic calcium concentration in bovine parathyroid cells. Prog Clin Biol Res 168: 139–144.
Butler PL, Mallampalli RK . (2010). Cross-talk between remodeling and de novo pathways maintains phospholipid balance through ubiquitination. J Biol Chem 285: 6246–6258.
Cardozo T, Pagano M . (2004). The SCF ubiquitin ligase: insights into a molecular machine. Nat Rev Mol Cell Biol 5: 739–751.
Cenciarelli C, Chiaur DS, Guardavaccaro D, Parks W, Vidal M, Pagano M . (1999). Identification of a family of human F-box proteins. Curr Biol 9: 1177–1179.
Chen BB, Coon TA, Glasser JR, Mallampalli RK . (2011). Calmodulin antagonizes a calcium-activated SCF ubiquitin E3 ligase subunit, FBXL2, to regulate surfactant homeostasis. Mol Cell Biol 22: 22.
Chen BB, Mallampalli RK . (2007). Calmodulin binds and stabilizes the regulatory enzyme, CTP: phosphocholine cytidylyltransferase. J Biol Chem 282: 33494–33506.
Chen BB, Mallampalli RK . (2009). Masking of a nuclear signal motif by monoubiquitination leads to mislocalization and degradation of the regulatory enzyme cytidylyltransferase. Mol Cell Biol 29: 3062–3075.
Choi J, Chiang A, Taulier N, Gros R, Pirani A, Husain M . (2006). A calmodulin-binding site on cyclin E mediates Ca2+-sensitive G1/s transitions in vascular smooth muscle cells. Circ Res 98: 1273–1281.
Choi J, Husain M . (2006). Calmodulin-mediated cell cycle regulation: new mechanisms for old observations. Cell Cycle 5: 2183–2186.
Diehl JA, Zindy F, Sherr CJ . (1997). Inhibition of cyclin D1 phosphorylation on threonine-286 prevents its rapid degradation via the ubiquitin-proteasome pathway. Genes Dev 11: 957–972.
Duan Y, He X, Yang H, Ji Y, Tao T, Chen J et al. (2010). Cyclin D3/CDK11(p58) complex involved in Schwann cells proliferation repression caused by lipopolysaccharide. Inflammation 33: 189–199.
Fang MZ, Lee MH, Lee YS, Kim YC, Lee BM, Cho MH . (2002). Low expression of cyclin D2 in G2/M-arrested and transformed proliferating Balb/3T3 cells. J Vet Med Sci 64: 201–205.
Gautschi O, Ratschiller D, Gugger M, Betticher DC, Heighway J . (2007). Cyclin D1 in non-small cell lung cancer: a key driver of malignant transformation. Lung Cancer 55: 1–14.
Glover DM, Leibowitz MH, McLean DA, Parry H . (1995). Mutations in aurora prevent centrosome separation leading to the formation of monopolar spindles. Cell 81: 95–105.
Hansen DV, Loktev AV, Ban KH, Jackson PK . (2004). Plk1 regulates activation of the anaphase promoting complex by phosphorylating and triggering SCFbetaTrCP-dependent destruction of the APC Inhibitor Emi1. Mol Biol Cell 15: 5623–5634.
Hochstrasser M . (2000). Biochemistry. All in the ubiquitin family. Science 289: 563–564.
Hu D, Valentine M, Kidd VJ, Lahti JM . (2007). CDK11(p58) is required for the maintenance of sister chromatid cohesion. J Cell Sci 120: 2424–2434.
Ilyin GP, Rialland M, Glaise D, Guguen-Guillouzo C . (1999). Identification of a novel Skp2-like mammalian protein containing F-box and leucine-rich repeats. FEBS Lett 459: 75–79.
Kahl CR, Means AR . (2003). Regulation of cell cycle progression by calcium/calmodulin-dependent pathways. Endocr Rev 24: 719–736.
Korkotian E, Segal M . (1996). Lasting effects of glutamate on nuclear calcium concentration in cultured rat hippocampal neurons: regulation by calcium stores. J Physiol 496 (Part 1): 39–48.
Lew DJ, Dulic V, Reed SI . (1991). Isolation of three novel human cyclins by rescue of G1 cyclin (Cln) function in yeast. Cell 66: 1197–1206.
Lin DI, Barbash O, Kumar KG, Weber JD, Harper JW, Klein-Szanto AJ et al. (2006). Phosphorylation-dependent ubiquitination of cyclin D1 by the SCF(FBX4-alphaB crystallin) complex. Mol Cell 24: 355–366.
Liu C, Kato Y, Zhang Z, Do VM, Yankner BA, He X . (1999). Beta-Trcp couples beta-catenin phosphorylation-degradation and regulates Xenopus axis formation. Proc Natl Acad Sci USA 96: 6273–6278.
Lu KP, Rasmussen CD, May GS, Means AR . (1992). Cooperative regulation of cell proliferation by calcium and calmodulin in Aspergillus nidulans. Mol Endocrinol 6: 365–374.
Malard V, Berenguer F, Prat O, Ruat S, Steinmetz G, Quemeneur E . (2007). Global gene expression profiling in human lung cells exposed to cobalt. BMC Genomics 8: 147.
Mallampalli RK, Ryan AJ, Salome RG, Jackowski S . (2000). Tumor necrosis factor-alpha inhibits expression of CTP:phosphocholine cytidylyltransferase. J Biol Chem 275: 9699–9708.
Myung SJ, Rerko RM, Yan M, Platzer P, Guda K, Dotson A et al. (2006). 15-Hydroxyprostaglandin dehydrogenase is an in vivo suppressor of colon tumorigenesis. Proc Natl Acad Sci USA 103: 12098–12102.
Ohtani K, DeGregori J, Nevins JR . (1995). Regulation of the cyclin E gene by transcription factor E2F1. Proc Natl Acad Sci USA 92: 12146–12150.
Okabe H, Lee SH, Phuchareon J, Albertson DG, McCormick F, Tetsu O . (2006). A critical role for FBXW8 and MAPK in cyclin D1 degradation and cancer cell proliferation. PLoS One 1: e128.
Papay J, Krenacs T, Moldvay J, Stelkovics E, Furak J, Molnar B et al. (2007). Immunophenotypic profiling of nonsmall cell lung cancer progression using the tissue microarray approach. Appl Immunohistochem Mol Morphol 15: 19–30.
Peeper DS, Upton TM, Ladha MH, Neuman E, Zalvide J, Bernards R et al. (1997). Ras signalling linked to the cell-cycle machinery by the retinoblastoma protein. Nature 386: 177–181.
Petretti C, Savoian M, Montembault E, Glover DM, Prigent C, Giet R . (2006). The PITSLRE/CDK11p58 protein kinase promotes centrosome maturation and bipolar spindle formation. EMBO Rep 7: 418–424.
Pszczolkowski MA, Lee WS, Liu HP, Chiang AS . (1999). Glutamate-induced rise in cytosolic calcium concentration stimulates in vitro rates of juvenile hormone biosynthesis in corpus allatum of Diploptera punctata. Mol Cell Endocrinol 158: 163–171.
Ray NB, Durairaj L, Chen BB, McVerry BJ, Ryan AJ, Donahoe M et al. (2010). Dynamic regulation of cardiolipin by the lipid pump Atp8b1 determines the severity of lung injury in experimental pneumonia. Nat Med 16: 1120–1127.
Rhoads AR, Friedberg F . (1997). Sequence motifs for calmodulin recognition. FASEB J 11: 331–340.
Ridsdale R, Tseu I, Wang J, Post M . (2001). CTP:phosphocholine cytidylyltransferase alpha is a cytosolic protein in pulmonary epithelial cells and tissues. J Biol Chem 276: 49148–49155.
Rosario CO, Ko MA, Haffani YZ, Gladdy RA, Paderova J, Pollett A et al. (2010). Plk4 is required for cytokinesis and maintenance of chromosomal stability. Proc Natl Acad Sci USA 107: 6888–6893.
Santra MK, Wajapeyee N, Green MR . (2009). F-box protein FBXO31 mediates cyclin D1 degradation to induce G1 arrest after DNA damage. Nature 459: 722–725.
Shan J, Zhao W, Gu W . (2009). Suppression of cancer cell growth by promoting cyclin D1 degradation. Mol Cell 36: 469–476.
Sicinska E, Aifantis I, Le Cam L, Swat W, Borowski C, Yu Q et al. (2003). Requirement for cyclin D3 in lymphocyte development and T cell leukemias. Cancer Cell 4: 451–461.
Skaar JR, Pagan JK, Pagano M . (2009). SnapShot: F box proteins I. Cell 137: 1160–1160 e1161.
Sterlacci W, Fiegl M, Hilbe W, Jamnig H, Oberaigner W, Schmid T et al. (2010). Deregulation of p27 and cyclin D1/D3 control over mitosis is associated with unfavorable prognosis in non-small cell lung cancer, as determined in 405 operated patients. J Thorac Oncol 5: 1325–1336.
Tanaka Y, Tanaka N, Saeki Y, Tanaka K, Murakami M, Hirano T et al. (2008). c-Cbl-dependent monoubiquitination and lysosomal degradation of gp130. Mol Cell Biol 28: 4805–4818.
Tanguay DA, Colarusso TP, Doughty C, Pavlovic-Ewers S, Rothstein TL, Chiles TC . (2001). Cutting edge: differential signaling requirements for activation of assembled cyclin D3-cdk4 complexes in B-1 and B-2 lymphocyte subsets. J Immunol 166: 4273–4277.
Taules M, Rius E, Talaya D, Lopez-Girona A, Bachs O, Agell N . (1998). Calmodulin is essential for cyclin-dependent kinase 4 (Cdk4) activity and nuclear accumulation of cyclin D1-Cdk4 during G1. J Biol Chem 273: 33279–33286.
Tsang WY, Spektor A, Luciano DJ, Indjeian VB, Chen Z, Salisbury JL et al. (2006). CP110 cooperates with two calcium-binding proteins to regulate cytokinesis and genome stability. Mol Biol Cell 17: 3423–3434.
Tyers M, Willems AR . (1999). One ring to rule a superfamily of E3 ubiquitin ligases. Science 284: 601, 603–604.
Wang C, Gale Jr M, Keller BC, Huang H, Brown MS, Goldstein JL et al. (2005). Identification of FBL2 as a geranylgeranylated cellular protein required for hepatitis C virus RNA replication. Mol Cell 18: 425–434.
Watanabe N, Arai H, Iwasaki J, Shiina M, Ogata K, Hunter T et al. (2005). Cyclin-dependent kinase (CDK) phosphorylation destabilizes somatic Wee1 via multiple pathways. Proc Natl Acad Sci USA 102: 11663–11668.
Weiderpass E . (2010). Lifestyle and cancer risk. J Prev Med Public Health 43: 459–471.
Wikenheiser KA, Clark JC, Linnoila RI, Stahlman MT, Whitsett JA . (1992). Simian virus 40 large T antigen directed by transcriptional elements of the human surfactant protein C gene produces pulmonary adenocarcinomas in transgenic mice. Cancer Res 52: 5342–5352.
Zhang Q, Tian L, Mansouri A, Korapati AL, Johnson TJ, Claret FX . (2005). Inducible expression of a degradation-resistant form of p27Kip1 causes growth arrest and apoptosis in breast cancer cells. FEBS Lett 579: 3932–3940.
Zhang S, Cai M, Xu S, Chen S, Chen X, Chen C et al. (2002). Interaction of p58(PITSLRE), a G2/M-specific protein kinase, with cyclin D3. J Biol Chem 277: 35314–35322.
Zheng N, Schulman BA, Song L, Miller JJ, Jeffrey PD, Wang P et al. (2002). Structure of the Cul1-Rbx1-Skp1-F boxSkp2 SCF ubiquitin ligase complex. Nature 416: 703–709.
Zi X, Agarwal R . (1999). Silibinin decreases prostate-specific antigen with cell growth inhibition via G1 arrest, leading to differentiation of prostate carcinoma cells: implications for prostate cancer intervention. Proc Natl Acad Sci USA 96: 7490–7495.
Acknowledgements
We thank AF Stewart for critical review of the manuscript and helpful suggestions. This material is based upon work supported, in part, by the US Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development. This work was supported by a Merit Review Award from the US Department of Veterans Affairs and National Institutes of Health R01 grants HL096376, HL097376 and HL098174 (to RKM). The contents presented do not represent the views of the Department of Veterans Affairs or the United States Government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website )
Supplementary information
Rights and permissions
About this article
Cite this article
Chen, B., Glasser, J., Coon, T. et al. F-box protein FBXL2 exerts human lung tumor suppressor-like activity by ubiquitin-mediated degradation of cyclin D3 resulting in cell cycle arrest. Oncogene 31, 2566–2579 (2012). https://doi.org/10.1038/onc.2011.432
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.432
Keywords
This article is cited by
-
FBXL2 promotes E47 protein instability to inhibit breast cancer stemness and paclitaxel resistance
Oncogene (2023)
-
Inhibition of USP10 induces myeloma cell apoptosis by promoting cyclin D3 degradation
Acta Pharmacologica Sinica (2023)
-
Fbxl8 suppresses lymphoma growth and hematopoietic transformation through degradation of cyclin D3
Oncogene (2021)
-
FBXL2 counteracts Grp94 to destabilize EGFR and inhibit EGFR-driven NSCLC growth
Nature Communications (2021)
-
EZH2 oncogenic mutations drive epigenetic, transcriptional, and structural changes within chromatin domains
Nature Genetics (2019)