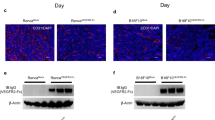
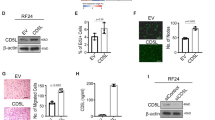
Abstract
Angiogenesis inhibiting agents are currently integral component of anticancer therapy. However, tumors, initially responsive to anti-angiogenic drugs or vascular targeting agents, can acquire resistance. The limited clinical efficacy might result from the heterogeneous nature of tumors or alternatively from the unique phenotype of tumor vascular cells, widely diverse from so-called ‘normal’ endothelium. Hence, defining the molecular mechanisms driving this diversity might provide a rational basis to design combinatory therapies that should be more effective in avoiding resistance. Herein, we demonstrated that tumor-derived endothelial cells (TECs) isolated from breast and kidney carcinomas retained an endothelial phenotype, but outspread independently of growth factors. Applying small interfering RNA approach, we demonstrated that interleukin (IL)-3, but not vascular endothelial growth factor, released by TECs, supports their autocrine growth and promotes in vivo vessel formation and tumor angiogenesis. Meanwhile, we found that the expression of the membrane-bound kit ligand (mbKitL) depends on IL-3, and it is crucial for adhesion of endothelial progenitor cells (EPCs) and inflammatory cells to TECs. These events required Akt activation. Finally, the finding that depletion of the mbKitL prevented EPC and inflammatory cell trafficking into vascular microenvironment, indicates that, as in bone marrow, the mbKitL can act as a membrane/adhesion molecule for c-Kit-expressing cells. These data provide evidences that an IL-3 autocrine loop can drive a tumor endothelial switch and that targeting IL-3 might confer a significant therapeutic advantage to hamper tumor angiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Avanzi GC, Brizzi MF, Giannotti J, Ciarletta A, Yang YC, Pegoraro L et al. (1990). M-07e human leukemic factor-dependent cell line provides a rapid and sensitive bioassay for the human cytokines GM-CSF and IL-3. J Cell Physiol 145: 458–464.
Baluk P, Morikawa S, Haskell A, Mancuso M, McDonald DM . (2003). Abnormalities of basement membrane on blood vessels and endothelial sprouts in tumors. Am J Pathol 163: 1801–1815.
Biancone L, Cantaluppi V, Duò D, Deregibus MC, Torre C, Camussi G . (2004). Role of L-selectin in the vascular homing of peripheral blood-derived endothelial progenitor cells. J Immunol 173: 5268–5274.
Brizzi MF, Formato L, Dentelli P, Rosso A, Pavan M, Garbarino G et al. (2001). Interleukin-3 stimulates migration and proliferation of vascular smooth muscle cells: a potential role in atherogenesis. Circulation 103: 549–554.
Brizzi MF, Garbarino G, Rossi PR, Pagliardi GL, Arduino C, Avanzi GC et al. (1993). Interleukin 3 stimulates proliferation and triggers endothelial-leukocyte adhesion molecule 1 gene activation of human endothelial cells. J Clin Invest 91: 2887–2892.
Bussolati B, Assenzio B, Deregibus MC, Camussi G . (2006). The proangiogenic phenotype of human tumor-derived endothelial cells depends on thrombospondin-1 downregulation via phosphatidylinositol 3-kinase/Akt pathway. J Mol Med 84: 852–863.
Bussolati B, Deambrosis I, Russo S, Deregibus MC, Camussi G . (2003). Altered angiogenesis and survival in human tumor-derived endothelial cells. FASEB J 17: 1159–1161.
Bussolati B, Deregibus MC, Camussi G . (2010). Characterization of molecular and functional alterations of tumor endothelial cells to design anti-angiogenic strategies. Curr Vasc Pharmacol 8: 220–232.
Butler JM, Kobayashi H, Rafii S . (2010). Instructive role of the vascular niche in promoting tumour growth and tissue repair by angiocrine factors. Nat Rev Cancer 10: 138–146.
De Palma M, Naldini L . (2006). Role of haematopoietic cells and endothelial progenitors in tumour angiogenesis. Biochim Biophys Acta 1766: 159–166.
Defilippi P, Rosso A, Dentelli P, Calvi C, Garbarino G, Tarone G et al. (2005). /{beta/}1 Integrin and IL-3R coordinately regulate STAT5 activation and anchora. J Cell Biol 168: 1099–1108.
Dentelli P, Del Sorbo L, Rosso A, Molinar A, Garbarino G, Camussi G et al. (1999). Human IL-3 stimulates endothelial cell motility and promotes in vivo new vessel formation. J Immunol 163: 2151–2159.
Dentelli P, Rosso A, Balsamo A, Colmenares Benedetto S, Zeoli A, Pegoraro M et al. (2007). C-KIT, by interacting with the membrane-bound ligand, recruits endothelial progenitor cells to inflamed endothelium. Blood 109: 4264–4271.
Dentelli P, Rosso A, Calvi C, Ghiringhello B, Garbarino G, Camussi G et al. (2004). IL-3 affects endothelial cell-mediated smooth muscle cell recruitment by increasing TGF beta activity: potential role in tumor vessel stabilization. Oncogene 23: 1681–1692.
Flanagan JG, Chan DC, Leder P . (1991). Transmembrane form of the kit ligand growth factor is determined by alternative splicing and is missing in the Sld mutant. Cell 4: 1025–1035.
Ferrara N, Kerbel RS . (2005). Angiogenesis as a therapeutic target. Nature 438: 967–974.
Folkman J . (2007). Angiogenesis: an organizing principle for drug discovery? Nat Rev Drug Discov 6: 273–286.
Gao D, Nolan D, McDonnell K, Vahdat L, Benezra R, Altorki N et al. (2009). Bone marrow-derived endothelial progenitor cells contribute to the angiogenic switch in tumor growth and metastatic progression. Biochim Biophys Acta 1796: 33–40.
Grange C, Bussolati B, Bruno S, Fonsato V, Sapino A, Camussi G . (2006). Isolation and characterization of human breast tumor-derived endothelial cells. Oncol Rep 15: 381–386.
Huang EJ, Nocka KH, Buck J, Besmer P . (1992). Differential expression and processing of two cell associated forms of the kit-ligand: KL-1 and KL-2. Mol Biol Cell 3: 349–362.
Kerbel RS . (2008). Tumor angiogenesis. N Engl J Med 358: 2039–2049.
Leek RD, Harris AL . (2002). Tumor-associated macrophages in breast cancer. J Mammary Gland Biol Neoplasia 7: 177–189.
Lin EY, Li JF, Gnatovskiy L, Deng Y, Zhu L, Grzesik DA et al. (2006). Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res 66: 11238–11246.
Luster AD, Alon R, von Andrian UH . (2005). Immune cell migration in inflammation: present and future therapeutic targets. Nat Immunol 6: 1182–1190.
Mantovani A, Allavena P, Sica A, Balkwill F . (2008). Cancer-related inflammation. Nature 454: 436–444.
Matsuda R, Takahashi T, Nakamura S, Sekido Y, Nishida K, Seto M et al. (1993). Expression of the c-kit protein in human solid tumors and in corresponding fetal and adult normal tissues. Am J Pathol 142: 339–346.
Nishie A, Ono M, Shono T, Fukushi J, Otsubo M, Onoue H et al. (1999). Macrophage infiltration and heme oxygenase-1 expression correlate with angiogenesis in human gliomas. Clinical Cancer Res 5: 1107–1113.
Nolan DJ, Ciarrocchi A, Mellick AS, Jaggi JS, Bambino K, Gupta S et al. (2007). Bone marrow-derived endothelial progenitor cells are a major determinant of nascent tumor neovascularization. Genes Dev 21: 1546–1558.
Ribatti D, Nico B, Crivellato E, Vacca A . (2007). The structure of the vascular network of tumors. Cancer Lett 248: 18–23.
Shchors K, Evan G . (2007). Tumor angiogenesis: cause or consequence of cancer? Cancer Res 67: 7059–7061.
Sica A, Rubino L, Mancino A, Larghi P, Porta C, Rimoldi M et al. (2007). Targeting tumour-associated macrophages. Expert Opin Ther Targets 11: 1219–1229.
St Croix B, Rago C, Velculescu V, Traverso G, Romans KE, Montgomery E et al. (2000). Genes expressed in human tumor endothelium. Science 289: 1197–1202.
Takakura N . (2006). Role of hematopoietic lineage cells as accessory components in blood vessel formation. Cancer Sci 97: 568–574.
Tögel F, Hu Z, Weiss K, Isaac J, Lange C, Westenfelder C . (2005). Administered mesenchymal stem cells protect against ischemic acute renal failure through differentiation-independent mechanisms. Am J Physiol 289: 31–42.
Togliatto G, Trombetta A, Dentelli P, Baragli A, Rosso A, Granata R et al. (2010). Unacylated ghrelin rescues endothelial progenitor cell function in individuals with type 2 diabetes. Diabetes 59: 1016–1025.
Tonini T, Rossi F, Claudio PP . (2003). Molecular basis of angiogenesis and cancer. Oncogene 22: 6549–6556.
Uberti B, Dentelli P, Rosso A, Defilippi P, Brizzi MF . (2010). Inhibition of β1 integrin and IL-3Rβ common subunit interaction hinders tumour angiogenesis. Oncogene 29: 6581–6590.
van Beijnum JR, Petersen K, Griffioen AW . (2009). Tumor endothelium is characterized by a matrix remodeling signature. Front Biosci 1: 216–225.
Vajkoczy P, Blum S, Lamparter M, Mailhammer R, Erber R, Engelhardt B et al. (2003). Multistep nature of microvascular recruitment of ex vivo-expanded embryonic endothelial progenitor cells during tumor angiogenesis. J Exp Med 197: 1755–1765.
Verfaillie CM . (2008). Bony endothelium: tumor-mediated transdifferentiation? Cancer Cell 14: 193–194.
Weber C, Koenen RR . (2006). Fine-tuning leukocyte responses: towards a chemokine ‘interactome’. Trends Immunol 27: 268–273.
Weidner N, Semple JP, Welch WR, Folkman J . (1991). Tumor angiogenesis and metastasis: correlation in invasive breast carcinoma. N Engl J Med 324: 1–7.
Yoder MC, Mead LE, Prater D, Krier TR, Mroueh KN, Li F et al. (2007). Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood 109: 1801–1809.
Yoder MC, Ingram DA . (2009). The definition of EPCs and other bone marrow cells contributing to neoangiogenesis and tumor growth: is there common ground for understanding the roles of numerous marrow-derived cells in the neoangiogenic process? Biochim Biophys Acta 1796: 50–54.
Zeoli A, Dentelli P, Rosso A, Togliatto G, Trombetta A, Damiano L et al. (2008). Interleukin-3 promotes expansion of hemopoietic-derived CD45+ angiogenic cells and their arterial commitment via STAT5 activation. Blood 112: 350–361.
Acknowledgements
This work was supported by grants of the Italian Association for Cancer Research (AIRC) to MFB and GC and by MIUR (Ministero dell’Università e Ricerca Scientifica) to MFB and GC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Dentelli, P., Rosso, A., Olgasi, C. et al. IL-3 is a novel target to interfere with tumor vasculature. Oncogene 30, 4930–4940 (2011). https://doi.org/10.1038/onc.2011.204
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.204
Keywords
This article is cited by
-
Transcriptome of rhesus macaque (Macaca mulatta) exposed to total-body irradiation
Scientific Reports (2021)
-
Nicotinamide inhibits melanoma in vitro and in vivo
Journal of Experimental & Clinical Cancer Research (2020)
-
Real-time metabolic profiling of oesophageal tumours reveals an altered metabolic phenotype to different oxygen tensions and to treatment with Pyrazinib
Scientific Reports (2020)
-
The pro- and anti-tumor roles of mesenchymal stem cells toward BRCA1-IRIS-overexpressing TNBC cells
Breast Cancer Research (2019)
-
A dual role for the N-terminal domain of the IL-3 receptor in cell signalling
Nature Communications (2018)