Abstract
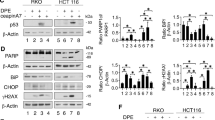
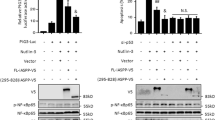
Stress response gene ATF3 is one of the p53 target genes and has a tumor suppressor role in cancer. However, the biological role of p53–ATF3 pathway is not well understood. Death receptor 5 (DR5) is a death domain-containing transmembrane receptor that triggers cell death upon binding to its ligand TRAIL (tumor necrosis factor-related apoptosis-inducing ligand), and a combination of TRAIL and agents that increase the expression of DR5 is expected as a novel anticancer therapy. In this report, we demonstrate that ATF3 is required for efficient DR5 induction upon DNA damage by camptothecin (CPT) in colorectal cancer cells. In the absence of ATF3, induction of DR5 messenger RNA and protein is remarkably abrogated, and this is associated with reduced cell death by TRAIL and CPT. By contrast, exogenous expression of ATF3 causes more rapid and elevated expression of DR5, resulting in enhanced sensitivity to apoptotic cell death by TRAIL/CPT. Reporter assay and DNA affinity precipitation assay demonstrate that at least three ATF/CRE motifs at the proximal promoter of the human DR5 gene are involved in the activation of DNA damage-induced DR5 gene transcription. Furthermore, ATF3 is shown to interact with p53 to form a complex on the DR5 gene by Re-chromatin immunoprecipitation assay. Taken together, our results provide a novel insight into the role of ATF3 as an essential co-transcription factor for p53 upon DNA damage, and this may represent a useful biomarker for TRAIL-based anticancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Amundson SA, Bittner M, Chen Y, Trent J, Meltzer P, Fornace Jr AJ . (1999). Fluorescent cDNA microarray hybridization reveals complexity and heterogeneity of cellular genotoxic stress responses. Oncogene 18: 3666–3672.
Ashkenazi A, Herbst RS . (2008). To kill a tumor cell: the potential of proapoptotic receptor agonists. J Clin Invest 118: 1979–1990.
Cai Y, Zhang C, Nawa T, Aso T, Tanaka M, Oshiro S et al. (2000). Homocysteine-responsive ATF3 gene expression in human vascular endothelial cells: activation of c-Jun NH(2)-terminal kinase and promoter response element. Blood 96: 2140–2148.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery Jr CA, Butel JS et al. (1992). Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356: 215–221.
Eskes R, Desagher S, Antonsson B, Martinou JC . (2000). Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol Cell Biol 20: 929–935.
Fan F, Jin S, Amundson SA, Tong T, Fan W, Zhao H et al. (2002). ATF3 induction following DNA damage is regulated by distinct signaling pathways and over-expression of ATF3 protein suppresses cells growth. Oncogene 21: 7488–7496.
Farczadi E, Szanto J, Kaszas I, Benyo I, Bodnar Z, Szlobodnyik J et al. (1999). Changes in apoptotic and mitotic activity in rectal carcinoma after short-term cytostatic therapy as possible predictive factors. Neoplasma 46: 219–223.
Fuchs C, Mitchell EP, Hoff PM . (2006). Irinotecan in the treatment of colorectal cancer. Cancer Treat Rev 32: 491–503.
Gliniak B, Le T . (1999). Tumor necrosis factor-related apoptosis-inducing ligand's antitumor activity in vivo is enhanced by the chemotherapeutic agent CPT-11. Cancer Res 59: 6153–6158.
Green DR, Evan GI . (2002). A matter of life and death. Cancer Cell 1: 19–30.
Hackl C, Lang SA, Moser C, Mori A, Fichtner-Feigl S, Hellerbrand C et al. (2010). Activating transcription factor-3 (ATF3) functions as a tumor suppressor in colon cancer and is up-regulated upon heat-shock protein 90 (HSP90) inhibition. BMC Cancer 10: 668.
Hai T, Wolfgang CD, Marsee DK, Allen AE, Sivaprasad U . (1999). ATF3 and stress responses. Gene Expr 7: 321–335.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Hsu JC, Laz T, Mohn KL, Taub R . (1991). Identification of LRF-1, a leucine-zipper protein that is rapidly and highly induced in regenerating liver. Proc Natl Acad Sci USA 88: 3511–3515.
Janz M, Hummel M, Truss M, Wollert-Wulf B, Mathas S, Johrens K et al. (2006). Classical Hodgkin lymphoma is characterized by high constitutive expression of activating transcription factor 3 (ATF3), which promotes viability of Hodgkin/Reed-Sternberg cells. Blood 107: 2536–2539.
Kawauchi J, Zhang C, Nobori K, Hashimoto Y, Adachi MT, Noda A et al. (2002). Transcriptional repressor activating transcription factor 3 protects human umbilical vein endothelial cells from tumor necrosis factor-alpha-induced apoptosis through down-regulation of p53 transcription. J Biol Chem 277: 39025–39034.
Kischkel FC, Lawrence DA, Chuntharapai A, Schow P, Kim KJ, Ashkenazi A . (2000). Apo2L/TRAIL-dependent recruitment of endogenous FADD and caspase-8 to death receptors 4 and 5. Immunity 12: 611–620.
LeBlanc H, Lawrence D, Varfolomeev E, Totpal K, Morlan J, Schow P et al. (2002). Tumor-cell resistance to death receptor-induced apoptosis through mutational inactivation of the proapoptotic Bcl-2 homolog Bax. Nat Med 8: 274–281.
Legarza K, Yang LX . (2006). New molecular mechanisms of action of camptothecin-type drugs. Anticancer Res 26: 3301–3305.
Lu D, Wolfgang CD, Hai T . (2006). Activating transcription factor 3, a stress-inducible gene, suppresses Ras-stimulated tumorigenesis. J Biol Chem 281: 10473–10481.
Miyazaki K, Inoue S, Yamada K, Watanabe M, Liu Q, Watanabe T et al. (2009). Differential usage of alternate promoters of the human stress response gene ATF3 in stress response and cancer cells. Nucleic Acids Res 37: 1438–1451.
Morita S, Kojima T, Kitamura T . (2000). Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther 7: 1063–1066.
Naka T, Sugamura K, Hylander BL, Widmer MB, Rustum YM, Repasky EA . (2002). Effects of tumor necrosis factor-related apoptosis-inducing ligand alone and in combination with chemotherapeutic agents on patients’ colon tumors grown in SCID mice. Cancer Res 62: 5800–5806.
Nesterov A, Nikrad M, Johnson T, Kraft AS . (2004). Oncogenic Ras sensitizes normal human cells to tumor necrosis factor-alpha-related apoptosis-inducing ligand-induced apoptosis. Cancer Res 64: 3922–3927.
Nicholson DW . (2000). From bench to clinic with apoptosis-based therapeutic agents. Nature 407: 810–816.
Nobori K, Ito H, Tamamori-Adachi M, Adachi S, Ono Y, Kawauchi J et al. (2002). ATF3 inhibits doxorubicin-induced apoptosis in cardiac myocytes: a novel cardioprotective role of ATF3. J Mol Cell Cardiol 34: 1387–1397.
Pan J, McEver RP . (1993). Characterization of the promoter for the human P-selectin gene. J Biol Chem 268: 22600–22608.
Pelzer AE, Bektic J, Haag P, Berger AP, Pycha A, Schafer G et al. (2006). The expression of transcription factor activating transcription factor 3 in the human prostate and its regulation by androgen in prostate cancer. J Urol 175: 1517–1522.
Pitti RM, Marsters SA, Ruppert S, Donahue CJ, Moore A, Ashkenazi A . (1996). Induction of apoptosis by Apo-2 ligand, a new member of the tumor necrosis factor cytokine family. J Biol Chem 271: 12687–12690.
Ravasi T, Suzuki H, Cannistraci CV, Katayama S, Bajic VB, Tan K et al. (2010). An atlas of combinatorial transcriptional regulation in mouse and man. Cell 140: 744–752.
Riley T, Sontag E, Chen P, Levine A . (2008). Transcriptional control of human p53-regulated genes. Nat Rev Mol Cell Biol 9: 402–412.
Scott N, Hale A, Deakin M, Hand P, Adab FA, Hall C et al. (1998). A histopathological assessment of the response of rectal adenocarcinoma to combination chemo-radiotherapy: relationship to apoptotic activity, p53 and bcl-2 expression. Eur J Surg Oncol 24: 169–173.
Sellers WR, Fisher DE . (1999). Apoptosis and cancer drug targeting. J Clin Invest 104: 1655–1661.
Sheikh MS, Burns TF, Huang Y, Wu GS, Amundson S, Brooks KS et al. (1998). p53-dependent and -independent regulation of the death receptor KILLER/DR5 gene expression in response to genotoxic stress and tumor necrosis factor alpha. Cancer Res 58: 1593–1598.
Sprick MR, Weigand MA, Rieser E, Rauch CT, Juo P, Blenis J et al. (2000). FADD/MORT1 and caspase-8 are recruited to TRAIL receptors 1 and 2 and are essential for apoptosis mediated by TRAIL receptor 2. Immunity 12: 599–609.
Sussman RT, Ricci MS, Hart LS, Sun SY, El-Deiry WS . (2007). Chemotherapy-resistant side-population of colon cancer cells has a higher sensitivity to TRAIL than the non-SP, a higher expression of c-Myc and TRAIL-receptor DR4. Cancer Biol Ther 6: 1490–1495.
Suzuki T, Fujisawa JI, Toita M, Yoshida M . (1993). The trans-activator tax of human T-cell leukemia virus type 1 (HTLV-1) interacts with cAMP-responsive element (CRE) binding and CRE modulator proteins that bind to the 21-base-pair enhancer of HTLV-1. Proc Natl Acad Sci USA 90: 610–614.
Takimoto R, El-Deiry WS . (2000). Wild-type p53 transactivates the KILLER/DR5 gene through an intronic sequence-specific DNA-binding site. Oncogene 19: 1735–1743.
Tamura K, Hua B, Adachi S, Guney I, Kawauchi J, Morioka M et al. (2005). Stress response gene ATF3 is a target of c-myc in serum-induced cell proliferation. Embo J 24: 2590–2601.
Turchi L, Aberdam E, Mazure N, Pouyssegur J, Deckert M, Kitajima S et al. (2008). Hif-2alpha mediates UV-induced apoptosis through a novel ATF3-dependent death pathway. Cell Death Differ 15: 1472–1480.
Wang J, Chun HJ, Wong W, Spencer DM, Lenardo MJ . (2001). Caspase-10 is an initiator caspase in death receptor signaling. Proc Natl Acad Sci USA 98: 13884–13888.
Wang S, El-Deiry WS . (2003). Requirement of p53 targets in chemosensitization of colonic carcinoma to death ligand therapy. Proc Natl Acad Sci USA 100: 15095–15100.
Wang Y, Engels IH, Knee DA, Nasoff M, Deveraux QL, Quon KC . (2004). Synthetic lethal targeting of MYC by activation of the DR5 death receptor pathway. Cancer Cell 5: 501–512.
Wei CL, Wu Q, Vega VB, Chiu KP, Ng P, Zhang T et al. (2006). A global map of p53 transcription-factor binding sites in the human genome. Cell 124: 207–219.
Wei MC, Lindsten T, Mootha VK, Weiler S, Gross A, Ashiya M et al. (2000). tBID, a membrane-targeted death ligand, oligomerizes BAK to release cytochrome c. Genes Dev 14: 2060–2071.
Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, Nicholl JK et al. (1995). Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3: 673–682.
Wu GS, Burns TF, McDonald III ER, Jiang W, Meng R, Krantz ID et al. (1997). KILLER/DR5 is a DNA damage-inducible p53-regulated death receptor gene. Nat Genet 17: 141–143.
Wu X, Nguyen BC, Dziunycz P, Chang S, Brooks Y, Lefort K et al. (2010). Opposing roles for calcineurin and ATF3 in squamous skin cancer. Nature 465: 368–372.
Xiang H, Fox JA, Totpal K, Aikawa M, Dupree K, Sinicropi D et al. (2002). Enhanced tumor killing by Apo2L/TRAIL and CPT-11 co-treatment is associated with p21 cleavage and differential regulation of Apo2L/TRAIL ligand and its receptors. Oncogene 21: 3611–3619.
Yamaguchi H, Wang HG . (2004). CHOP is involved in endoplasmic reticulum stress-induced apoptosis by enhancing DR5 expression in human carcinoma cells. J Biol Chem 279: 45495–45502.
Yan C, Lu D, Hai T, Boyd DD . (2005). Activating transcription factor 3, a stress sensor, activates p53 by blocking its ubiquitination. Embo J 24: 2425–2435.
Yin X, Dewille JW, Hai T . (2008). A potential dichotomous role of ATF3, an adaptive-response gene, in cancer development. Oncogene 27: 2118–2127.
Yoon K, Lee SO, Cho SD, Kim K, Khan S, Safe S . (2011). Activation of nuclear TR3 (Nr4a1) by a diindolylmethane analog induces apoptosis and proapoptotic genes in pancreatic cancer cells and tumors. Carcinogenesis 32: 836–842.
Yoshida T, Maeda A, Tani N, Sakai T . (2001). Promoter structure and transcription initiation sites of the human death receptor 5/TRAIL-R2 gene. FEBS Lett 507: 381–385.
Zhang C, Gao C, Kawauchi J, Hashimoto Y, Tsuchida N, Kitajima S . (2002). Transcriptional activation of the human stress-inducible transcriptional repressor ATF3 gene promoter by p53. Biochem Biophys Res Commun 297: 1302–1310.
Acknowledgements
We thank Dr WS El-Deiry for a kind gift of valuable plasmid pGL2-Full. This work was supported in part by a grant-in-aid for Research on Priority Areas from the Ministry of Education, Culture, Sports, Science, and Technology of Japan [18012015, 18055008, 21590302] to SK and [22300330] to JM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Taketani, K., Kawauchi, J., Tanaka-Okamoto, M. et al. Key role of ATF3 in p53-dependent DR5 induction upon DNA damage of human colon cancer cells. Oncogene 31, 2210–2221 (2012). https://doi.org/10.1038/onc.2011.397
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.397
Keywords
This article is cited by
-
The transcription factor ATF3 switches cell death from apoptosis to necroptosis in hepatic steatosis in male mice
Nature Communications (2023)
-
Activating transcription factor 3 mediates apoptotic functions through a p53-independent pathway in human papillomavirus 18 infected HeLa cells
Virus Genes (2022)
-
Inhibitory role of ATF3 in gastric cancer progression through regulating cell EMT and stemness
Cancer Cell International (2021)
-
YIPF2 promotes chemotherapeutic agent-mediated apoptosis via enhancing TNFRSF10B recycling to plasma membrane in non-small cell lung cancer cells
Cell Death & Disease (2020)
-
Adenylate kinase 4 promotes bladder cancer cell proliferation and invasion
Clinical and Experimental Medicine (2019)