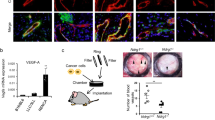
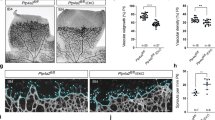
Abstract
Angiogenesis is regulated by highly coordinated function of various proteins with pro- and anti-angiogenic functions. Among the many cytoplasmic signaling proteins that are activated by VEGFR-2, activation of PLCγ1 is considered to have a pivotal role in angiogenic signaling. In previous study we have identified c-Cbl as a negative regulator of PLCγ1 in endothelial cells, the biochemical and biological significance of c-Cbl, however, in angiogenesis in vivo and molecular mechanisms involved were remained elusive. In this study, we report that genetic inactivation of c-Cbl in mice results in enhanced tumor angiogenesis and retinal neovascularization. Endothelial cells derived from c-Cbl null mice displayed elevated cell proliferation and tube formation in response to VEGF stimulation. Loss of c-Cbl also resulted in robust activation of PLCγ1 and increased intracellular calcium release. c-Cbl-dependent ubiquitination selectively inhibited tyrosine phosphorylation of PLCγ1 and mostly refrained from ubiquitin-mediated degradation. Hence, we propose c-Cbl as an angiogenic suppressor protein where upon activation it uniquely modulates PLCγ1 activation by ubiquitination and subsequently inhibits VEGF-driven angiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Adams RH, Alitalo K . (2007). Molecular regulation of angiogenesis and lymphangiogenesis. Nat Rev Mol Cell Biol 8: 464–478.
Fukumura D, Jain RK . (2007). Tumor microvasculature and microenvironment: Targets for anti-angiogenesis and normalization. Microvasc Res 74: 72–84.
Fang D, Liu Y-C . (2001). Proteolysis-independent regulation of PI3K by Cbl-b-mediated ubiquitination in T cells. Nat Immunol 2: 870–875.
Feshchenko EA, Langdon WY, Tsygankov AY . (1998). Fyn, Yes, and Syk phosphorylation sites in c-Cbl map to the same tyrosine residues that become phosphorylated in activated T cells. J Biol Chem 273: 8323–8331.
Folkman J . (1990). What is the evidence that tumors are angiogenesis dependent? J Natl Cancer Inst 82: 4–6.
Funakoshi T, Birsner AE, D'Amato RJ . (2006). Antiangiogenic effect of oral 2-methoxyestradiol on choroidal neovascularization in mice. Exp Eye Res 83: 1102–1107.
Graham LJ, Stoica BA, Shapiro M, DeBell KE, Rellahan B, Laborda J et al. (1998). Sequences surrounding the Src-homology 3 domain of phospholipase C gamma-1 increase the domain's association with Cbl. Biochem Biophys Res Commun 249: 537–541.
Graham LJ, Veri M-C, DeBell KE, Noviello C, Rawat R, Jen S et al. (2003). 70Z/3 Cbl induces PLC gamma1 activation in T lymphocytes via an alternate Lat- and Slp-76-independent signaling mechanism. Oncogene 22: 2493–2503.
Hochstrasser M . (2009). Origin and function of ubiquitin-like proteins. Nature 458: 422–429.
Husain D, Meyer R, Mehta M, Pfeifer WM, Chou E, Navruzbekov G et al. (2010). Role of c-Cbl Dependent Regulation of Phospholipase C gamma 1 Activation in Experimental Choroidal Neovascularization. Invest Ophthalmol Vis Sci 51: 6803–6809.
Ji Q-S, Winnier GE, Niswender KD, Horstman D, Wisdom R, Magnusen MA et al. (1997). Essential role of the tyrosine kinase substrate phospholipase C-gamma1 inmammalian growth and development. Proc Natl Acad Sci USA 94: 2999–3003.
Lawson ND, Mugford JW, Diamond BA, Weinstein BM . (2003). phospholipase C gamma-1 is required downstream of vascular endothelial growth factor during arterial development. Genes Dev 17: 1346–1351.
Liao H-J, Kume T, McKay C, Xu M-J, Ihle JN, Carpenter G . (2002). Absence of erythrogenesis and vasculogenesis in Plc gamma-deficient mice. J Biol Chem 277: 9335–9341.
Meyer RD, Latz C, Rahimi N . (2003). Recruitment and activation of PLCγ1 by vascular endothelial growth factor receptor-2 are required for tubulogenesis and differentiation of endothelial cells. J Biol Chem 278: 16347–16355.
Meyer RD, Sacks DB, Rahimi N . (2008). IQGAP1-dependent signaling pathway regulates endothelial cell proliferation and angiogenesis. PLoS One 3: e3848.
Meyer RD, Singh A, Majnoun F, Latz C, Lashkari K, Rahimi N . (2004). Substitution of C-terminus of VEGFR-2 with VEGFR-1 promotes VEGFR-1 activation and endothelial cell proliferation. Oncogene 23: 5523–5531.
Miura-Shimura Y, Duan L, Rao NL, Reddi AL, Shimura H, Rottapel R et al. (2003). Cbl-mediated ubiquitinylation and negative regulation of Vav. J Biol Chem 278: 38495–38504.
Miyazaki T, Sanjay A, Neff L, Tanaka S, Horne WC, Baron R . (2004). Src kinase activity is essential for osteoclast function. J Biol Chem 279: 17660–17666.
Murphy MA, Schnall RG, Venter DJ, Barnett L, Bertoncello I, Thien CB et al. (1998). Tissue hyperplasia and enhanced T-cell signalling via ZAP-70 in c-Cbl-deficient mice. Mol Cell Biol 18: 4872–4882.
Naramura M, Jang IK, Kole H, Huang F, Haines D, Gu H . (2002). c-Cbl and Cbl-b regulate T cell responsiveness by promoting ligand-induced TCR down-modulation. Nat Immunol 3: 1192–1199.
Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L . (2006). VEGF receptor signalling - in control of vascular function. Nat Rev Mol Cell Biol 7: 359–371.
Rahimi N . (2006). VEGFR-1 and VEGFR-2: two non-identical twins with a unique physiognomy. Front Biosci 11: 818–829.
Rahimi N, Dayanir V, Lashkari K . (2000). Receptor chimeras indicate that the vascular endothelial growth factor receptor-1 (VEGFR-1) modulates mitogenic activity of VEGFR-2 in endothelial cells. J Biol Chem 275: 16986–16992.
Rahimi N, Golde TE, Meyer RD . (2009). Identification of ligand-induced proteolytic cleavage and ectodomain shedding of VEGFR-1/FLT1 in leukemic cancer cells. Cancer Res 69: 2607–2614.
Ravid T, Hochstrasser M . (2008). Diversity of degradation signals in the ubiquitin–proteasome system. Nat Rev Mol Cell Biol 9: 679–690.
Rebecchi MJ, Pentyala SN . (2000). Structure, function, and control of phosphoinositide-specific phospholipase C. Physiol Rev 80: 1291–1335.
Sakurai Y, Ohgimoto K, Kataoka Y, Yoshida N, Shibuya M . (2005). Essential role of Flk-1 (VEGF receptor 2) tyrosine residue 1173 in vasculogenesis in mice. Proc Natl Acad Sci USA 102: 1076–1081.
Schwartz AL, Ciechanover A . (2009). Targeting proteins for destruction by the ubiquitin system: implications for human pathobiology. Annu Rev Pharmacol Toxicol 49: 73–79.
Singh AJ, Meyer RD, Band H, Rahimi N . (2005). The carboxyl terminus of VEGFR-2 is required for PKC-mediated downregulation. Mol Biol Cell 16: 2106–2118.
Singh AJ, Meyer RD, Navruzbekov G, Shelke R, Duan L, Band H et al. (2007). A critical role for the E3-ligase activity of c-Cbl in VEGFR-2-mediated PLCgamma1 activation and angiogenesis. Proc Natl Acad Sci USA 104: 5413–5418.
Swaminathan G, Tsygankov AY . (2006). The Cbl family proteins: ring leaders in regulation of cell signaling. J Cell Physiol 209: 21–43.
Takahashi T, Yamaguchi S, Chida K, Shibuya M . (2001). A single autophosphorylation site on KDR/Flk-1 is essential for VEGF-A-dependent activation of PLCγ1 and DNA synthesis in vascular endothelial cells. EMBO J 20: 2768–2778.
Teckchandani AM, Panetti TS, Tsygankov AY . (2005). c-Cbl regulates migration of v-Abl-transformed NIH 3T3 fibroblasts via Rac1. Exp Cell Res 307: 247–258.
Thien CBF, Langdon WY . (2005). c-Cbl and Cbl-b ubiquitin ligases: substrate diversity and negative regulation of signaling responses. Biochem J 391: 153–165.
Woodman SE, Ashton AW, Schubert W, Lee H, Williams TM, Medina FA et al. (2003). Caveolin-1 knockout mice show an impaired angiogenic response to exogenous stimuli. Am J Pathol 162: 2059–2068.
Zeng H, Qin L, Zhao D, Tan X, Manseau EJ, Van Hoang M et al. (2006). Orphan nuclear receptor TR3/Nur77 regulates VEGF-A-induced angiogenesis through its transcriptional activity. J Exp Med 203: 719–729.
Zhang H, He Y, Dai S, Xu Z, Luo Y, Wan T et al. (2008). AIP1 functions as an endogenous inhibitor of VEGFR2-mediated signaling and inflammatory angiogenesis in mice. J Clin Invest 118: 3904–3916.
Acknowledgements
This study was supported by grants from the National Institutes of Health (NIH/NEI) to NR and Massachusetts Lions Foundation grant to Department of Ophthalmology, and also by BU CTSI grant (NR). The c-Cbl knockout mice were generously provided by Jeffrey Chiang (National Cancer Institute, Frederick, MD).Author contribution: Rosana D Meyer, Deeba Husain and Nader Rahimi all performed experiments. Nader Rahimi wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Meyer, R., Husain, D. & Rahimi, N. c-Cbl inhibits angiogenesis and tumor growth by suppressing activation of PLCγ1. Oncogene 30, 2198–2206 (2011). https://doi.org/10.1038/onc.2010.597
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.597
Keywords
This article is cited by
-
Mechanisms and regulation of endothelial VEGF receptor signalling
Nature Reviews Molecular Cell Biology (2016)